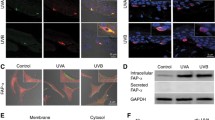
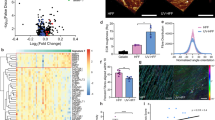
Abstract
Melanoma represents a malignant disease with steadily increasing incidence. UV-irradiation is a recognized key factor in melanoma initiation. Therefore, the efficient prevention of UV tissue damage bears a critical potential for melanoma prevention. In this study, we tested the effect of UV irradiation of normal keratinocytes and their consequent interaction with normal and cancer-associated fibroblasts isolated from melanoma, respectively. Using this model of UV influenced microenvironment, we measured melanoma cell migration in 3-D collagen gels. These interactions were studied using DNA microarray technology, immunofluorescence staining, single cell electrophoresis assay, viability (dead/life) cell detection methods, and migration analysis. We observed that three 10 mJ/cm2 fractions at equal intervals over 72 h applied on keratinocytes lead to a 50% increase (p < 0.05) in in vitro invasion of melanoma cells. The introduction cancer-associated fibroblasts to such model further significantly stimulated melanoma cells in vitro invasiveness to a higher extent than normal fibroblasts. A panel of candidate gene products responsible for facilitation of melanoma cells invasion was defined with emphasis on IL-6, IL-8, and CXCL-1. In conclusion, this study demonstrates a synergistic effect between cancer microenvironment and UV irradiation in melanoma invasiveness under in vitro condition.








Similar content being viewed by others
References
Adini I, Ghosh K, Adini A, Chi ZL, Yoshimura T, Benny O, Connor KM, Rogers MS, Bazinet L, Birsner AE, Bielenberg DR, D’Amato RJ (2014) Melanocyte-secreted fibromodulin promotes an angiogenic microenvironment. J Clin Investig 124:425–436
Adini I, Adini A, Bazinet L, Watnick RS, Bielenberg DR, D’Amato RJ (2015) Melanocyte pigmentation inversely correlates with MCP-1 production and angiogenesis-inducing potential. FASEB J 29:662–670
Barrientos S, Stojadinovic O, Golinko MS, Brem H, Tomic-Canic M (2008) Growth factors and cytokines in wound healing. Wound Repair Regen 16:585–601
Benke EM, Ji Y, Patel V, Wang H, Miyazaki H, Yeudall WA (2010) VEGF-C contributes to head and neck squamous cell carcinoma growth and motility. Oral Oncol 46:e19–e24
Brash DE (2015) UV signature mutations. Photochem Photobiol 91:15–26
Brennan M, Bhatti H, Nerusu KC, Bhagavathula N, Kang S, Fisher GJ, Varani J, Voorhees JJ (2003) Matrix metalloproteinase-1 is the major collagenolytic enzyme responsible for collagen damage in UV-irradiated human skin. Photochem Photobiol 78:43–48
Brenner M, Degitz K, Besch R, Berking C (2005) Differential expression of melanoma-associated growth factors in keratinocytes and fibroblasts by ultraviolet A and ultraviolet B radiation. Br J Dermatol 153:733–739
Catania A (2007) The melanocortin system in leukocyte biology. J Leukocyte Biol 81:383–392
Cheli Y, Giuliano S, Botton T, Rocchi S, Hofman V, Hofman P, Bahadoran P, Bertolotto C, Ballotti R (2011) Mitf is the key molecular switch between mouse or human melanoma initiating cells and their differentiated progeny. Oncogene 30:2307–2318
Childs BG, Durik M, Baker DJ, van Deursen JM (2015) Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med 21:1424–1435
Chou WC, Takeo M, Rabbani P, Hu H, Lee W, Chung YR, Carucci J, Overbeek P, Ito M (2013) Direct migration of follicular melanocyte stem cells to the epidermis after wounding or UVB irradiation is dependent on Mc1r signaling. Nat Med 19:924–929
D’Orazio J, Jarrett S, Amaro-Ortiz A, Scott T (2013) UV Radiation and the Skin. Int J Mol Sci 14:12222–12248
Di Cesare S, Marshall JC, Logan P, Antecka E, Faingold D, Maloney SC, Burnier MN Jr (2007) Expression and migratory analysis of 5 human uveal melanoma cell lines for CXCL12, CXCL8, CXCL1, and HGF. J Carcinogen 6:2
