Abstract
Main conclusion
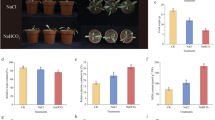
The comparison of the changes of the lipid content in plant cell boundary membranes demonstrates a substantial role of the vacuolar membrane in response to hyperosmotic stress.
Abstract
Comparison of variations in the lipid content of plant cell boundary membranes (vacuolar and plasma membranes) isolated from beet root tissues (Beta vulgaris L.) was conducted after the effect of hyperosmotic stress. Both types of membranes participate in the formation of protective mechanisms, but the role of the vacuolar membrane was considered as more essential. This conclusion was connected with more significant adaptive variations in the content and composition of sterols and fatty acids in the vacuolar membrane (although some of the adaptive variations, especially, in the composition of phospholipids and glycoglycerolipids were similar for both types of membranes). In the plasma membrane under hyperosmotic stress, the increase in the content of sphingolipids was noted that was not observed in the tonoplast.
Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
Abbreviations
- DGDG:
-
Digalactosyldiacylglycerol
- FA:
-
Fatty acid
- GL:
-
Glycoglycerolipid
- MGDG:
-
Monogalactosyldiacylglycerol
- PC:
-
Phosphatidylcholine
- PE:
-
Phosphatidylethanolamine
- SFA:
-
Saturated fatty acid
- USFA:
-
Unsaturated fatty acid
References
Alsher RG, Donahue JL, Cramer CL (1997) Reactive oxygen species and antioxidants: relationships in green cells. Physiol Plant 100:224–233. https://doi.org/10.1111/j.1399-3054.1997.tb04778.x
Bohn M, Luthje S, Sperling P, Heinz E, Dorffling K (2007) Plasma membrane lipid alterations induced by cold acclimation and abscisic acid treatment of winter wheat seedlings differing in frost resistance. J Plant Physiol 164(2):146–156. https://doi.org/10.1016/j.jplph.2005.12.008
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Carmona-Salazar L, Cahoon RE, Gasca-Pineda J, González-Solís A, Vera-Estrella R, Treviño V, Cahoon BE, Gavilanes-Ruiz M (2021) Plasma and vacuolar membrane sphingolipidomes: composition and insights on the role of main molecular species. Plant Physiol 186(1):624–636. https://doi.org/10.1093/plphys/kiab064
Cassim AM, Gouguet P, Gronnier J, Laurent N, Germain V, Grison M, Boutté Y, Gerbeau-Pissot P, Simon-Plas F, Mongrand S (2019) Plant lipids: key players of plasma membrane organization and function. Prog Lipid Res 73:1–27. https://doi.org/10.1016/j.plipres.2018.11.002
Cassim AM, Grison M, Ito Y, Simon-Plas F, Mongrand S, Boutté Y (2020) Sphingolipids in plants: a guidebook on their function in membrane architecture, cellular processes, and environmental or developmental responses. FEBS Lett 594:3719–3738
Christie W (1988) Equivalent chain lengths of methyl ester derivatives of fatty acids on gas chromatography: a reappraisal. J Chromatogr A 447:305–314. https://doi.org/10.1016/0021-9673(88)90040-4
Folch J, Lees M, Sloan Stanley GH (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226:497–509. https://doi.org/10.1016/S0021-9258(18)64849-5
Fujimotoat M, Hayashi T, Su TP (2012) The role of cholesterol in the association of endoplasmic reticulum membranes with mitochondria. Biochem Biophys Res Commun 417:635–639. https://doi.org/10.1016/j.bbrc.2011.12.022
Gigon A, Matos A-R, Laffray D, Zuily-Fodil Y, Pham-Thi A-T (2004) Effect of drought on lipid metabolism in the leaves of Arabidopsis thaliana (ecotype Columbia). Ann Bot 94:345–351. https://doi.org/10.1093/aob/mch150
Guo Q, Liu L, Rupasinghe TWT, Roessner U, Barkla BJ (2022) Salt stress alters membrane lipid content and lipid biosynthesis pathways in the plasma membrane and tonoplast. Plant Physiol 189:805–826. https://doi.org/10.1093/plphys/kiac123
