Abstract
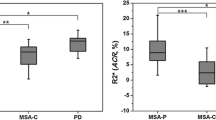
It is a matter of debate whether increased brain iron levels are the cause or only the consequence of neurodegenerative process in degenerative parkinsonism. The aim of this study is to characterize disease-related changes in volumes and iron-related R2* values of basal ganglia and thalamus. 13 patients with progressive supranuclear palsy (PSP), 15 with a parkinsonian variant of multiple system atrophy (MSA-p), 29 with Parkinson’s disease (PD), and 21 age-matched controls underwent 3-Tesla MRI. The R2* values and volumes were calculated for the selected subcortical structures (caudate nucleus, putamen, globus pallidus, and thalamus) using an automated region-based analysis. Voxel-based analysis was also performed to visualize a topographical correlation of R2* value and volume. The PSP group had significantly higher R2* values in globus pallidus and caudate nucleus (p < 0.05), whereas the MSA-p group had higher R2* values in putamen (p < 0.001) than PD and controls. The globus pallidus in PSP and the putamen in MSA-p were the most significant areas of atrophy to differentiate PSP, MSA-p and PD (AUC = 0.856, 0.832, respectively, p < 0.001). The R2* values in both structures increased in parallel with the extent of atrophy. They were negatively correlated with volumes in putamen (r = −0.777, p < 0.001) and globus pallidus (r = −0.409, p = 0.025) of MSA-p, and globus pallidus (r = −0.4, p = 0.043) of PSP. Voxel-based analysis identified higher R2* values in more severely atrophic sub-regions in these structures. We observed topographical differences of iron deposition as well as atrophy between MSA-p and PSP. Increased iron levels were related to the structural atrophy in basal ganglia. Our results imply that iron accumulation is likely an epiphenomenon of the degenerative process.


Similar content being viewed by others
References
Berg D, Hochstrasser H (2006) Iron metabolism in Parkinsonian syndromes. Mov Disord 21:1299–1310
Dusek P, Jankovic J, Le W (2012) Iron dysregulation in movement disorders. Neurobiol Dis 46:1–18
Sian-Hülsmann J, Mandel S, Youdim MB, Riederer P (2011) The relevance of iron in the pathogenesis of Parkinson’s disease. J Neurochem 118:939–957
Vymazal J, Righini A, Brooks RA, Canesi M, Mariani C, Leonardi M et al (1999) T1 and T2 in the brain of healthy subjects, patients with Parkinson disease and patients with multiple system atrophy: relation to iron content. Radiology 211:489–495
Boelmans K, Holst B, Hackius M, Finsterbusch J, Gerloff C, Fiehler J, Münchau A (2012) Brain iron deposition fingerprints in Parkinson’s disease and progressive supranuclear palsy. Mov Disord 27:421–427
Foroutan P, Murray ME, Fujioka S, Schweitzer KJ, Dickson DW, Wszolek ZK, Grant SC (2013) Progressive supranuclear palsy: high-field-strength MR microscopy in the human substantia nigra and globus pallidus. Radiology 266:280–288
Han YH, Lee JH, Kang BM, Mun CW, Baik SK, Shin YI, Park KH (2013) Topographical differences of brain iron deposition between progressive supranuclear palsy and parkinsonian variant multiple system atrophy. J Neurol Sci 325:29–35
Dumas EM, Versluis MJ, van den Bogaard SJ, van Osch MJ, Hart EP, van Roon-Mom WM, van Buchem MA, Webb AG, van der Grond J, Roos RA, TRACK-HD investigators (2012) Elevated brain iron is independent from atrophy in Huntington’s disease. Neuroimage 61:558–564
Khalil M, Enzinger C, Langkammer C, Tscherner M, Wallner-Blazek M, Jehna M, Ropele S, Fuchs S, Fazekas F (2009) Quantitative assessment of brain iron by R(2)* relaxometry in patients with clinically isolated syndrome and relapsing-remitting multiple sclerosis. Mult Scler 15:1048–1054
Smith SM, Jenkinson M, Woolrich MW et al (2004) Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23:S208–S219
Patenaude B, Smith SM, Kennedy DN, Jenkinson M (2011) A Bayesian model of shape and appearance for subcortical brain segmentation. Neuroimage 56:907–922
Péran P, Cherubini A, Luccichenti G, Hagberg G, Démonet JF, Rascol O, Celsis P, Caltagirone C, Spalletta G, Sabatini U (2009) Volume and iron content in basal ganglia and thalamus. Hum Brain Mapp 30:2667–2675
Aquino D, Bizzi A, Grisoli M, Garavaglia B, Bruzzone MG, Nardocci N, Savoiardo M, Chiapparini L (2009) Age-related iron deposition in the basal ganglia: quantitative analysis in healthy subjects. Radiology 252:165–172
Langkammer C, Krebs N, Goessler W, Scheurer E, Ebner F, Yen K, Fazekas F, Ropele S (2010) Quantitative MR imaging of brain iron: a postmortem validation study. Radiology 257:455–462
Litvan I, Agid Y, Calne D, Campbell G, Dubois B, Duvoisin RC et al (1996) Clinical research criteria for the diagnosis of progressive supranuclear palsy (Steel-Richardson-Olszewski syndrome): report of the NINDSSPSP international workshop. Neurology 47:1–9
Gilman S, Low PA, Quinn N, Albanese A, Ben-Shlomo Y, Fowler CJ et al (1999) Consensus statement on the diagnosis of multiple system atrophy. J Neurol Sci 163:94–98
