Abstract
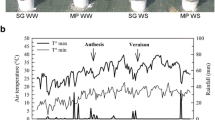
Fast and efficient recovery from water stress is a key determinant of plant adaptation to changing meteorological conditions modulating transpiration, i.e. air temperature and humidity. We analysed transcriptomic responses during rehydration after water stress in grapevine leaf petioles, where embolism formation and repair commonly take place, and where metabolic changes related to embolism recovery are expected to be particularly important. We compared gene expression of recovering plants with irrigated controls, upon high and low transpiration conditions, using cDNA microarrays. In parallel, we assessed the daily dynamics of water relations, embolism formation and repair, and leaf abscisic acid concentration. In recovering plants, the most affected gene categories were secondary metabolism, including genes linked to flavonoid biosynthesis; sugar metabolism and transport, and several aquaporin genes. The physiological dynamics of recovery were lower and the number of differentially expressed probes was much lower upon low transpiration than found in actively transpiring grapevines, suggesting the existence of a more intense metabolic reorganization upon high transpiration conditions and of a signal eliciting these responses. In plants recovering under high transpiration, abscisic acid concentrations significantly increased, and, in parallel, transcripts linked to abscisic acid metabolism and signalling (ABA-8′-hydroxylase, serine-threonine kinases, RD22 proteins) were upregulated; a trend that was not observed upon low transpiration. Our results show that recovery from water stress elicits complex transcriptomic responses in grapevine. The increase observed in abscisic acid cellular levels could represent a signal triggering the activation of responses to rehydration after stress.






Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- IRR:
-
Irrigated
- WS:
-
Water stress
- REC:
-
Recovery
- HCFM:
-
Hydraulic conductance flow meter
- qRT-PCR:
-
Quantitative real-time PCR
References
Ageorges A, Fernandez L, Vialet S, Merdinoglou D, Terrier N, Romieu C (2006) Four specific isogenes of the anthocyanin metabolic pathway are systematically co-expressed with the red colour of grape berries. Plant Sci 170:372–383
Almeida-Rodriguez AM, Hacke UG, Laur J (2011) Influence of evaporative demand on aquaporin expression and root hydraulics of hybrid poplar. Plant Cell Environ 34:1318–1331
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, Kasarskis A, Lewis S, Matese JC, Richardson JE, Ringwald M, Rubin GM, Sherlock G (2000) Gene Ontology: tool for the unification of biology. Nat Rev Genet 25:25–29
Baiges I, Schaffner AR, Mas A (2001) Eight cDNA encoding putative aquaporins in Vitis hybrid Richter-110 and their differential expression. J Exp Bot 52:1949–1951
Brodersen CR, McElrone AJ, Choat B, Matthews MA, Shackel KA (2010) The dynamics of embolism repair in xylem: in vivo visualizations using High-Resolution Computed Tomography. Plant Physiol 154:1088–1095
Bucci SJ, Scholz FG, Goldstein G, Meinzer FC, Sternberg LDL (2003) Dynamic changes in hydraulic conductivity in petioles of two savanna tree species: factors and mechanisms contributing to the refilling of embolized vessels. Plant Cell Environ 26:1633–1645
Carra A, Gambino G, Schubert A (2007) A cetyltrimethylammonium bromide-based method to extract low-molecular-weight RNA from polysaccharide-rich plant tissues. Anal Biochem 360:318–320
Chaves MM, Santos TP, Souza CR, Ortuno MF, Rodrigues ML, Lopes CM, Maroco JP, Pereira JS (2007) Deficit irrigation in grapevine improves water-use efficiency while controlling vigour and production quality. Ann Appl Biol 150:237–252
Choat B, Gambetta GA, Shackel KA, Matthews MA (2009) Vascular function in grape berries across development and its relevance to apparent hydraulic isolation. Plant Physiol 151:1677–1687
Choat B, Drayton WM, Brodersen C, Matthews MA, Shackel KA, Wada H, McElrone AJ (2010) Measurement of vulnerability to water stress-induced cavitation in grapevine: a comparison of four techniques applied to a long-vesseled species. Plant Cell Environ 33:1502–1512
