Abstract
Background
Leber congenital amaurosis (LCA) is a severe retinal dystrophy, typically manifesting in the first year of life. Mutations in more than 18 genes have been reported to date. In recent studies, biallelic mutations in NMNAT1 encoding nicotinamide mononucleotide adenylyltransferase 1 have been found to cause LCA.
Purpose
To broaden the knowledge regarding the phenotype of NMNAT1-associated LCA.
Methods
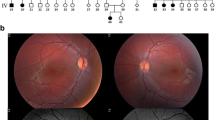
Clinical ophthalmologic examinations were performed in two sisters with LCA. Whole exome sequencing was performed in one of the affected girls, with subsequent segregation analysis in the affected sister and unaffected parents. The literature was reviewed for reports of NMNAT1-associated LCA.
Results
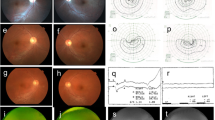
Exome sequencing revealed the known NMNAT1 mutation c.25G>A (p.Val9Met) in a homozygous state. Segregation analysis showed the same homozygous mutation in the affected younger sister. Both parents were found to be heterozygous carriers of the mutation. The two girls both presented with severe visual impairment, nystagmus, central atrophy of the pigment epithelium, and pigment clumping in the periphery before the age of 6 months. Retinal vessels were attenuated. Both children were hyperopic. In the older sister, differential diagnosis included an inflammatory origin, but electrophysiology in her as well as her sister confirmed a diagnosis of LCA. Pallor of the optic nerve head was not present at birth but developed progressively.
Conclusions
We confirmed a diagnosis of NMNAT1-associated LCA in two siblings through identification of the mutation (c.25G>A [p. Val9Met]) in a homozygous state. In infants with non-detectable electroretinogram (ERG), along with severe congenital visual dysfunction or blindness and central pigment epithelium atrophy with pigment clumping resembling scarring due to chorioretinitis, LCA due to NMNAT1 mutations should be considered.



Similar content being viewed by others
References
Michaelides M, Holder GE, Moore A (2005) Leber congenital amaurosis. In: Taylor D, Hoyt GS (eds) Pediatric Ophthalmology and Strabismus. Elsevier Saunders, Edinburgh London New York Oxford Philadelphia St. Louis Sydney Toronto, pp. 539-540
Russell-Eggitt IM, Morris AAM (2005) Retinal dystrophies with systemic associations.In: Taylor D, Hoyt GS (eds) Pediatric Ophthalmology and Strabismus. Elsevier Saunders, Edinburgh London New York Oxford Philadelphia St. Louis Sydney Toronto, pp. 558-574
Weleber RG, Francis PJ, Trzupek KM, Beattie C (2004) Leber Congenital Amaurosis. [Updated 2013 May 2]. In: Pagon RA, Adam MP, Ardinger HH, Wallace SE, Amemiya A, Bean LJH, Bird TD, Dolan CR, Fong CT, Smith RJH, Stephens K (eds)GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2014. http://www.ncbi.nlm.nih.gov/books/NBK1298/ Accessed 7 May 2015
Falk MJ, Zhang Q, Nakamaru-Ogiso E, Kannabiran C, Fonseca-Kelly Z, Chakarova C, Audo I, Mackay DS, Zeitz C, Borman AD, Staniszewska M, Shukla R, Palavalli L, Mohand-Said S, Waseem NH, Jalali S, Perin JC, Place E, Ostrovsky J, Xiao R, Bhattacharya SS, Consugar M, Webster AR, Sahel JA, Moore AT, Berson EL, Liu Q, Gai X, Pierce EA (2012) NMNAT1 mutations cause Leber congenital amaurosis. Nat Genet 44(9):1040–1045. doi:10.1038/ng.2361
