Abstract
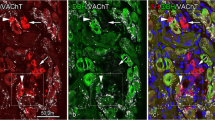
We investigated the three-dimensional architectures of P2X2-/P2X3-immunoreactive nerve terminals in the rat carotid body using immunohistochemistry with confocal laser microscopy. Nerve endings immunoreactive for P2X2 and P2X3 were associated with clusters of type I cells, whereas some nerve endings were sparsely distributed in a few clusters. Most nerve endings surrounding type I cells were hederiform in shape and extended several flattened axon terminals, which were polygonal or pleomorphic in shape and contained P2X2-/P2X3-immunoreactive products. Three-dimensional reconstruction views revealed that some flattened nerve endings with P2X3 immunoreactivity formed arborized, sac- or goblet-like terminal structures and were attached to type I cells immunoreactive for tyrosine hydroxylase (TH). However, P2X3-immunoreactive axon terminals were sparsely distributed in type I cells immunoreactive for dopamine beta-hydroxylase. Multi-immunolabeling for P2X2, S100, and TH revealed that P2X2-immunoreactive axon terminals were attached to TH-immunoreactive type I cells on the inside of type II cells with S100 immunoreactivity. These results revealed the detailed morphology of P2X2-/P2X3-immunoreactive nerve terminals and suggest that sensory nerve endings may integrate chemosensory signals from clustered type I cells with their variform nerve terminals.






Similar content being viewed by others
References
Biscoe TJ, Pallot DJ (1982) The carotid body chemoreceptor: an investigation in the mouse. Q J Exp Physiol 67:557–576
Biscoe TJ, Stehbens WE (1966) Ultrastructure of the carotid body. J Cell Biol 30:563–578
Brouns I, Oztay F, Pintelon I, De Proost I, Lembrechts R, Timmermans JP, Adriaensen D (2009) Neurochemical pattern of the complex innervation of neuroepithelial bodies in mouse lungs. Histochem Cell Biol 131:55–74. doi:10.1007/s00418-008-0495-7
Buttigieg J, Nurse CA (2004) Detection of hypoxia-evoked ATP release from chemoreceptor cells of the rat carotid body. Biochem Biophys Res Commun 322:82–87. doi:10.1016/j.bbrc.2004.07.081
Fitzgerald RS, Shirahata M, Chang I (2006) The impact of PCO2 and H+ on the release of acetylcholine from the cat carotid body. Neurosci Lett 397:205–259. doi:10.1016/j.neulet.2005.12.024
Gonzalez C, Almaraz L, Obeso A, Rigual R (1994) Carotid body chemoreceptors: from natural stimuli to sensory discharges. Physiol Rev 74:829–898
Gourine AV (2005) On the peripheral and central chemoreception and control of breathing: an emerging role of ATP. J Physiol 568:715–724. doi:10.1113/jphysiol.2005.095968
Habeck JO, Kummer W (1993) Neuronal and neuroendocrine markers in the human carotid body in health and disease. Adv Exp Med Biol 337:31–35
Hess A, Zapata P (1972) Innervation of the cat carotid body: normal and experimental studies. Fed Proc 31:1365–1382
Iturriaga R, Alcayaga J (2004) Neurotransmission in the carotid body: transmitters and modulators between glomus cells and petrosal ganglion nerve terminals. Brain Res Brain Res Rev 47:46–53. doi:10.1016/j.brainresrev.2004.05.007
Kataoka S, Toyono T, Seta Y, Toyoshima K (2006) Expression of ATP-gated P2X3 receptors in rat gustatory papillae and taste buds. Arch Histol Cytol 69:281–288. doi:10.1679/aohc.69.281
Kato K, Yokoyama T, Yamaguchi-Yamada M, Yamamoto Y (2013) Short-term hypoxia transiently increases dopamine β-hydroxylase immunoreactivity in glomus cells of the rat carotid body. J Histochem Cytochem 61:55–62. doi:10.1369/0022155412464639
Kondo H (1976) Innervation of the carotid body of the adult rat. Cell Tissue Res 173:1–15
Kondo H, Iwanaga T, Nakajima T (1982) Immunocytochemical study on the localization of neuron-specific enolase and S-100 protein in the carotid body of rats. Cell Tissue Res 227:291–295
Kumar P, Prabhakar NR (2012) Peripheral chemoreceptors: function and plasticity of the carotid body. Compr Physiol 2:141–219. doi:10.1002/cphy.c100069
Lahiri S, Roy A, Baby SM, Hoshi T, Semenza GL, Prabhakar NR (2006) Oxygen sensing in the body. Prog Biophys Mol Biol 91:249–286. doi:10.1016/j.pbiomolbio.2005.07.001
