Abstract
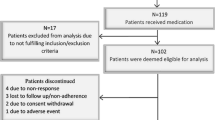
Enhancing quality of life (QoL) is an important objective of disease-modifying therapies in multiple sclerosis (MS). Strategies to substantiate the effect on QoL and depression have been suggested, including injection devices and nursing support. This study assesses QoL and depression in MS patients treated with interferon beta-1b (IFNB-1b) and evaluates the impact of different elements of a patient support programme and of coping strategies on QoL and depression. A prospective, observational, 2-year cohort study was conducted. MS patients were eligible if they had previously switched to IFNB-1b. Data were collected every 6 months. For the measurement of QoL the Functional Assessment of MS (FAMS) was used. Depression symptoms were assessed using the Center for Epidemiologic Studies Depression Scale (CES-D); coping strategies were assessed using the 66-item version of Ways of Coping Questionnaire. A total of 1,077 patients were recruited into the study. Seven hundred (65 %) patients completed the study. Within the subgroup completing questionnaires on QoL (N = 472) and depression (N = 363), QoL increased (110.4 vs. 115.8, p < 0.001), and the proportion of depressed patients decreased from 53.7 to 43.3 % (p < 0.001). Modelling QoL and depressions, the use of the autoinjector Betaject® over time showed a positive association with QoL (p = 0.049). The support from a nurse was positively associated with lower depressive symptoms (p = 0.039). The coping strategies ‘planful problem-solving’ and ‘positive reappraisal’ were associated with higher QoL and lower depressive symptoms. Patients on IFNB-1b treatment who were included in the patient support programme and completed the study showed an improvement in QoL. Moreover, compared to baseline the proportion of depressive patients decreased. Coping strategies as well as supportive elements such as autoinjectors and nurses had a significant impact on QoL and depression. However, the study had the general limitations of a non-controlled design.



Similar content being viewed by others
References
Borras C, Rio J, Porcel J, Barrios M, Tintore M, Montalban X (1999) Emotional state of patients with relapsing-remitting MS treated with interferon beta-1b. Neurology 52:1636–1639
Cella DF, Dineen K, Arnason B, Reder A, Webster KA, Karabatsos G, Chang C, Lloyd S, Steward J, Stefoski D (1996) Validation of the functional assessment of multiple sclerosis quality of life instrument. Neurology 47:129–139
European Study Group on interferon beta-1b in secondary progressive MS (1998) Placebo-controlled multicentre randomised trial of interferon beta-1b in treatment of secondary progressive multiple sclerosis. European Study Group on interferon beta-1b in secondary progressive MS. Lancet 352:1491–1497
Folkman S, Lazarus R (1988) Manual for the ways of coping questionnaire. Consulting Psychology Press, Paolo Alto
Forbes A, While A, Mathes L, Griffiths P (2006) Evaluation of a MS specialist nurse programme. Int J Nurs Stud 43:985–1000
Freeman JA, Thompson AJ, Fitzpatrick R, Hutchinson M, Miltenburger C, Beckmann K, Dahlke F, Kappos L, Polman C, Pozzilli C (2001) Interferon-beta1b in the treatment of secondary progressive MS: impact on quality of life. Neurology 57:1870–1875
Ghubash R, Daradkeh TK, Al Naseri KS, Al Bloushi NB, Al Daheri AM (2000) The performance of the Center for Epidemiologic Study Depression Scale (CES-D) in an Arab female community. Int J Soc Psychiatry 46:241–249
Hourai Y, Brochet B, Grau G, Epicure Study Group (2004) Reduction of injection site reactions of MS patients newly starten on Beterferon® therapy with the Betaject® and Betaject®Light autoinjectors. Mult Scler 10(Suppl 2):252
