Abstract
Main conclusion
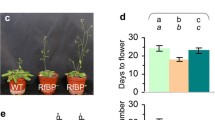
Our findings suggested that ClWRKY48 promoted the expression level of Arabidopsis phosphate transporter genes, enhanced phosphate uptake, and delayed the transition from the vegetative stage to the reproductive phase in Arabidopsis.
Abstract
Phosphorus (P) is an essential mineral for plants that influences their growth and development. ClWRKY48, one of the most highly expressed genes in the leaf, was identified by RT-PCR from Chinese fir [Cunninghamia lanceolata (Lamb.) Hook] (C. lanceolata). Furthermore, when treating C. lanceolata with increasing phosphate (Pi) concentration, the expression level of ClWRKY48 rose in leaves, the trends followed the increasing phosphate concentration treatment. ClWRKY48 is a transcription factor in C. lanceolata, according to the results of a yeast one hybridization experiment. Based on subcellular localization studies, ClWRKY48 is a nuclear-localized protein. Under Pi deficiency conditions, the phosphorus concentration of ClWRKY48 overexpressing Arabidopsis increased by 43.2–51.1% compared to the wild-type. Moreover, under Pi limiting conditions, the phosphate transporter genes AtPHT1;1 (Arabidopsis Phosphate transporter 1;1), AtPHT1;4, and AtPHO1 (Arabidopsis PHOSPHATE 1) were expressed 2.1–2.5, 2.2–2.7, and 6.7–7.3-fold greater than the wild-type in ClWRKY48 transgenic Arabidopsis, respectively. Under Pi-sufficient conditions, the phosphorus concentration and phosphate transporter genes of ClWRKY48 overexpression in Arabidopsis are not significantly different from the wild type. These findings indicated that ClWRKY48 increased phosphate absorption in transgenic Arabidopsis. Furthermore, compared to the wild type, the ClWRKY48 transgenic Arabidopsis not only had a delayed flowering time characteristic but also had lower expression of flowering-related genes AtFT (FLOWERING LOCUS T), AtFUL (FRUITFUL), and AtTSF (TWIN SISTER OF FT). Our findings show that ClWRKY48 enhances phosphate absorption and slows the transition from the vegetative to the reproductive stage in ClWRKY48 transgenic Arabidopsis.






Similar content being viewed by others
Data availability
All the data generated or analyzed during this study are included in this published article.
Abbreviations
- Ade:
-
Adenine
- FT :
-
FLOWERING LOCU S T
- FUL :
-
FRUITFUL
- GFP:
-
Green fluorescence protein
- His:
-
Histidine
- PHO1:
-
PHOSPHATE 1
- PHT:
-
Phosphate transporter
- Pi:
-
Phosphate
- SD:
-
Yeast synthetic dropout medium
- TF:
-
Transcription factor
- Trp:
-
Tryptophane
- TSF :
-
TWIN SISTER OF FT
References
Arpat AB, Magliano P, Wege S, Rouached H, Stefanovic A, Poirier Y (2012) Functional expression of PHO1 to the Golgi and trans-Golgi network and its role in export of inorganic phosphate. Plant J 71(3):479–491. https://doi.org/10.1111/j.1365-313X.2012.05004.x
Ayadi A, David P, Arrighi JFO, Chiarenza S, Thibaud MC, Nussaume L, Marin E (2015) Reducing the genetic redundancy of Arabidopsis PHOSPHATE TRANSPORTER1 transporters to study phosphate uptake and signaling. Plant Physiol 167(4):1511–1526. https://doi.org/10.1104/pp.114.252338
Baek D, Chun HJ, Yun DJ, Kim MC (2017) Cross-talk between phosphate starvation and other environmental stress signaling pathways in plants. Mol Cells 40(10):697–705. https://doi.org/10.14348/molcells.2017.0192
