Abstract
Background
To determine the correlation between the area of morphological changes on the macular surface, the depth of intraretinal changes and the best-corrected visual acuity (BCVA) in patients with idiopathic epiretinal membrane.
Methods
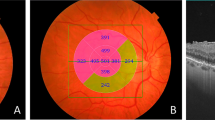
In this prospective cross-sectional study, 38 patients underwent BCVA testing with a Snellen eye chart. The depth of traction, photoreceptor outer segment layer (PROS) thickness, central macular thickness (CMT) and presence of intraretinal cysts were measured using the Avanti RTVue XR and 3D Widefield Enface OCT instruments. Enface sections were performed at the inner limiting membrane (ILM) line level (ILM offset). Patients were categorized into two groups: in group 1 (n = 21), the depth of traction was within 90 μm from the ILM, and in group 2 (n = 17) it was beyond 90 μm from the ILM. The main outcome measure was the correlation between area of epiretinal traction and BCVA according to the depth of traction.
Results
In group 1, the depth of traction was 55.15 ± 14.28 μm, the area of traction was 38.95 ± 13.63 mm2, PROS thickness was 51.20 ± 7.23 μm, and CMT was 362.65 ± 41.08 μm. In group 2, the depth of traction was 112.24 ± 10.89 μm, the area of traction was 25.18 ± 4.07 mm2, PROS thickness was 50.24 ± 9.01 μm, and mean CMT was 534.29 ± 126.81 μm. Statistically significant differences in depth of traction, area of traction and CMT were found between the two groups (P < 0.001). The relationship between structure and function between the area of traction and BCVA was better explained in group 2 (r = 0.814, P < 0.001).
Conclusions
We found that intraretinal changes were induced by the ERM, and varied according to the depth of traction measured with en face analysis. In the case of traction deeper than 90 μm, we found a significant correlation between the inner area of the epiretinal traction and BCVA. We believe that retinal stress induced by epiretinal traction is better characterized by combining information provided by sagittal and transverse OCT scans, thus defining its prognostic significance in ERM.




Similar content being viewed by others
References
Gass JDM (1996) Macular dysfucntions caused by vitreous and vitreretinal interface abnormalities. Stereoscopic atlas of macular diseases. 4th ed. St. Louis, MO: Mosby 938-951.
Mitchell P, Smith W, Chey T, Wang JJ, Chang A (1997) Prevalence and associations of epiretinal membranes. The Blue Mountains Eye Study, Australia. Ophthalmology 104(6):1033–1040
Aung KZ, Makeyeva G, Adams MK et al (2013) The prevalence and risk factors of epiretinal membranes: the Melbourne Collaborative Cohort Study. Retina 33(5):1026–1034
Duan XR, Liang YB, Friedman DS et al (2009) Prevalence and associations of epiretinal membranes in a rural Chinese adult population: the Handan Eye Study. Invest Ophthalmol Vis Sci 50(5):2018–2023
Fraser-Bell S, Guzowski M, Rochtchina E, Wang JJ, Mitchell P (2003) Five-year cumulative incidence and progression of epiretinal membranes: the Blue Mountains Eye Study. Ophthalmology 110(1):34–40
Fraser-Bell S, Ying-Lai M, Klein R, Varma R (2004) Prevalence and associations of epiretinal membranes in latinos: the Los Angeles Latino Eye Study. Invest Ophthalmol Vis Sci 45(6):1732–1736