IFNB Multiple Sclerosis Study Group (1993) Interferon beta-1b is effective in relapsing-remitting multiple sclerosis. I. Clinical results of a multicenter, randomized, double-blind, placebo-controlled trial. The IFNB Multiple Sclerosis Study Group. Neurology 43:655–661
Jacobs LD, Cookfair DL, Rudick RA, Herndon RM, Richert JR, Salazar AM, Fischer JS, Goodkin DE, Granger CV, Simon JH (1995) A phase III trial of intramuscular recombinant interferon beta as treatment for exacerbating-remitting multiple sclerosis: design and conduct of study and baseline characteristics of patients. Multiple Sclerosis Collaborative Research Group (MSCRG). Mult Scler 1:118–135
Johnson KP, Brooks BR, Cohen JA, Ford CC, Goldstein J, Lisak RP, Myers LW, Panitch HS, Rose JW, Schiffer RB (1995) Copolymer 1 reduces relapse rate and improves disability in relapsing-remitting multiple sclerosis: results of a phase III multicenter, double-blind placebo-controlled trial. The Copolymer 1 Multiple Sclerosis Study Group. Neurology 45:1268–1276
Jongen PJ, Sindic C, Carton H, Zwanikken C, Lemmens W, Borm G (2010) Improvement of health-related quality of life in relapsing remitting multiple sclerosis patients after 2 years of treatment with intramuscular interferon-beta-1a. J Neurol 257:584–589
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33:1444–1452
Lily O, McFadden E, Hensor E, Johnson M, Ford H (2006) Disease-specific quality of life in multiple sclerosis: the effect of disease modifying treatment. Mult Scler 12:808–813
Lynch SG, Kroencke DC, Denney DR (2001) The relationship between disability and depression in multiple sclerosis: the role of uncertainty, coping, and hope. Mult Scler 7:411–416
Mikol D, Lopez-Bresnahan M, Taraskiewicz S, Chang P, Rangnow J (2005) A randomized, multicentre, open-label, parallel-group trial of the tolerability of interferon beta-1a (Rebif) administered by autoinjection or manual injection in relapsing-remitting multiple sclerosis. Mult Scler 11:585–591
Miller A, Dishon S (2006) Health-related quality of life in multiple sclerosis: the impact of disability, gender and employment status. Qual Life Res 15:259–271
Miller D, Rudick RA, Hutchinson M (2010) Patient-centered outcomes: translating clinical efficacy into benefits on health-related quality of life. Neurology 74(Suppl 3):S24–S35
Mohr DC, Goodkin DE, Gatto N, Van der Wende J (1997) Depression, coping and level of neurological impairment in multiple sclerosis. Mult Scler 3:254–258
Molyneux PD, Barker GJ, Barkhof F, Beckmann K, Dahlke F, Filippi M, Ghazi M, Hahn D, MacManus D, Polman C, Pozzilli C, Kappos L, Thompson AJ, Wagner K, Yousry T, Miller DH (2001) Clinical-MRI correlations in a European trial of interferon beta-1b in secondary progressive MS. Neurology 57:2191–2197
Montel SR, Bungener C (2007) Coping and quality of life in one hundred and thirty five subjects with multiple sclerosis. Mult Scler 13:393–401
Murrell SA, Himmelfarb S, Wright K (1983) Prevalence of depression and its correlates in older adults. Am J Epidemiol 117:173–185
Neilley LK, Goodin DS, Goodkin DE, Hauser SL (1996) Side effect profile of interferon beta-1b in MS: results of an open label trial. Neurology 46:552–554
Nortvedt MW, Riise T (2003) The use of quality of life measures in multiple sclerosis research. Mult Scler 9:63–72
Nortvedt MW, Riise T, Myhr KM, Nyland HI (1999) Quality of life in multiple sclerosis: measuring the disease effects more broadly. Neurology 53:1098–1103
Paolicelli D, Direnzo V, Trojano M (2009) Review of interferon beta-1b in the treatment of early and relapsing multiple sclerosis. Biologics 3:369–376
