Abstract
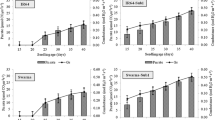
In Asian cultivated rice (Oryza sativa), distinct mechanisms to survive flooding are activated in two groups of varieties. Submergence-tolerant rice varieties possessing the SUBMERGENCE1A (SUB1A) gene display reduced growth during flash floods at the seedling stage and resume growth after the flood recedes, whereas deepwater rice varieties possessing the SNORKEL1 (SK1) and SNORKEL2 (SK2) genes display enhanced growth based on internodal elongation during prolonged submergence at the mature stage. In this study, we investigated the occurrence of these growth responses to submergence in the wild rice species Oryza grandiglumis, which is native to the Amazon floodplains. When subjected to gradual submergence, adult plants of O. grandiglumis accessions showed enhanced internodal elongation with rising water level and their growth response closely resembled that of deepwater varieties of O. sativa with high floating capacity. On the other hand, when subjected to complete submergence, seedlings of O. grandiglumis accessions displayed reduced shoot growth and resumed normal growth after desubmergence, similar to the response of submergence-tolerant varieties of O. sativa. Neither SUB1A nor the SK genes were detected in the O. grandiglumis accessions. These results indicate that the O. grandiglumis accessions are capable of adapting successfully to flooding by activating two contrasting mechanisms as the situation demands and that each mechanism of adaptation to flooding is not mediated by SUB1A or the SK genes.








Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- ERF:
-
Ethylene response factor
- FR13A:
-
Flood Resistant 13A
- GA:
-
Gibberellin
- HA II:
-
Habiganj Aman II
- QTL:
-
Quantitative trait locus
- SUB1:
-
SUBMERGENCE1
- SK1:
-
SNORKEL1
- SK2:
-
SNORKEL2
- TN1:
-
Taichung Native 1
References
Akimoto M, Shimamoto Y, Morishima H (1998) Population genetic structure of wild rice Oryza glumaepatula distributed in the Amazon flood area influenced by its life-history traits. Mol Ecol 7:1371–1381
Azuma T, Hirano T, Deki Y, Uchida N, Yasuda T, Yamaguchi T (1995) Involvement of the decrease in levels of abscisic acid in the internodal elongation of submerged floating rice. J Plant Physiol 146:323–328
Azuma T, Ueno S, Uchida N, Yasuda T (1997) Gibberellin-induced elongation and osmoregulation in internodes of floating rice. Physiol Plant 99:517–522
Azuma T, Uchida N, Yasuda T (2001) Low levels of oxygen promote intermodal elongation in floating rice independently of enhanced ethylene production. Plant Growth Regul 34:181–186
Bailey-Serres J, Voesenek LACJ (2008) Flooding stress: acclimations and genetic diversity. Annu Rev Plant Biol 59:313–339
Bailey-Serres J, Fukao T, Ronald P, Ismail A, Heuer S, Mackill D (2010) Submergence tolerant rice: SUB1’s journey from landrace to modern cultivar. Rice 3:138–147
Bleecker AB, Schuette JL, Kende H (1986) Anatomical analysis of growth and developmental patterns in the internodal of deepwater rice. Planta 169:490–497
Catling D (1992) Rice in deep water. MacMillan Press, London and Basingstoke
Choi D (2011) Molecular events underlying coordinated hormone action in submergence escape response of deepwater rice. J Plant Biol 54:365–372
Cox MC, Peeters AJM, Voesenek LACJ (2006) The stimulating effects of ethylene and auxin on petiole elongation and on hyponastic curvature are independent processes in submerged Rumex palustris. Plant, Cell Environ 29:282–290
Fukao T, Bailey-Serres J (2008) Submergence tolerance conferred by Sub1A is mediated by SLR1 and SLRL1 restriction of gibberellin responses in rice. Proc Natl Acad Sci USA 105:16814–16819
Fukao T, Xu K, Ronald PC, Bailey-Serres J (2006) A variable cluster of ethylene response factor-like genes regulates metabolic and developmental acclimation responses to submergence in rice. Plant Cell 18:2021–2034