Bao W, Qu Y, Shan X, Wan Y (2016) Screening and validation of housekeeping genes of the root and cotyledon of Cunninghamia lanceolata under abiotic stresses by using quantitative real-time PCR. Int J Mol Sci. https://doi.org/10.3390/ijms17081198
Bustos R, Castrillo G, Linhares F, Puga MI, Rubio V, Perez-Perez J, Solano R, Leyva A, Paz-Ares J (2010) A central regulatory system largely controls transcriptional activation and repression responses to phosphate starvation in Arabidopsis. PLOS Genet 6(9):e1001102. https://doi.org/10.1371/journal.pgen.1001102
Ceulemans T, Bode S, Bollyn J, Harpole S, Coorevits K, Peeters G, Van Acker K, Smolders E, Boeckx P, Honnay O (2017) Phosphorus resource partitioning shapes phosphorus acquisition and plant species abundance in grasslands. Nat Plants 3(2):16224. https://doi.org/10.1038/nplants.2016.224
Chen YF, Li LQ, Xu Q, Kong YH, Wang H, Wu WH (2009) The WRKY6 transcription factor modulates PHOSPHATE1 expression in response to low Pi stress in Arabidopsis. Plant Cell 21(11):3554–3566. https://doi.org/10.1105/tpc.108.064980
Chen J, Nolan TM, Ye H, Zhang M, Tong H, Xin P, Chu J, Chu C, Li Z, Yin Y (2017) Arabidopsis WRKY46, WRKY54, and WRKY70 transcription factors are involved in brassinosteroid-regulated plant growth and drought responses. Plant Cell 29(6):1425–1439. https://doi.org/10.1105/tpc.17.00364
Chen F, Hu Y, Vannozzi A, Wu K, Cai H, Qin Y, Mullis A, Lin Z, Zhang L (2018) The WRKY transcription factor family in model plants and crops. Crit Rev Plant Sci 36(5–6):311–335. https://doi.org/10.1080/07352689.2018.1441103
Chiou TJ, Lin SI (2011) Signaling network in sensing phosphate availability in plants. Annu Rev Plant Biol 62:185–206. https://doi.org/10.1146/annurev-arplant-042110-103849
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium -mediated transformation of Arabidopsis thaliana. Plant J 16(6):735–743. https://doi.org/10.1046/j.1365-313x.1998.00343.x
Dai X, Wang Y, Zhang WH (2016) OsWRKY74, a WRKY transcription factor, modulates tolerance to phosphate starvation in rice. J Exp Bot 67(3):947–960. https://doi.org/10.1093/jxb/erv515
Devaiah BN, Karthikeyan AS, Raghothama KG (2007) WRKY75 transcription factor is a modulator of phosphate acquisition and root development in Arabidopsis. Plant Physiol 143(4):1789–1801. https://doi.org/10.1104/pp.106.093971
Dou L, Zhang X, Pang C, Song M, Wei H, Fan S, Yu S (2014) Genome-wide analysis of the WRKY gene family in cotton. Mol Genet Genom 289(6):1103–1121. https://doi.org/10.1007/s00438-014-0872-y
Eulgem T, Rushton PJ, Robatzek S, Somssich IE (2000) The WRKY superfamily of plant transcription factors. Trends Plant Sci 5(5):199–206. https://doi.org/10.1016/s1360-1385(00)01600-9
Gao K, Khan WU, Li J, Huang S, Yang X, Guo T, Guo B, Wu RQ, An XM (2022) Identification and validation of reliable reference genes for gene expression studies in koelreuteria paniculate. Genes 13(714):1–12. https://doi.org/10.3390/genes13050714
Gonzalez D, Postma J, Wissuwa M (2021) Cost-benefit analysis of the Upland-Rice root architecture in relation to phosphate: 3D simulations highlight the importance of S-Type lateral roots for reducing the Pay-Off time. Front Plant Sci 12:641835. https://doi.org/10.3389/fpls.2021.641835
Guo C, Guo R, Xu X, Gao M, Li X, Song J, Zheng Y, Wang X (2014) Evolution and expression analysis of the grape (Vitis vinifera L.) WRKY gene family. J Exp Bot 65(6):1513–1528. https://doi.org/10.1093/jxb/eru007