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinicopathological study of 100 cases. J Neurol Neurosurg Psychiatry 55:181–184
Helms G, Dathe H, Weiskopf N, Dechent P (2011) Identification of signal bias in the variable flip angle method by linear display of the algebraic Ernst equation. Magn Reson Med 66:669–677
Weiskopf N, Helms G (2008) Multi-parameter mapping of the human brain at 1 mm resolution in less than 20 minutes In: International Society for Magnetic Resonance in Medicine, editor. Proceedings of the 16th Scientific Meeting ISMRM, p 2241
Draganski B, Ashburner J, Hutton C, Kherif F, Frackowiak RS, Helms G, Weiskopf N (2011) Regional specificity of MRI contrast parameter changes in normal ageing revealed by voxel-based quantification (VBQ). Neuroimage 55:1423–1434
Popescu V, Battaglini M, Hoogstrate WS, Verfaillie SC, Sluimer IC, van Schijndel RA, van Dijk BW, Cover KS, Knol DL, Jenkinson M, Barkhof F, de Stefano N, Vrenken H, MAGNIMS Study Group (2012) Optimizing parameter choice for FSL-Brain Extraction Tool (BET) on 3D T1 images in multiple sclerosis. Neuroimage 61:1484–1494
Mechelli A, Price CJ, Friston KJ, Ashburner J (2005) Voxel-based morphometry of the human brain: methods and applications. Curr Med Imaging Rev 1:1–9
Péran P, Cherubini A, Assogna F, Piras F, Quattrocchi C, Peppe A, Celsis P, Rascol O, Démonet JF, Stefani A, Pierantozzi M, Pontieri FE, Caltagirone C, Spalletta G, Sabatini U (2010) Magnetic resonance imaging markers of Parkinson’s disease nigrostriatal signature. Brain 133:3423–3433
Dexter DT, Jenner P, Schapira AH, Marsden CD (1992) Alterations in levels of iron, ferritin, and other trace metals in neurodegenerative diseases affecting the basal ganglia. The Royal Kings and Queens Parkinson’s disease research group. Ann Neurol 32:S94–S100
Wang Y, Butros SR, Shuai X, Dai Y, Chen C, Liu M et al (2012) Different iron-deposition patterns of multiple system atrophy with predominant parkinsonism and idiopathetic Parkinson diseases demonstrated by phase-corrected susceptibility-weighted imaging. Am J Neuroradiol 33:266–273
Matsusue E, Fujii S, Kanasaki Y, Sugihara S, Miyata H, Ohama E et al (2008) Putaminal lesion in multiple system atrophy: postmortem MR pathological correlations. Neuroradiology 50:559–567
Galazka-Friedman J, Bauminger ER, Szlachta K, Friedman A (2012) The role of iron in neurodegeneration-Mössbauer spectroscopy, electron microscopy, enzyme-linked immunosorbent assay and neuroimaging studies. IOP Publ J Phys Condens Matter. http://stacks.iop.org/JPhysCM/24/244106. Accessed 20 Jun 2012
Messina D, Cerasa A, Condino F, Arabia G, Novellino F, Nicoletti G, Salsone M, Morelli M, Lanza PL, Quattrone A (2011) Patterns of brain atrophy in Parkinson’s disease, progressive supranuclear palsy and multiple system atrophy. Parkinsonism Relat Disord 17:172–176
Saini J, Bagepally BS, Sandhya M, Pasha SA, Yadav R, Thennarasu K, Pal PK (2013) Subcortical structures in progressive supranuclear palsy: vertex-based analysis. Eur J Neurol 20:493–501
Qian ZM, Shen X (2001) Brain iron transport and neurodegeneration. Trends Mol Med 7:103–108
Pérez M, Valpuesta JM, de Garcini EM, Quintana C, Arrasate M, López Carrascosa JL et al (1998) Ferritin is associated with the aberrant tau filaments present in progressive supranuclear palsy. Am J Pathol 152:1531–1539
Olivares D, Huang X, Branden L, Greig NH, Rogers JT (2009) Physiological and pathological role of alpha-synuclein in Parkinson’s disease through iron mediated oxidative stress; the role of a putative iron-responsive element. Int J Mol Sci 10:1226–1260
Graham JM, Paley MN, Grünewald RA, Hoggard N, Griffiths PD (2000) Brain iron deposition in Parkinson’s disease imaged using the PRIME magnetic resonance sequence. Brain 123:2423–2431
Tenner MS, Spiller M, Koenig SH, Valsamis MP, Childress S, Brown RD 3rd, Kasoff SS (1995) Calcification can shorten T2, but not T1, at magnetic resonance imaging fields. Results of a relaxometry study of calcified human meningiomas. Invest Radiol 30:345–353
Haacke EM, Cheng NY, House MJ, Liu Q, Neelavalli J, Ogg RJ et al (2005) Imaging iron stores in the brain using magnetic resonance imaging. Magn Reson Imaging 23:1–25
Karas GB, Scheltens P, Rombouts SA, Visser PJ, van Schijndel RA, Fox NC, Barkhof F (2004) Global and local gray matter loss in mild cognitive impairment and Alzheimer’s disease. Neuroimage 23:708–716
Acknowledgments
This study was supported by a grant from the Pusan National University Yangsan Hospital (2012).
Conflicts of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
J.-H. Lee and Y.-H. Han contributed equally to this study as first authors.
An erratum to this article is available at http://dx.doi.org/10.1007/s00415-015-7670-2.
Rights and permissions
About this article
Cite this article
Lee, JH., Han, YH., Kang, BM. et al. Quantitative assessment of subcortical atrophy and iron content in progressive supranuclear palsy and parkinsonian variant of multiple system atrophy. J Neurol 260, 2094–2101 (2013). https://doi.org/10.1007/s00415-013-6951-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-013-6951-x