Dornelles S, Goldim J, Cestari T (2004) Determination of the minimal erythema dose and colorimetric measurements as indicators of skin sensitivity to UV-B radiation. Photochem Photobiol 79:540–544
Drigeard Desgarnier MC, Fournier F, Droit A, Rochette PJ (2017) Influence of a pre-stimulation with chronic low-dose UVB on stress response mechanisms in human skin fibroblasts. PLoS One 12:e0173740
Dvořánková B, Szabo P, Lacina L, Kodet O, Matoušková E, Smetana K Jr (2012) Fibroblasts prepared from different types of malignant tumors stimulate expression of luminal marker keratin 8 in the EM-G3 breast cancer cell line. Histochem Cell Biol 137:679–685
Dvořánková B, Szabo P, Kodet O, Strnad H, Kolář M, Lacina L, Krejčí E, Naňka O, Šedo A, Smetana K Jr (2017) Intercellular crosstalk in human malignant melanoma. Protoplasma 254:1143–1150
Finger EC, Cheng CF, Williams TR, Rankin EB, Bedogni B, Tachiki L, Spong S, Giaccia AJ, Powell MB (2014) CTGF is a therapeutic target for metastatic melanoma. Oncogene 33:1093–1100
Fisher GJ, Datta SC, Talwar HS, Wang ZQ, Varani J, Kang S, Voorhees JJ (1996) Molecular basis of sun-induced premature skin ageing and retinoid antagonism. Nature 379:335–339
Fisher GJ, Datta S, Wang Z, Li XY, Quan T, Chung JH, Kang S, Voorhees JJ (2000) c-Jun-dependent inhibition of cutaneous procollagen transcription following ultraviolet irradiation is reversed by all-trans retinoic acid. J Clin Investig 106:663–670
Gentleman RC, Carey VJ, Bates DM, Bolstad B, Dettling M, Dudoit S, Ellis B, Gautier L, Ge Y, Gentry J, Hornik K, Hothorn T, Huber W, Iacus S, Irizarry R, Leisch F, Li C, Maechler M, Rossini AJ, Sawitzki G, Smith C, Smyth G, Tierney L, Yang JY, Zhang J (2004) Bioconductor: open software development for computational biology and bioinformatics. Genome Biol 5:R80
Ghosh K, Capell BC (2016) The senescence-associated secretory phenotype: critical effector in skin cancer and aging. J Investig Dermatol 136:2133–2139
Golan T, Messer AR, Amitai-Lange A, Melamed Z, Ohana R, Bell RE, Kapitansky O, Lerman G, Greenberger S, Khaled M, Amar N, Albrengues J, Gaggioli C, Gonen P, Tabach Y, Sprinzak D, Shalom-Feuerstein R, Levy C (2015) Interactions of melanoma cells with distal keratinocytes trigger metastasis via notch signaling inhibition of MITF. Mol Cell 59:664–676
Gyori BM, Venkatachalam G, Thiagarajan PS, Hsu D, Clement MV (2014) OpenComet: an automated tool for comet assay image analysis. Redox Biol 2:457–465
Hammer R, Harper DAT, Ryan PD (2001) Paleontological statistics software package for education and data analysis. Palaeontol Electron 4:1–9
Hochberg M, Gilead L, Markel G, Nemlich Y, Feiler Y, Enk CD, Denichenko P, Karni R, Ingber A (2013) Insulin-like growth factor-binding protein-7 (IGFBP7) transcript: A-to-I editing events in normal and cancerous human keratinocytes. Arch Dermatol Res 305:519–528
Iida J, Wilhelmson KL, Ng J, Lee P, Morrison C, Tam E, Overall CM, McCarthy JB (2007) Cell surface chondroitin sulfate glycosaminoglycan in melanoma: role in the activation of pro-MMP-2 (pro-gelatinase A). Biochem J 403:553–563
Jackson SP, Bartek J (2009) The DNA-damage response in human biology and disease. Nature 461:1071–1078
Jobe NP, Rösel D, Dvořánková B, Kodet O, Lacina L, Mateu R, Smetana K, Brábek J (2016) Simultaneous blocking of IL-6 and IL-8 is sufficient to fully inhibit CAF-induced human melanoma cell invasiveness. Histochem Cell Biol 146:205–217
Johnson NR, Wang Y (2013) Controlled delivery of heparin-binding EGF-like growth factor yields fast and comprehensive wound healing. J Control Release 166:124–129
Kamp H, Geilen CC, Sommer C, Blume-Peytavi U (2003) Regulation of PDGF and PDGF receptor in cultured dermal papilla cells and follicular keratinocytes of the human hair follicle. Exp Dermatol 12:662–672
Kant V, Kumar D, Kumar D, Prasad R, Gopal A, Pathak NN, Kumar P, Tandan SK (2015) Topical application of substance P promotes wound healing in streptozotocin-induced diabetic rats. Cytokine 73:144–155