Delk NA, Johnson KA, Chowdhury NI, Braam J (2005) CML24, regulated in expression by diverse stimuli, encodes a potential Ca2+ sensor that functions in responses to abscisic acid, daylength, and ion stress. Plant Physiol 139:240–253
Domec JC, Scholz FG, Bucci SJ, Meinzer FC, Goldstein G, Villalobos-Vega R (2006) Diurnal and seasonal variation in root xylem embolism in neotropical savanna woody species: impact on stomatal control of plant water status. Plant Cell Environ 29:26–35
Downton WJS, Loveys BR, Grant WJR (1988) Stomatal closure fully accounts for the inhibition of photosynthesis by abscisic-acid. New Phytol 108:263–266
Flexas J, Baron M, Bota J, Ducruet JM, Galle A, Galmes J, Jimenez M, Pou A, Ribas-Carbo M, Sajnani C, Tomas M, Medrano H (2009) Photosynthesis limitations during water stress acclimation and recovery in the drought-adapted Vitis hybrid Richter-110 (V-berlandierixV-rupestris). J Exp Bot 60:2361–2377
Fournier-Level A, Le Cunff L, Gomez C, Doligez A, Ageorges A, Roux C, Bertrand Y, Souquet JM, Cheynier V, This P (2009) Quantitative genetic bases of anthocyanin variation in grape (Vitis vinifera L. ssp. sativa) berry: a quantitative trait locus to quantitative trait nucleotide integrated study. Genetics 183:1127–1139
Galle A, Florez-Sarasa I, Tomas M, Pou A, Medrano H, Ribas-Carbo M, Flexas J (2009) The role of mesophyll conductance during water stress and recovery in tobacco (Nicotiana sylvestris): acclimation or limitation? J Exp Bot 60:2379–2390
Giribaldi M, Geny L, Delrot S, Schubert A (2010) Proteomic analysis of the effects of ABA treatments on ripening Vitis vinifera berries. J Exp Bot 61:2447–2458
Gomez C, Conejero G, Torregrosa L, Cheynier V, Terrier N, Ageorges A (2011) In vivo grapevine anthocyanin transport involves vesiclemediated trafficking and the contribution of anthoMATE transporters and GST. Plant J 67:960–970
Holbrook NM, Zwieniecki MA (1999) Embolism repair and xylem tension: do we need a miracle? Plant Physiol 120:7–10
Hugueney P, Provenzano S, Verries C, Ferrandino A, Meudec E, Batelli G, Merdinoglu D, Cheynier V, Schubert A, Ageorges A (2009) A novel cation-dependent O-methyltransferase involved in anthocyanin methylation in grapevine. Plant Physiol 150:2057–2070
Jang JY, Lee SH, Rhee JY, Chung GC, Ahn SJ, Kang HS (2007) Transgenic Arabidopsis and tobacco plants overexpressing an aquaporin respond differently to various abiotic stresses. Plant Mol Biol 64:621–632
Jeong ST, Goto-Yamamoto N, Kobayashi S, Esaka A (2004) Effects of plant hormones and shading on the accumulation of anthocyanins and the expression of anthocyanin biosynthetic genes in grape berry skins. Plant Sci 167:247–252
Jordao AM, Ricardo-da-Silva JM, Laureano O (2001) Evolution of proanthocyanidins in bunch stems during berry development (Vitis vinifera L.). Vitis 40:17–22
Kaldenhoff R, Fischer M (2006) Functional aquaporin diversity in plants. Biochim Biophys Acta Biomembr 1758:1134–1141
Kaldenhoff R, Ribas-Carbo M, Flexas J, Lovisolo C, Heckwolf M, Uehlein N (2008) Aquaporins and plant water balance. Plant Cell Environ 31:658–666
Kirschbaum MUF (1988) Recovery of photosynthesis from water-stress in Eucaliptus pauciflora—a process in 2 stages. Plant Cell Environ 11:685–694
Kobayashi S, Ishimaru M, Hiraoka K, Honda C (2002) Myb-related genes of the Kyoho grape (Vitis labruscana) regulate anthocyanin biosynthesis. Planta 215:924–933
Kobayashi Y, Yamamoto S, Minami H, Kagaya Y, Hattori T (2004) Differential activation of the rice sucrose nonfermenting1-related protein kinase2 family by hyperosmotic stress and abscisic acid. Plant Cell 16:1163–1177
Kobayashi Y, Murata M, Minami H, Yamamoto S, Kagaya Y, Hobo T, Yamamoto A, Hattori T (2005) Abscisic acid-activated SNRK2 protein kinases function in the gene-regulation pathway of ABA signal transduction by phosphorylating ABA response element-binding factors. Plant J 44:939–949
Lehti-Shiu MD, Zou C, Hanada K, Shiu SH (2009) Evolutionary history and stress regulation of plant receptor-like kinase/pelle genes. Plant Physiol 150:12–26