Hattori Y, Nagai K, Furukawa S, Song XJ, Kawano R, Sakakibara H, Wu J, Matsumoto T, Yoshimura A, Kitano H, Matsuoka M, Mori H, Ashikari M (2009) The ethylene response factors SNORKEL1 and SNORKEL2 allow rice to adapt to deep water. Nature 460:1026–1030
Jackson MB, Ram PC (2003) Physiological and molecular basis of susceptibility and tolerance of rice plants to complete submergence. Ann Bot 91:227–241
Kende H, van der Knaap E, Cho HT (1998) Deepwater rice: a model plant to study stem elongation. Plant Physiol 118:1105–1110
Kihara H, Katayama TC, Tsunewaki K (1962) Floating habit of 10 strains of wild and cultivated rice. Jpn J Genet 37:1–9
Leopold AC, Kriedemann PE (1975) Plant growth and development, 2nd edn. McGraw-Hill, NewYork
Manzur ME, Grimoldi AA, Insausti P, Striker GG (2009) Escape from water or remain quiescent? Lotus tenuis changes its strategy depending on depth of submergence. Ann Bot 104:1163–1169
Niroula RK, Pucciariello C, Ho VT, Novi G, Fukao T, Perata P (2012) SUB1A-dependent and -independent mechanisms are involved in the flooding tolerance of wild rice species. Plant J 72:282–293
Peeters AJM, Cox MCH, Benschop JJ, Vreeburg RAM, Bou J, Voesenek LACJ (2002) Submergence research using Rumex palustris as a model; looking back and going forward. J Exp Bot 368:391–398
Piedade MTF, Junk WJ, Long SP (1991) The productivity of the C4 grass Echinochloa polystachya on the Amazon floodplain. Ecology 72:1456–1463
Ram PC, Singh AK, Singh BB, Singh VK, Singh HP, Setter TL, Singh VP, Singh PK (1999) Environmental characterization of floodwater in eastern India: relevance to submergence tolerance in lowland rice. Exp Agric 35:141–152
Septiningsih EM, Pamplona AM, Sanchez DL, Neeraja CN, Vergara GV, Heuer S, Ismail AM, Mackill DJ (2009) Development of submergence-tolerant rice cultivars: the Sub1 locus and beyond. Ann Bot 103:151–160
Setter TL, Laureles EV (1996) The beneficial effect of reduced elongation growth on submergence tolerance of rice. J Exp Bot 47:1551–1559
Setter TL, Kupkanchanakul T, Kupkanchanakul K, Bhekasut P, Wiengweera A, Greenway H (1987) Concentrations of CO2 and O2 in floodwater and in internodal lacunae of floating rice growing at 1-2 metre water depths. Plant, Cell Environ 10:767–776
Singh N, Dang TT, Vergara GV, Pandey DM, Sanchez D, Neeraja CN, Septiningsih EM, Mendioro M, Tecson-Mendoza EM, Ismail AM, Mackill DJ, Heuer S (2010) Molecular marker survey and expression analyses of the rice submergence-tolerance gene SUB1A. Theor Appl Genet 121:1441–1453
van Veen H, Mustroph A, Barding GA, Vergeer-van Eijk M, Welschen-Evertman RAM, Pedersen O, Visser EJW, Larive CK, Pierik R, Bailey-Serres J, Voesenek LACJ, Sasidharan R (2013) Two Rumex species from contrasting hydrological niches regulate flooding tolerance through distinct mechanisms. Plant Cell 25:4691–4707
Vaughan DA (1994) The Wild Relatives of Rice: A genetic resources handbook. International Rice Research Institute, Manila
Voesenek LACJ, Benschop JJ, Bou J, Cox MCH, Groeneveld HW, Millenaar FF, Vreeburg RAM, Peeters AJM (2003) Interactions between plant hormones regulate submergence-induced shoot elongation in the flooding-tolerant dicot Rumex palustris. Ann Bot 91:205–211
Xu K, Xu X, Fukao T, Canlas P, Maghirang-Rodriguez R, Heuer S, Ismail AM, Bailey-Serres J, Ronald PC, Mackill DJ (2006) Sub1A is an ethylene-response-factor-like gene that confers submergence tolerance to rice. Nature 442:705–708
Acknowledgments
We thank Dr N. Mori (Kobe University, Japan) for providing seeds of FR13A. This work was supported by the Japan Society for the Promotion of Science KAKENHI (24580022 to T.A.).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Okishio, T., Sasayama, D., Hirano, T. et al. Growth promotion and inhibition of the Amazonian wild rice species Oryza grandiglumis to survive flooding. Planta 240, 459–469 (2014). https://doi.org/10.1007/s00425-014-2100-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-014-2100-8