Kawasaki R, Wang JJ, Aung T et al (2008) Prevalence of age-related macular degeneration in a Malay population: the Singapore Malay Eye Study. Ophthalmology 115(10):1735–1741
Kawasaki R, Wang JJ, Ji GJ et al (2008) Prevalence and risk factors for age-related macular degeneration in an adult Japanese population: the Funagata study. Ophthalmology 115(8):1376–1381, 1381 e1371-1372
Kawasaki R, Wang JJ, Mitchell P et al (2008) Racial difference in the prevalence of epiretinal membrane between Caucasians and Asians. Br J Ophthalmol 92(10):1320–1324
Kawasaki R, Wang JJ, Sato H et al (2009) Prevalence and associations of epiretinal membranes in an adult Japanese population: the Funagata study. Eye (Lond) 23(5):1045–1051
Koh V, Cheung CY, Wong WL et al (2012) Prevalence and risk factors of epiretinal membrane in Asian Indians. Invest Ophthalmol Vis Sci 53(2):1018–1022
Li Y, Xu L, Wang YX, You QS, Yang H, Jonas JB (2008) Prevalence of age-related maculopathy in the adult population in China: the Beijing eye study. Am J Ophthalmol 146(2):329
McCarty DJ, Mukesh BN, Chikani V et al (2005) Prevalence and associations of epiretinal membranes in the visual impairment project. Am J Ophthalmol 140(2):288–294
Ng CH, Cheung N, Wang JJ et al (2011) Prevalence and risk factors for epiretinal membranes in a multi-ethnic United States population. Ophthalmology 118(4):694–699
Xie XW, Xu L, Wang YX, Jonas JB (2008) Prevalence and associated factors of diabetic retinopathy. The Beijing Eye Study 2006. Graefes Arch Clin Exp Ophthalmol 246(11):1519–1526
You Q, Xu L, Jonas JB (2008) Prevalence and associations of epiretinal membranes in adult Chinese: the Beijing Eye Study. Eye (Lond) 22(7):874–879
You QS, Xu L, Jonas JB (2008) Prevalence of crowded optic discs in adult Chinese. The Beijing Eye Study. Graefes Arch Clin Exp Ophthalmol 246(9):1291–1293
You QS, Xu L, Wang YX, Jonas JB (2009) Prevalence of optic disc drusen in an adult Chinese population: the Beijing Eye Study. Acta Ophthalmol 87(2):227–228
Zhu XF, Peng JJ, Zou HD et al (2012) Prevalence and risk factors of idiopathic epiretinal membranes in Beixinjing blocks, Shanghai, China. PLoS One 7(12):e51445
Klein R, Klein BE, Wang Q, Moss SE (1994) The epidemiology of epiretinal membranes. Trans Am Ophthalmol Soc 92:403–425, discussion 425-430
Sebag J (2008) Vitreoschisis. Graefes Arch Clin Exp Ophthalmol 246(3):329–332
Sebag J, Gupta P, Rosen RR, Garcia P, Sadun AA (2007) Macular holes and macular pucker: the role of vitreoschisis as imaged by optical coherence tomography/scanning laser ophthalmoscopy. Trans Am Ophthalmol Soc 105:121–129, discusion 129-131
Rutka JT, Murakami M, Dirks PB et al (1997) Role of glial filaments in cells and tumors of glial origin: a review. J Neurosurg 87(3):420–430
Bringmann A, Pannicke T, Grosche J et al (2006) Muller cells in the healthy and diseased retina. Prog Retin Eye Res 25(4):397–424
Hiscott PS, Grierson I, Trombetta CJ, Rahi AH, Marshall J, McLeod D (1984) Retinal and epiretinal glia--an immunohistochemical study. Br J Ophthalmol 68(10):698–707
Lu YB, Iandiev I, Hollborn M et al (2011) Reactive glial cells: increased stiffness correlates with increased intermediate filament expression. FASEB J 25(2):624–631
Reichenbach A, Bringmann A (2010) Muller cells in the healthy retina. Muller cells in the healthy and diseased retina. London, UK: Springer Science+Business media 53-55.