Lovisolo C, Schubert A (1998) Effects of water stress on vessel size and xylem hydraulic conductivity in Vitis vinifera L. J Exp Bot 49:693–700
Lovisolo C, Schubert A (2006) Mercury hinders recovery of shoot hydraulic conductivity during grapevine rehydration: evidence from a whole-plant approach. New Phytol 172:469–478
Lovisolo C, Perrone I, Hartung W, Schubert A (2008a) An abscisic acid-related reduced transpiration promotes gradual embolism repair when grapevines are rehydrated after drought. New Phytol 180:642–651
Lovisolo C, Tramontini S, Flexas J, Schubert A (2008b) Mercurial inhibition of root hydraulic conductance in Vitis spp. rootstocks under water stress. Environ Exp Bot 63:178–182
Lovisolo C, Perrone I, Carra A, Ferrandino A, Flexas J, Medrano H, Schubert A (2010) Drought-induced changes in development and function of grapevine (Vitis spp.) organs and in their hydraulic and non-hydraulic interactions at the whole-plant level: a physiological and molecular update. Funct Plant Biol 37:98–116
Mahdieh M, Mostajeran A (2009) Abscisic acid regulates root hydraulic conductance via aquaporin expression modulation in Nicotiana tabacum. J Plant Physiol 166:1993–2003
Martre P, Morillon R, Barrieu F, North GB, Nobel PS, Chrispeels MJ (2002) Plasma membrane aquaporins play a significant role during recovery from water deficit. Plant Physiol 130:2101–2110
Matsui A, Ishida J, Morosawa T, Mochizuki Y, Kaminuma E, Endo TA, Okamoto M, Nambara E, Nakajima M, Kawashima M, Satou M, Kim JM, Kobayashi N, Toyoda T, Shinozaki K, Seki M (2008) Arabidopsis transcriptome analysis under drought, cold, high-salinity and ABA treatment conditions using a tiling array. Plant Cell Physiol 49:1135–1149
Melcher PJ, Goldstein G, Meinzer FC, Yount DE, Jones TJ, Holbrook NM, Huang CX (2001) Water relations of coastal and estuarine Rhizophora mangle: xylem pressure potential and dynamics of embolism formation and repair. Oecologia 126:182–192
Montalvo-Hernandez L, Piedra-Ibarra E, Gomez-Silva L, Lira-Carmona R, Acosta-Gallegos JA, Vazquez-Medrano J, Xoconostle-Cazares B, Ruiz-Medrano R (2008) Differential accumulation of mRNAs in drought-tolerant and susceptible common bean cultivars in response to water deficit. New Phytol 177:102–113
Morillon R, Chrispeels MJ (2001) The role of ABA and the transpiration stream in the regulation of the osmotic water permeability of leaf cells. Proc Natl Acad Sci USA 98:14138–14143
Nambara E, Marion-Poll A (2005) Abscisic acid biosynthesis and catabolism. Ann Rev Plant Biol 56:165–185
Oono Y, Seki M, Nanjo T, Narusaka M, Fujita M, Satoh R, Satou M, Sakurai T, Ishida J, Akiyama K, Iida K, Maruyama K, Satoh S, Yamaguchi-Shinozaki K, Shinozaki K ((2003)) Monitoring expression profiles of Arabidopsis gene expression during rehydration process after dehydration using ca. 7000 full-length cDNA microarray. Plant J 34:868–887
Parent B, Hachez C, Redondo E, Simonneau T, Chaumont F, Tardieu F (2009) Drought and abscisic acid effects on aquaporin content translate into changes in hydraulic conductivity and leaf growth rate: a trans-scale approach. Plant Physiol 149:2000–2012
Sakr S, Alves G, Morillon RL, Maurel K, Decourteix M, Guilliot A, Fleurat-Lessard P, Julien JL, Chrispeels MJ (2003) Plasma membrane aquaporins are involved in winter embolism recovery in walnut tree. Plant Physiol 133:630–641
Sakurai-Ishikawa J, Murai-Hatano M, Hayashi H, Ahamed A, Fukushi K, Matsumoto T, Kitagawa Y (2011) Transpiration from shoots triggers diurnal changes in root aquaporin expression. Plant Cell Environ 34:1150–1163
Salleo S, LoGullo MA, DePaoli D, Zippo M (1996) Xylem recovery from cavitation-induced embolism in young plants of Laurus nobilis: a possible mechanism. New Phytol 132:47–56
Salleo S, Lo Gullo MA, Trifilo P, Nardini A (2004) New evidence for a role of vessel-associated cells and phloem in the rapid xylem refilling of cavitated stems of Laurus nobilis L. Plant Cell Environ 27:1065–1076