Perrault I, Hanein S, Zanlonghi X, Serre V, Nicouleau M, Defoort-Delhemmes S, Delphin N, Fares-Taie L, Gerber S, Xerri O, Edelson C, Goldenberg A, Duncombe A, Le Meur G, Hamel C, Silva E, Nitschke P, Calvas P, Munnich A, Roche O, Dollfus H, Kaplan J, Rozet JM (2012) Mutations in NMNAT1 cause Leber congenital amaurosis with early-onset severe macular and optic atrophy. Nat Genet 44(9):975–977. doi:10.1038/ng.2357
Chiang PW, Wang J, Chen Y, Fu Q, Zhong J, Chen Y, Yi X, Wu R, Gan H, Shi Y, Chen Y, Barnett C, Wheaton D, Day M, Sutherland J, Heon E, Weleber RG, Gabriel LA, Cong P, Chuang K, Ye S, Sallum JM, Qi M (2012) Exome sequencing identifies NMNAT1 mutations as a cause of Leber congenital amaurosis. Nat Genet 44(9):972–974. doi:10.1038/ng.2370
Koenekoop RK, Wang H, Majewski J, Wang X, Lopez I, Ren H, Chen Y, Li Y, Fishman GA, Genead M, Schwartzentruber J, Solanki N, Traboulsi EI, Cheng J, Logan CV, McKibbin M, Hayward BE, Parry DA, Johnson CA, Nageeb M, Finding of Rare Disease Genes (FORGE) Canada Consortium, Poulter JA, Mohamed MD, Jafri H, Rashid Y, Taylor GR, Keser V, Mardon G, Xu H, Inglehearn CF, Fu Q, Toomes C, Chen R (2012) Mutations in NMNAT1 cause Leber congenital amaurosis and identify a new disease pathway for retinal degeneration. Nat Genet 44(9):1035–1039. doi:10.1038/ng.2356
Kaur A (2013) Novel NMNAT1 mutations causing Leber congenital amaurosis identified. Clin Genet 83(1):33–34. doi:10.1111/cge.12043
Siemiatkowska AM, van den Born LI, van Genderen MM, Bertelsen M, Zobor D, Rohrschneider K, van Huet RA, Nurohmah S, Klevering BJ, Kohl S, Faradz SM, Rosenberg T, den Hollander AI, Collin RW, Cremers FP (2014) Novel compound heterozygous NMNAT1 variants associated with Leber congenital amaurosis. Mol Vis 20:753–759, eCollection 2014
Siemiatkowska AM, Schuurs-Hoeijmakers JH, Bosch DG, Boonstra FN, Riemslag FC, Ruiter M, de Vries BB, den Hollander AI, Collin RW, Cremers FP (2014) Nonpenetrance of the most frequent autosomal recessive leber congenital amaurosis mutation in NMNAT1. JAMA Ophthalmol 132(8):1002–1004. doi:10.1001/jamaophthalmol.2014.983
Lau C, Niere M, Ziegler M (2009) The NMN/NaMN adenylyltransferase (NMNAT) protein family. Front Biosci (Landmark Ed) 14:410–431
Coleman MP, Freeman MR (2010) Wallerian degeneration, wld(s), and nmnat. Annu Rev Neurosci 33:245–267. doi:10.1146/annurev-neuro-060909-153248
Zhai RG, Cao Y, Hiesinger PR, Zhou Y, Mehta SQ, Schulze KL, Verstreken P, Bellen HJ (2006) Drosophila NMNAT maintains neural integrity independent of its NAD synthesis activity. PLoS Biol 4(12), e416
Avery MA, Sheehan AE, Kerr KS, Wang J, Freeman MR (2009) Wld S requires Nmnat1 enzymatic activity and N16-VCP interactions to suppress Wallerian degeneration. J Cell Biol 184(4):501–513. doi:10.1083/jcb.200808042
Marmor MF, Fulton AB, Holder GE, Miyake Y, Brigell M, Bach M (2009) ISCEV Standard for full-field clinical electroretinography (2008 update). Doc Ophthalmol 118:69–77. doi:10.1007/s10633-008-9155-4
Holder G, Robson A (2005) Paediatric electrophysiology: a practical approach. In: Lorenz B, Moore AT (eds) Paediatric Ophthalmology and Neuroophthalmology. Essentials in Ophthalmology. Springer, Berlin, pp 133–155
Dharmaraj S, Leroy BP, Sohocki MM, Koenekoop RK, Perrault I, Anwar K, Khaliq S, Devi RS, Birch DG, De Pool E, Izquierdo N, Van Maldergem L, Ismail M, Payne AM, Holder GE, Bhattacharya SS, Bird AC, Kaplan J, Maumenee IH (2004) The phenotype of Leber congenital amaurosis in patients with AIPL1 mutations. Arch Ophthalmol 122(7):1029–1037