Kenawy N, Wong D, Stappler T et al (2010) Does the presence of an epiretinal membrane alter the cleavage plane during internal limiting membrane peeling? Ophthalmology 117(2):320–323 e321
Zhang Z, Dong F, Zhao C et al (2015) Natural course of vitreomacular traction syndrome observed by spectral-domain optical coherence tomography. Can J Ophthalmol 50(2):172–179
Kim JH, Kang SW, Kong MG, Ha HS (2013) Assessment of retinal layers and visual rehabilitation after epiretinal membrane removal. Graefes Arch Clin Exp Ophthalmol 251(4):1055–1064
Dyer MA, Cepko CL (2000) Control of Muller glial cell proliferation and activation following retinal injury. Nat Neurosci 3(9):873–880
Francke M, Faude F, Pannicke T et al (2001) Electrophysiology of rabbit Muller (glial) cells in experimental retinal detachment and PVR. Invest Ophthalmol Vis Sci 42(5):1072–1079
Mamballikalathil I, Mann C, Guidry C (2000) Tractional force generation by porcine Muller cells: paracrine stimulation by retinal pigment epithelium. Invest Ophthalmol Vis Sci 41(2):529–536
McGillem GS, Dacheux RF (1998) Migration of retinal microglia in experimental proliferative vitreoretinopathy. Exp Eye Res 67(3):371–375
Erickson PA, Fisher SK, Guerin CJ, Anderson DH, Kaska DD (1987) Glial fibrillary acidic protein increases in Muller cells after retinal detachment. Exp Eye Res 44(1):37–48
Lewis GP, Fisher SK (2003) Up-regulation of glial fibrillary acidic protein in response to retinal injury: its potential role in glial remodeling and a comparison to vimentin expression. Int Rev Cytol 230:263–290
Lewis GP, Guerin CJ, Anderson DH, Matsumoto B, Fisher SK (1994) Rapid changes in the expression of glial cell proteins caused by experimental retinal detachment. Am J Ophthalmol 118(3):368–376
Charteris DG, Downie J, Aylward GW, Sethi C, Luthert P (2007) Intraretinal and periretinal pathology in anterior proliferative vitreoretinopathy. Graefes Arch Clin Exp Ophthalmol 245(1):93–100
McLeod D, Hiscott PS, Grierson I (1987) Age-related cellular proliferation at the vitreoretinal juncture. Eye (Lond) 1(Pt 2):263–281
Gupta P, Sadun AA, Sebag J (2008) Multifocal retinal contraction in macular pucker analyzed by combined optical coherence tomography/scanning laser ophthalmoscopy. Retina 28(3):447–452
Shiono A, Kogo J, Klose G et al (2013) Photoreceptor outer segment length: a prognostic factor for idiopathic epiretinal membrane surgery. Ophthalmology 120(4):788–794
Michalewski J, Michalewska Z, Cisiecki S, Nawrocki J (2007) Morphologically functional correlations of macular pathology connected with epiretinal membrane formation in spectral optical coherence tomography (SOCT). Graefes Arch Clin Exp Ophthalmol 245(11):1623–1631
Mitamura Y, Hirano K, Baba T, Yamamoto S (2009) Correlation of visual recovery with presence of photoreceptor inner/outer segment junction in optical coherence images after epiretinal membrane surgery. Br J Ophthalmol 93(2):171–175
Falkner-Radler CI, Glittenberg C, Hagen S, Benesch T, Binder S (2010) Spectral-domain optical coherence tomography for monitoring epiretinal membrane surgery. Ophthalmology 117(4):798–805
Inoue M, Morita S, Watanabe Y et al (2010) Inner segment/outer segment junction assessed by spectral-domain optical coherence tomography in patients with idiopathic epiretinal membrane. Am J Ophthalmol 150(6):834–839
Okamoto F, Sugiura Y, Okamoto Y, Hiraoka T, Oshika T (2012) Associations between metamorphopsia and foveal microstructure in patients with epiretinal membrane. Invest Ophthalmol Vis Sci 53(11):6770–6775
Duker JS, Kaiser PK, Binder S et al (2013) The International Vitreomacular Traction Study Group classification of vitreomacular adhesion, traction, and macular hole. Ophthalmology 120(12):2611–2619
Acknowledgments
The contributions of the authors were as follows: design and conduct of the study (MRR, GlC); collection (FA, DM, CC), management (FA, DM), analysis (FA, DM, MR), and interpretation of the data (MRR, FA, DM, GC); preparation (MRR, GlC), review, or approval of the manuscript (MRR, GiC); and decision to submit the manuscript for publication (MRR, GlG, GC, MR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements) or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Funding
No funding was received for this research.
Rights and permissions
About this article
Cite this article
Romano, M.R., Cennamo, G., Amoroso, F. et al. Intraretinal changes in the presence of epiretinal traction. Graefes Arch Clin Exp Ophthalmol 255, 31–38 (2017). https://doi.org/10.1007/s00417-016-3413-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-016-3413-z