Schultz HR (2003) Differences in hydraulic architecture account for near-isohydric and anisohydric behaviour of two field-grown Vitis vinifera L. cultivars during drought. Plant Cell Environ 26:1393–1405
Schultz HR, Matthews MA (1988) Resistance to water transport in shoots of Vitis vinifera L.—relation to growth at low water potential. Plant Physiol 88:718–724
Secchi F, Zwieniecki MA (2010) Patterns of PIP gene expression in Populus trichocarpa during recovery from xylem embolism suggest a major role for the PIP1 aquaporin subfamily as moderators of refilling process. Plant Cell Environ 33:1285–1297
Secchi F, Zwieniecki MA (2011) Sensing embolism in xylem vessels: the role of sucrose as a trigger for refilling. Plant Cell Environ 34:514–524
Seki M, Ishida J, Narusaka M, Fujita M, Nanjo T, Umezawa T, Kamiya A, Nakajima M, Enju A, Sakurai T, Satou M, Akiyama K, Yamaguchi-Shinozaki K, Carninci P, Kawai J, Hayashizaki Y, Shinozaki K (2002) Monitoring the expression pattern of around 7, 000 Arabidopsis genes under ABA treatments using a full-length cDNA microarray. Funct Integr Genomics 2:282–291
Shatil-Cohen A, Attia Z, Moshelion M (2011) Bundle-sheath cell regulation of xylem-mesophyll water transport via aquaporins under drought stress: a target of xylem-borne ABA? Plant J 67:72–80
Soar CJ, Speirs J, Maffei SM, Loveys BR (2004) Gradients in stomatal conductance, xylem sap ABA and bulk leaf ABA along canes of Vitis vinifera cv. Shiraz: molecular and physiological studies investigating their source. Funct Plant Biol 31:659–669
Soar CJ, Speirs J, Maffei SM, Penrose AB, McCarthy MG, Loveys BR (2006) Grape vine varieties Shiraz and Grenache differ in their stomatal response to VPD: apparent links with ABA physiology and gene expression in leaf tissue. Aust J Grape Wine Res 12:2–12
Trainotti L, Bonghi C, Ziliotto F, Zanin D, Rasori A, Casadoro G, Ramina A, Tonutti P (2006) The use of microarray mu PEACH1.0 to investigate transcriptome changes during transition from pre-climacteric to climacteric phase in peach fruit. Plant Sci 170:606–613
Tusher VG, Tibshirani R, Chu G (2001) Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci USA 98:5116–5121
Tyree MT, Patino S, Bennink J, Alexander J (1995) Dynamic measurements of root hydraulic conductance using a high-pressure flowmeter in the laboratory and field. J Exp Bot 46:83–94
Vandeleur RK, Mayo G, Shelden MC, Gilliham M, Kaiser BN, Tyerman SD (2009) The role of plasma membrane intrinsic protein aquaporins in water transport through roots: diurnal and drought stress responses reveal different strategies between isohydric and anisohydric cultivars of grapevine. Plant Physiol 149:445–460
Vicre M, Farrant JM, Driouich A (2004) Insights into the cellular mechanisms of desiccation tolerance among angiosperm resurrection plant species. Plant Cell Environ 27:1329–1340
Zenoni S, Ferrarini A, Giacomelli E, Xumerle L, Fasoli M, Malerba G, Bellin D, Pezzotti M, Delledonne M (2010) Characterization of transcriptional complexity during berry development in Vitis vinifera using RNA-seq. Plant Physiol 152:1787–1795
Zufferey V, Cochard H, Ameglio T, Spring JL, Viret O (2011) Diurnal cycles of embolism formation and repair in petioles of grapevine (Vitis vinifera cv. Chasselas). J Exp Bot 62:3885–3894
Zurek DM, Clouse SD (1994) Molecular cloning and characterization of a brassinosteroid-regulated gene from elongating soybean (Glycine max L.) epicotyls. Plant Physiol 104:161–170
Acknowledgments
The authors wish to thank Dr. Claudio Bonghi from the University of Padova, for assistance in designing the microarray protocols, Vivai Cooperativi Rauscedo—Italy, for providing grapevines, and two anonymous reviewers for having improved former versions of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
I. Perrone and C. Pagliarani equally contributed to this paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Perrone, I., Pagliarani, C., Lovisolo, C. et al. Recovery from water stress affects grape leaf petiole transcriptome. Planta 235, 1383–1396 (2012). https://doi.org/10.1007/s00425-011-1581-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-011-1581-y