Hartmann MA (1998) Plant sterols and the membrane environment. Trends Plant Sci 3:170–175. https://doi.org/10.1016/S1360-1385(98)01233-3
Hodzic A, Rappolt M, Amenitsch H, Laggner P, Pabst G (2008) Differential modulation of membrane structure and fluctuations by plant sterols and cholesterol. Biophys J 94(10):3935–3944. https://doi.org/10.1529/biophysj.107.123224
Kates M (1972) Techniques of lipidology. In: Work TS, Work E (eds) Laboratory techniques in biochemistry and molecular biology. North-Holland Publ, North-Holland
Kerkeb L, Donaire JP, Venema K, Rodríguez-Rosales MP (2001) Tolerance to NaCl induces changes in plasma membrane lipid composition, fluidity and H+-ATPase activity of tomato calli. Physiol Plant 113:217–224. https://doi.org/10.1034/j.1399-3054.2001.1130209.x
Kumar M, Ali K, Dahuia A, Tyagi A (2015) Role of phytosterols in drought stress tolerance in rice. Plant Physiol Biochem 96:83–89. https://doi.org/10.1016/j.plaphy.2015.07.014
Larsson C, Widell S, Kjellbon P (1987) Preparation of high-purity plasma membranes. Methods Enzymol 14:558–568. https://doi.org/10.1016/0076-6879(87)48054-3
Michaelson LV, Napier JA, Molino D, Faure JD (2016) Plant sphingolipids: their importance in cellular organization and adaptation. Biochim Biophys Acta 1861:1329–1335. https://doi.org/10.1016/j.bbalip.2016.04.003. (Epub 2016 Apr 13)
Mongrand S, Morel J, Laroche J, Claverol S, Carde J-P, Hartmann M-A, Bonneu M, Simon-Plas F, Lessire R, Bessoule J-J (2004) Lipid rafts in higher plant cells. J Biol Chem 279:36277–36286. https://doi.org/10.1074/jbc.M403440200
Narayanan S, Tamura PJ, Roth MR, Vara Prasad PV, Welti R (2016) Wheat leaf lipid composition during heat stress: I. High day and night temperatures result in major lipid alterations. Plant Cell Environm 39:787–803. https://doi.org/10.1111/pce.12649
Nurminsky VN, Ozolina NV, Nesterkina IS, Kolesnikova EV, Korzun AM, Chernyshov MY, Tikhonov NV, Tarkov MS, Salyaev RK (2011) Stability of plant vacuolar membranes under the conditions of osmotic stress and influence of redox agents. Biochem (Moscow). Suppl SER A Membr Cell Biol 5:185–190
Okazaki Y, Saito K (2014) Roles of lipids as signaling molecules and mitigators during stress response in plants. Plant J 79:584–596. https://doi.org/10.1111/tpj.12556
Omoto E, Iwasaki Y, Miyaki H, Taniguchi M (2016) Salinity induces membrane structure and lipid changes in maize mesophyll and bundle sheath chloroplasts. Physiol Plant 157(1):13–23. https://doi.org/10.1111/ppl.12404
Orvar BL, Sangwan V, Omann F, Dhindsa RS (2000) Early steps in cold sensing by plant cells: the role of actin cytoskeleton and membrane fluidity. Plant J 23:785–794. https://doi.org/10.1046/j.1365-313x.2000.00845.x
Ozolina NV, Gurina VV, Nesterkina IS, Nurminsky VN (2020) Variations in the content of tonoplast lipids under abiotic stress. Planta 251:107. https://doi.org/10.1007/s00425-020-03399-x
Pham-Thi A-T, Borrel-Flood C, Veira da Silva J, Justin AM, Mazliak P (2006) Effects of drought on [1-14C]-oleic and [1-14C]-linoleic acid desaturation in cotton leaves. Physiol Plant 69:147–150. https://doi.org/10.1111/j.1399-3054.1987.tb01958.x
Reszczynska E, Hanaka A (2020) Lipids composition in plant membranes. Cell Biochem Biophys 78:401–414. https://doi.org/10.1007/s12013-020-00947-w
Ristic Z, Ashworth EN (1993) Changes in leaf ultrastructure and carbohydrates in Arabidopsis thaliana L. (Heyn) cv. Columbia during rapid cold acclimation. Protoplasma 172:111–123. https://doi.org/10.1007/BF01379368
Salyaev RK, Kuzevanov VYA, Khaptagaev SB, Kopytchuk VN (1981) Isolation and purification of vacuoles and vacuolar membranes from plant cells. Russ J Plant Physiol (russ.) 28:1295–1305
Shuler I, Milon A, Nakatani Y, Ourisson G, Albrecht AM, Benveniste P, Hartman MA (1991) Differential effects of plant sterols on water permeability and on acyl chain phosphatidylcholine bilayers. Proc Natl Acad Sci USA 88:6926–6930. https://doi.org/10.1073/pnas.88.16.6926