Hall BG (2013) Building phylogenetic trees from molecular data with MEGA. Mol Biol Evol 30(5):1229–1235. https://doi.org/10.1093/molbev/mst012
He H, Dong Q, Shao Y, Jiang H, Zhu S, Cheng B, Xiang Y (2012) Genome-wide survey and characterization of the WRKY gene family in Populus trichocarpa. Plant Cell Rep 31(7):1199–1217. https://doi.org/10.1007/s00299-012-1241-0
Huang S, Gao Y, Liu J, Peng X, Niu X, Fei Z, Cao S, Liu Y (2012) Genome-wide analysis of WRKY transcription factors in Solanum lycopersicum. Mol Genet Genom 287(6):495–513. https://doi.org/10.1007/s00438-012-0696-6
Ishiguro S, Nakamura K (1994) Characterization of a cDNA encoding a novel DNA-binding protein, SPF1, that recognizes SP8 sequences in the 5′ upstream regions of genes coding for sporamin and β-amylase from sweet potato. Mol Gene Genet 244(6):563–571. https://doi.org/10.1007/BF00282746
Jiang J, Ma S, Ye N, Jiang M, Cao J, Zhang J (2017) WRKY transcription factors in plant responses to stresses. J Integ Plant Biol 59(2):86–101. https://doi.org/10.1111/jipb.12513
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Evolution Genet Analy 33(7):1870–1874. https://doi.org/10.1093/molbev/msw054
Li W, Wang H, Yu D (2016) Arabidopsis WRKY transcription factors WRKY12 and WRKY13 oppositely regulate flowering under short-day conditions. Mol Plant 9(11):1492–1503. https://doi.org/10.1016/j.molp.2016.08.003
Liu J, Yang L, Luan M, Wang Y, Zhang C, Zhang B, Shi J, Zhao FG, Lan W, Luan S (2015) A vacuolar phosphate transporter essential for phosphate homeostasis in Arabidopsis. PNAS 112(47):E6571–E6578. https://doi.org/10.1073/pnas.1514598112
Nam M, Lee JH, Choi HS, Lim HS, Moon JS, Lee SH (2013) Complete genome sequence of keunjorong mosaic virus, a potyvirus from Cynanchum wilfordii. Arch Virol 158(8):1817–1820. https://doi.org/10.1007/s00705-013-1652-4
Paz-Ares J, Puga MI, Rojas-Triana M, Martinez-Hevia I, Diaz S, Poza-Carrion C, Minambres M, Leyva A (2022) Plant adaptation to low phosphorus availability: core signaling, crosstalks, and applied implications. Mol Plant 15(1):104–124. https://doi.org/10.1016/j.molp.2021.12.005
Raghothama KG (1999) Phosphate acquisition. Annu Rev Plant Physiol Plant Mol Biol 50:665–693. https://doi.org/10.1146/annurev.arplant.50.1.665
Rushton PJ, Somssich IE, Ringler P, Shen QJ (2010) WRKY transcription factors. Trends Plant Sci 15(5):247–258. https://doi.org/10.1016/j.tplants.2010.02.006
Schachtman DP, Reid RJ, Ayling SM (1998) Phosphorus uptake by plants: from soil to cell. Plant Physiol 116(2):447–453. https://doi.org/10.1104/pp.116.2.447
Schmutz J, Cannon SB, Schlueter J, Ma J, Mitros T, Nelson W, Hyten DL, Song Q, Thelen JJ, Cheng J, Xu D, Hellsten U, May GD, Yu Y, Sakurai T, Umezawa T, Bhattacharyya MK, Sandhu D, Valliyodan B, Lindquist E, Peto M, Grant D, Shu S, Goodstein D, Barry K, Futrell-Griggs M, Abernathy B, Du J, Tian Z, Zhu L, Gill N, Joshi T, Libault M, Sethuraman A, Zhang XC, Shinozaki K, Nguyen HT, Wing RA, Cregan P, Specht J, Grimwood J, Rokhsar D, Stacey G, Shoemaker RC, Jackson SA (2010) Genome sequence of the palaeopolyploid soybean. Nature 463(7278):178–183. https://doi.org/10.1038/nature08670
Song Y, Ai CR, Jing SJ, Yu DQ (2010) Research progress on functional analysis of rice WRKY genes. Rice Sci 17(1):60–72. https://doi.org/10.1016/s1672-6308(08)60105-5