McDonald DM, Mitchell RA (1975) The innervations of glomus cells, ganglion cells and blood vessels in the rat carotid body: a quantitative ultrastructural analysis. J Neurocytol 4:177–230
Nakakura-Ohshima K, Maeda T, Ohshima H, Noda T, Takano Y (1995) Postnatal development of periodontal Ruffini endings in rat incisors: an immunoelectron microscopic study using protein gene product 9.5 (PGP 9.5)-antibody. J Comp Neurol 362:551–564. doi:10.1002/cne.903620409
Nishi K, Stensaas LJ (1974) The ultrastructure and source of nerve endings in the carotid body. Cell Tissue Res 154:303–319
Nurse CA (2005) Neurotransmission and neuromodulation in the chemosensory carotid body. Auton Neurosci 120:1–9. doi:10.1016/j.autneu.2005.04.008
Nurse CA (2010) Neurotransmitter and neuromodulatory mechanisms at peripheral arterial chemoreceptors. Exp Physiol 95:657–667. doi:10.1113/expphysiol.2009.049312
Piskuric NA, Nurse CA (2012) Effects of chemostimuli on [Ca2+]i responses of rat aortic body type I cells and endogenous local neurons: comparison with carotid body cells. J Physiol 590:2121–2135. doi:10.1113/jphysiol.2012.229468
Piskuric NA, Nurse CA (2013) Expanding role of ATP as a versatile messenger at carotid and aortic body chemoreceptors. J Physiol 591:415–422. doi:10.1113/jphysiol.2012.234377
Prasad M, Fearon IM, Zhang M, Laing M, Vollmer C, Nurse CA (2001) Expression of P2X2 and P2X3 receptor subunits in rat carotid body afferent neurones: role in chemosensory signalling. J Physiol (Lond) 537:667–677. doi:10.1111/j.1469-7793.2001.00667.x
Rong W, Gourine AV, Cockayne DA, Xiang Z, Ford AP, Spyer KM, Burnstock G (2003) Pivotal role of nucleotide P2X2 receptor subunit of the ATP-gated ion channel mediating ventilatory responses to hypoxia. J Neurosci 23:11315–11321
Schoch S, Gundelfinger ED (2006) Molecular organization of the presynaptic active zone. Cell Tissue Res 326:379–391. doi:10.1007/s00441-006-0244-y
Takahashi N, Nakamura N, Yamamoto Y (2016) Morphology of P2X3-immunoreactive nerve endings in the rat laryngeal mucosa. Histochem Cell Biol 145:131–146. doi:10.1007/s00418-015-1371-x
Takahashi-Iwanaga H (2000) Three-dimensional microanatomy of longitudinal lanceolate endings in rat vibrissae. J Comp Neurol 426:259–269. doi:10.1002/1096-9861(20001016)426:23.0.CO;2-N
Takaki F, Nakamuta N, Kusakabe T, Yamamoto Y (2015) Sympathetic and sensory innervation of small intensely fluorescent (SIF) cells in rat superior cervical ganglion. Cell Tissue Res 359:441–451. doi:10.1007/s00441-014-2051-1
Yang R, Montoya A, Bond A, Walton J, Kinnamon JC (2012) Immunocytochemical analysis of P2X2 in rat circumvallate taste buds. BMC Neurosci 13:51. doi:10.1186/1471-2202-13-51
Yokoyama T, Nakamuta N, Kusakabe T, Yamamoto Y (2014) Vesicular glutamate transporter 2-immunoreactive afferent nerve terminals in the carotid body of the rat. Cell Tissue Res 358:271–275. doi:10.1007/s00441-014-1921-x
Zhang M, Nurse CA (2004) CO2/pH chemosensory signaling in co-cultures of rat carotid body receptors and petrosal neurons: role of ATP and ACh. J Neurophysiol 92:3433–3445. doi:10.1152/jn.01099.2003
Zhang M, Piskuric NA, Vollmer C, Nurse CA (2012) P2Y2 receptor activation opens pannexin-1 channels in rat carotid body type II cells: potential role in amplifying the neurotransmitter ATP. J Physiol 590:4335–4350. doi:10.1113/jphysiol.2012.236265
Acknowledgments
This work was supported by Grants-in-Aid from the Japan Society for the Promotion of Science to TK (25350823) and TY (16K21306) and Grants-in-Aid for Strategic Medical Science Research (S1491001, 2014-2018) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yokoyama, T., Saino, T., Nakamuta, N. et al. Three-dimensional architectures of P2X2-/P2X3-immunoreactive afferent nerve terminals in the rat carotid body as revealed by confocal laser scanning microscopy. Histochem Cell Biol 146, 479–488 (2016). https://doi.org/10.1007/s00418-016-1458-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-016-1458-z