Hanein S, Perrault I, Gerber S, Tanguy G, Barbet F, Ducroq D, Calvas P, Dollfus H, Hamel C, Lopponen T, Munier F, Santos L, Shalev S, Zafeiriou D, Dufier JL, Munnich A, Rozet JM, Kaplan J (2004) Leber congenital amaurosis: comprehensive survey of the genetic heterogeneity, refinement of the clinical definition, and genotype-phenotype correlations as a strategy for molecular diagnosis. Hum Mutat 23:306–317
Perrault I, Hanein S, Gerber S, Barbet F, Ducroq D, Dollfus H, Hamel C, Dufier JL, Munnich A, Kaplan J, Rozet JM (2004) Retinal dehydrogenase 12 (RDH12) mutations in leber congenital amaurosis. Am J Hum Genet 75:639–646
Mackay DS, Dev Borman A, Moradi P, Henderson RH, Li Z, Wright GA, Waseem N, Gandra M, Thompson DA, Bhattacharya SS, Holder GE, Webster AR, Moore AT (2011) RDH12 retinopathy: novel mutations and phenotypic description. Mol Vis 17:2706–2716
Weleber RG, Michaelides M, Trzupek KM, Stover NB, Stone EM (2011) The phenotype of severe early childhood onset retinal dystrophy (SECORD) from mutation of RPE65 and differentiation from leber congenital amaurosis. Invest Ophthalmol Vis Sci 52(1):292–302. doi:10.1167/iovs.10-6106
McKibbin M, Ali M, Mohamed MD, Booth AP, Bishop F, Pal B, Springell K, Raashid Y, Jafri H, Inglehearn CF (2010) Genotype-phenotype correlation for leber congenital amaurosis in Northern Pakistan. Arch Ophthalmol 128(1):107–113. doi:10.1001/archophthalmol.2010.309
Hull S, Arno G, Plagnol V, Chamney S, Russell-Eggitt I, Thompson D, Ramsden SC, Black GC, Robson A, Holder GE, Moore AT, Webster AR (2014) The phenotypic variability of retinal dystrophies associated with mutations in CRX, with report of a novel macular dystrophy phenotype. Invest Ophthalmol Vis Sci 55(10):6934–6944. doi:10.1167/iovs.14-14715
Corton M, Nishiguchi KM, Avila-Fernández A, Nikopoulos K, Riveiro-Alvarez R, Tatu SD, Ayuso C, Rivolta C (2013) Exome sequencing of index patients with retinal dystrophies as a tool for molecular diagnosis. PLoS One 8(6):e65574. doi:10.1371/journal.pone.0065574
Volk A, Conboy E, Wical B, Patterson M, Kirmani S (2015) Whole-exome sequencing in the clinic: lessons from six consecutive cases from the Clinician's perspective. MolSyndromol 6(1):23–31. doi:10.1159/000371598
Acknowledgments
The authors are indebted to the patients and their family for the participation in this study.
Compliance with ethical standards
ᅟ
Funding
AEV provided financial support in the form of “Bausteinförderung 3.2” funding by the Medical Faculty of the University of Ulm. The sponsor had no role in the design or conduct of this research.
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers' bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements) or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. Hedergott and A. E. Volk are equally contributing first authors.
C. Kubisch and A. Neugebauer are equally contributing last authors.
Rights and permissions
About this article
Cite this article
Hedergott, A., Volk, A.E., Herkenrath, P. et al. Clinical and genetic findings in a family with NMNAT1-associated Leber congenital amaurosis: case report and review of the literature. Graefes Arch Clin Exp Ophthalmol 253, 2239–2246 (2015). https://doi.org/10.1007/s00417-015-3174-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-015-3174-0