Su T, Xu Q, Zhang FC, Chen Y, Li LQ, Wu WH, Chen YF (2015) WRKY42 modulates phosphate homeostasis through regulating phosphate translocation and acquisition in Arabidopsis. Plant Physiol 167(4):1579–1591. https://doi.org/10.1104/pp.114.253799
Tiziani R, Mimmo T, Valentinuzzi F, Pii Y, Celletti S, Cesco S (2020) Root handling affects carboxylates exudation and phosphate uptake of white lupin roots. Front Plant Sci 11:584568. https://doi.org/10.3389/fpls.2020.584568
Wang H, Xu Q, Kong YH, Chen Y, Duan JY, Wu WH, Chen YF (2014) Arabidopsis WRKY45 transcription factor activates PHOSPHATE TRANSPORTER1;1 expression in response to phosphate starvation. Plant Physiol 164(4):2020–2029. https://doi.org/10.1104/pp.113.235077
Wei Y, Shi H, Xia Z, Tie W, Ding Z, Yan Y, Wang W, Hu W, Li K (2016) Genome-Wide identification and expression analysis of the WRKY gene family in cassava. Front Plant Sci 7:25. https://doi.org/10.3389/fpls.2016.00025
Wendrich JR, Yang BJ, Vandamme N, Verstaen K, Smet W, de Velde CV, Minne M, Wybouw B, Mor E, Arents HE, Nolf J, Duyse JV, Isterdael GV, Maere S, Saeys Y, De Rybel B (2020) Vascular transcription factors guide plant epidermal responses to limiting phosphate conditions. Science 370(6518):1–12. https://doi.org/10.1126/science.aay4970
Yamasaki K, Kigawa T, Watanabe S, Inoue M, Yamasaki T, Seki M, Shinozaki K, Yokoyama S (2012) Structural basis for sequence-specific DNA recognition by an Arabidopsis WRKY transcription factor. J Biol Chem 287(10):7683–7691. https://doi.org/10.1074/jbc.M111.279844
Yang X, Zhang K, Nvsvrot T, Zhang Y, Cai G, Huang L, Ren W, Ding Y, Hammond JP, Shi L, Wang N (2022) Phosphate (Pi) stress-responsive transcription factors PdeWRKY6 and PdeWRKY65 regulate the expression of PdePHT1;9 to modulate tissue Pi concentration in poplar. Plant J 111(6):1753–1767. https://doi.org/10.1111/tpj.15922
Yu Y, Liu Z, Wang L, Kim S-G, Seo PJ, Qiao M, Wang N, Li S, Cao X, Park C-M, Xiang F (2016) WRKY71 accelerates flowering via the direct activation of FLOWERING LOCUS T and LEAFY in Arabidopsis thaliana. Plant J 85(1):96–106. https://doi.org/10.1111/tpj.13092
Zhang L, Chen L, Yu D (2017) Transcription factor WRKY75 interacts with DELLA proteins to affect flowering. Plant Physiol 176(1):790–803. https://doi.org/10.1104/pp.17.00657
Zhang J, Gu M, Liang R, Shi X, Chen L, Hu X, Wang S, Dai X, Qu H, Li H, Xu G (2021) OsWRKY21 and OsWRKY108 function redundantly to promote phosphate accumulation through maintaining the constitutive expression of OsPHT1;1 under phosphate-replete conditions. New Phytol 229(3):1598–1614. https://doi.org/10.1111/nph.16931
Acknowledgements
This work was supported by the National Natural Science Foundation of China (32171767), the Scientific Research Project Foundation of the Education Department of Hunan Province of China (21B0252), the Natural Science Foundation of Hunan Province of China (2020JJ5975), and the Undergraduate Students’ Research and Innovative Experiment Program of Hunan Province of China (2022-2799).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest with this manuscript.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tang, W., Wang, J., Lv, Q. et al. Overexpression of ClWRKY48 from Cunninghamia lanceolata improves Arabidopsis phosphate uptake. Planta 257, 87 (2023). https://doi.org/10.1007/s00425-023-04120-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-023-04120-4