Su K, Bremer DJ, Jeannotte R, Welti R, Yang C (2009) Membrane lipid composition and heat tolerance in cool-season turgrasses, including a hybrid bluegrass. J Am Soc Hortic Sci 134:511–520. https://doi.org/10.21273/JASHS.134.5.511
Valitova JN, Sulkarnayeva AG, Minibaeva FV (2016) Plant sterols: diversity, biosynthesis and phusiological functions. Biochem Mosc 18:1050–1068. https://doi.org/10.1134/S0006297916080046
Vaskovsky VE, Latyshev NA (1975) Modified Jungnickel’s reagent for detecting phospholipids and other phosphorus compounds on thin-layer chromatograms. J Chromatogr 115:246–249. https://doi.org/10.1016/s0021-9673(00)89042-1
Verhoek B, Haas R, Wrage K, Linscheid M, Heinz F (1983) Lipids and enzymatic activities in vacuolar membranes isolated via protoplasts from oat primary leaves. Z Naturforschg 38:770–777
Vivancos M, Moreno JJ (2005) Beta-isosterol modulates antioxidant enzyme response in RAW 264.7 macrophages. Free Radic Biol Med 39:91–97. https://doi.org/10.1016/j.freeradbiomed.2005.02.025
Wang T, Hicks KB, Moreau R (2002) Antioxidant activity of photosterols, oryzanol, and other phytosterols conjugates. J Am Oil Chem Soc 7:1201–1206. https://doi.org/10.1007/s11746-002-0628-x
Wegener A, Gimbel W, Werner T, Hani J, Ernst D, Sandermann H (1997) Molecular cloning of ozone-inducible protein from Pinus sylvestris L. with high sequence similarity to vertebrate 3-hydroxy-3-methylglutaryl-CoA-syntase. Biochim Biophys Acta 1350:247–252. https://doi.org/10.1016/s0005-2760(96)00161-0
Wu J, Seliskar DM, Gallagher JL (2005) The response of plasma membrane lipid composition in callus of the halophyte Spartina patens (Poaceae) to salinity stress. Am J Bot 92:852–858. https://doi.org/10.3732/ajb.92.5.852
Yamaguchi M, Kasamo K (2001) Modulation in the activity of purified tonoplast H+-ATPase by tonoplast glycolipids prepared from cultured rice (Oryza sztiva L. var. Boro) cells. Plant Cell Physiol 42(5):516–523. https://doi.org/10.1093/pcp/pce064
YuL ZC, Fan J, Shanklin J, Xu C (2021) Mechanisms and functions of membrane lipid remodeling in plants. Plant J 107(1):37–53. https://doi.org/10.1111/tpj.15273
Zhigacheva IV, Burlakova EB, Misharina TA, Terenina MB, Krikunova NI, Generozova IP, Shugaev AG, Fattakhov SG (2013) Fatty acid composition of membrane lipids and energy metabolism in mitochondria of pea seedlings under water deficit. Russ J Plant Physiol 60:205–213. https://doi.org/10.1134/S1607672911020104
Zhou Y, Pan X, Qu H, Underhill SJ (2014) Low temperature alters plasma membrane lipid composition and ATPase activity of pineapple fruit during blackheart development. J Bioenerg Biomembr 46:59–69. https://doi.org/10.1007/s10863-013-9538-4
Nikulina GN (1965) Survey of the methods of quantitative analysis of phosphorus with formation of molybdenum blue. (Russ.) Nauka Publ.
Vladimirov YuA, Archakov AI, Frank GM (1972) Lipid peroxidation in biological membranes (Russ.). Nauka Publ.
Acknowledgements
This investigation was conducted with the use of the equipment of Central Analytical Center “Bioanalytics” of Siberian Institute of Plant Physiology and Biochemistry (Siberian Branch of Russian Academy of Sciences, Irkutsk) within the framework of the project supported by state budget financing (state registration number 122041100052-0).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare no conflicts of interests.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ozolina, N.V., Kapustina, I.S., Gurina, V.V. et al. Comparison of the functions of plasma membrane and vacuolar membrane lipids in plant cell protection against hyperosmotic stress. Planta 258, 39 (2023). https://doi.org/10.1007/s00425-023-04191-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-023-04191-3