Kim EJ, Kim YK, Kim JE, Kim S, Kim M-K, Park C-H, Chung JH (2011) UV Modulation of subcutaneous fat metabolism. J Investig Dermatol 131:1720–1726
Kodet O, Dvořánková B, Krejčí E, Szabo P, Dvořák P, Štork J, Krajsová I, Dundr P, Smetana K Jr, Lacina L (2013) Cultivation-dependent plasticity of melanoma phenotype. Tumour Biol 34:3345–3355
Kodet O, Lacina L, Krejčí E, Dvořánková B, Grim M, Štork J, Kodetová D, Vlček Č, Šáchová J, Kolář M, Strnad H, Smetana K Jr (2015) Melanoma cells influence the differentiation pattern of human epidermal keratinocytes. Mol Cancer 14:1
Kolář M, Szabo P, Dvořánková B, Lacina L, Gabius H-J, Strnad H, Sáchová J, Vlček C, Plzák J, Chovanec M, Cada Z, Betka J, Fík Z, Pačes J, Kovářová H, Motlík J, Jarkovská K, Smetana K Jr (2012) Upregulation of IL-6, IL-8 and CXCL-1 production in dermal fibroblasts by normal/malignant epithelial cells in vitro, immunohistochemical and transcriptomic analyses. Biol Cell 104:738–751
Kondo T, Hearing VJ (2011) Update on the regulation of mammalian melanocyte function and skin pigmentation. Expert Rev Dermatol 6:97–108
Krejčí E, Kodet O, Szabo P, Borský J, Smetana K Jr, Grim M, Dvořánková B (2015) In vitro differences of neonatal and later postnatal keratinocytes and dermal fibroblasts. Physiol Res 64:561–569
Kučera J, Dvořánková B, Smetana K Jr, Szabo P, Kodet O (2015) Fibroblasts isolated from the malignant melanoma influence phenotype of normal human keratinocytes. J Appl Biomed 13:195–198
La Porta CA (2012) CXCR6: the role of environment in tumor progression. Challenges for therapy. Stem Cell Rev 8:1282–1285
Lacina L, Plzak J, Kodet O, Szabo P, Chovanec M, Dvorankova B, Smetana K Jr (2015) Cancer microenvironment: What can we learn from the stem cell niche. Int J Mol Sci 16:24094–24110
Lacina L, Kodet O, Dvořánková B, Szabo P, Smetana K Jr (2017) Ecology of melanoma cells. Histol Histopathol. https://doi.org/10.14670/HH-11-926
Lo JA, Fisher DE (2014) The melanoma revolution: from UV carcinogenesis to a new era in therapeutics. Science 346:945–949
Marchetti D, Nicolson GL (1997) Human melanoma cell invasion: selected neurotrophin enhancement of invasion and heparanase activity. J Investig Dermatol Symp Proc 2:99–105
Mateu R, Živicová V, Drobná Krejčí E, Grim M, Strnad H, Vlček Č, Kolář M, Lacina L, Gál P, Borský J, Smetana K Jr, Dvořánková B (2016) Functional differences between neonatal and adult fibroblasts and keratinocytes. Int J Mol Med 38:1063–1074
Merkel EA, Gerami P (2017) Malignant melanoma of sun-protected sites: a review of clinical, histological, and molecular features. Lab Investig 97:630–635
Ortiz-Montero P, Londoño-Vallejo A, Vernot JP (2017) Senescence-associated IL-6 and IL-8 cytokines induce a self- and cross-reinforced senescence/inflammatory milieu strengthening tumorigenic capabilities in the MCF-7 breast cancer cell line. Cell Commun Signal 15:17
Peppicelli S, Bianchini F, Calorini L (2014) Inflammatory cytokines induce vascular endothelial growth factor-C expression in melanoma-associated macrophages and stimulate melanoma lymph node metastasis. Oncol Lett 8:1133–1138
Quan T, Qin Z, Xia W, Shao Y, Voorhees JJ, Fisher GJ (2009) Matrix-degrading metalloproteinases in photoaging. J Investig Dermatol Symp Proc 14:20–24
Rastrelli M, Tropea S, Rossi CR, Alaibac M (2014) Melanoma: epidemiology, risk factors, pathogenesis, diagnosis and classification. In Vivo 28:1005–1011
Rothhammer T, Poser I, Soncin F, Bataille F, Moser M, Bosserhoff AK (2005) Bone morphogenic proteins are overexpressed in malignant melanoma and promote cell invasion and migration. Cancer Res 65:448–456
Rünger TM (2016) Mechanisms of melanoma promotion by ultraviolet radiation. J Investig Dermatol 136:1751–1752
Sabbatino F, Wang Y, Wang X, Flaherty KT, Yu L, Pepin D, Scognamiglio G, Pepe S, Kirkwood JM, Cooper ZA, Frederick DT, Wargo JA, Ferrone S, Ferrone CR (2014) PDGFRα up-regulation mediated by sonic hedgehog pathway activation leads to BRAF inhibitor resistance in melanoma cells with BRAF mutation. Oncotarget 5:1926–1941
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Shafritz TA, Rosenberg LC, Yannas IV (1994) Specific effects of glycosaminoglycans in an analog of extracellular matrix that delays wound contraction and induces regeneration. Wound Repair Regen 2:270–276
Smetana K Jr, Dvořánková B, Lacina L (2016a) Phylogeny, regeneration, ageing and cancer: role of microenvironment and possibility of its therapeutic manipulation. Folia Biol 59:207–216
Smetana K Jr, Lacina L, Szabo P, Dvořánková B, Brož P, Šedo A (2016b) Ageing as an important risk factor for cancer. Anticancer Res 36:5009–5017
Smyth GK (2006) Linear models and empirical Bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol 3:3
Tice RR, Agurell E, Anderson D, Burlinson B, Hartmann A, Kobayashi H, Miyamae Y, Rojas E, Ryu JC, Sasaki YF (2000) Single cell gel/comet assay: guidelines for in vitro and in vivo genetic toxicology testing. Environ Mol Mutagen 35:206–221
Trylcova J, Busek P, Smetana K Jr, Balaziova E, Dvorankova B, Mifkova A, Sedo A (2015) Effect of cancer-associated fibroblasts on the migration of glioma cells in vitro. Tumour Biol 36:5873–5879
Vartanian A, Stepanova E, Grigorieva I, Solomko E, Baryshnikov A, Lichinitser M (2011) VEGFR1 and PKCα signaling control melanoma vasculogenic mimicry in a VEGFR2 kinase-independent manner. Melanoma Res 21:91–98
Volkmer B, Greinert R (2011) UV and Children’s skin. Prog Biophys Mol Biol 107:386–388
Wang JX, Fukunaga-Kalabis M, Herlyn M (2016) Crosstalk in skin: melanocytes, keratinocytes, stem cells, and melanoma. J Cell Commun Signal 10:191–196
Wu XJ, Zhu JW, Jing J, Xue D, Liu H, Zheng M, Lu ZF (2014) VEGF165 modulates proliferation, adhesion, migration and differentiation of cultured human outer root sheath cells from central hair follicle epithelium through VEGFR-2 activation in vitro. J Dermatol Sci 73:152–160
Zelba H, Weide B, Martens A, Derhovanessian E, Bailur JK, Kyzirakos C, Pflugfelder A, Eigentler TK, Di Giacomo AM, Maio M, Aarntzen EH, de Vries J, Sucker A, Schadendorf D, Büttner P, Garbe C, Pawelec G (2014) Circulating CD4+ T cells that produce IL4 or IL17 when stimulated by melan-A but not by NY-ESO-1 have negative impacts on survival of patients with stage IV melanoma. Clin Cancer Res 20:4390–4399
Zhang C, Zhang F, Tsan R, Fidler IJ (2009) Transforming growth factor-beta2 is a molecular determinant for site-specific melanoma metastasis in the brain. Cancer Res 69:828–835
Acknowledgements
This publication is a result of the project implementation: “The equipment for metabolomic and cell analyses”, registration number CZ.1.05/2.1.00/19.0400, supported by Research and Development for Innovations Operational Programme (RDIOP) co-financed by European regional development fund and the state budget of the Czech Republic. This study was also supported by the Grant Agency of the Czech Republic (Project no. 16-05534S), AZV 16-29032A, the Charles University (project of Specific University Research, GAUK 165015 and PROGRESS 28 and UNCE 23014) and by the Ministry of Education, Youth and Sports of CR within the National Sustainability Program II (Project BIOCEV-FAR reg. no. LQ1604), and by the project BIOCEV (CZ.1.05/1.1.00/02.0109). The part of the study was performed by the equipment for metabolomics and cell analyses (Grant no. CZ.1.05/2.1.00/19.0400) supported by the Research and Development for Innovations Operational Program, co-financed by the European regional development fund and the state budget of the Czech Republic.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jobe, N.P., Živicová, V., Mifková, A. et al. Fibroblasts potentiate melanoma cells in vitro invasiveness induced by UV-irradiated keratinocytes. Histochem Cell Biol 149, 503–516 (2018). https://doi.org/10.1007/s00418-018-1650-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-018-1650-4