Parkin D, Jacoby A, McNamee P, Miller P, Thomas S, Bates D (2000) Treatment of multiple sclerosis with interferon beta: an appraisal of cost-effectiveness and quality of life. J Neurol Neurosurg Psychiatry 68:144–149
Paty DW, Li DK (1993) Interferon beta-1b is effective in relapsing-remitting multiple sclerosis. II. MRI analysis results of a multicenter, randomized, double-blind, placebo-controlled trial. UBC MS/MRI Study Group and the IFNB Multiple Sclerosis Study Group. Neurology 43:662–667
Porcel J, Rio J, Sanchez-Betancourt A, Arevalo MJ, Tintore M, Tellez N, Borras C, Nos C, Montalban X (2006) Long-term emotional state of multiple sclerosis patients treated with interferon beta. Mult Scler 12:802–807
Pozzilli C, Schweikert B, Ecari U, Oentrich W (2011) Supportive strategies to improve adherence to IFN beta-1b in Multiple Sclerosis—Results of the BetaPlus observational cohort study. J Neurol Sci. doi:10.1016/j.jns.2011.04.026:
PRISMS Study Group (1998) Randomised double-blind placebo-controlled study of interferon beta-1a in relapsing/remitting multiple sclerosis. PRISMS (prevention of relapses and disability by interferon beta-1a subcutaneously in multiple sclerosis) Study Group. Lancet 352:1498–1504
Putzki N, Fischer J, Gottwald K, Reifschneider G, Ries S, Siever A, Hoffmann F, Kafferlein W, Kausch U, Liedtke M, Kirchmeier J, Gmund S, Richter A, Schicklmaier P, Niemczyk G, Wernsdorfer C, Hartung HP (2009) Quality of life in 1,000 patients with early relapsing-remitting multiple sclerosis. Eur J Neurol 16:713–720
Rabe-Hesketh S, Skrondal A, Skrondal A (2008) Multilevel and longitudinal modeling using stata. Stata Press Publication, College Station
Radloff L (1977) The CES-D Scale: a self-report depression for research in the general population. Appl Psychol Meas 1:385–401
Rudick RA, Miller D, Hass S, Hutchinson M, Calabresi PA, Confavreux C, Galetta SL, Giovannoni G, Havrdova E, Kappos L, Lublin FD, Miller DH, O’Connor PW, Phillips JT, Polman CH, Radue EW, Stuart WH, Wajgt A, Weinstock-Guttman B, Wynn DR, Lynn F, Panzara MA (2007) Health-related quality of life in multiple sclerosis: effects of natalizumab. Ann Neurol 62:335–346
Rudick RA, Polman CH (2009) Current approaches to the identification and management of breakthrough disease in patients with multiple sclerosis. Lancet Neurol 8:545–559
Shinar D, Gross CR, Price TR, Banko M, Bolduc PL, Robinson RG (1986) Screening for depression in stroke patients: the reliability and validity of the Center for Epidemiologic Studies Depression Scale. Stroke 17:241–245
Simone IL, Ceccarelli A, Tortorella C, Bellacosa A, Pellegrini F, Plasmati I, De Caro MF, Lopez M, Girolamo F, Livrea P (2006) Influence of interferon beta treatment on quality of life in multiple sclerosis patients. Health Qual Life Outcomes 4:96
Thompson AJ, Toosy AT, Ciccarelli O (2010) Pharmacological management of symptoms in multiple sclerosis: current approaches and future directions. Lancet Neurol 9:1182–1199
Turpin KV, Carroll LJ, Cassidy JD, Hader WJ (2007) Deterioration in the health-related quality of life of persons with multiple sclerosis: the possible warning signs. Mult Scler 13:1038–1045
Vermersch P, de Seze J, Delisse B, Lemaire S, Stojkovic T (2002) Quality of life in multiple sclerosis: influence of interferon-beta1 a (Avonex) treatment. Mult Scler 8:377–381
Vickrey BG, Hays RD, Harooni R, Myers LW, Ellison GW (1995) A health-related quality of life measure for multiple sclerosis. Qual Life Res 4:187–206
Zivadinov R, Zorzon M, Tommasi MA, Nasuelli D, Bernardi M, Monti-Bragadin L, Cazzato G (2003) A longitudinal study of quality of life and side effects in patients with multiple sclerosis treated with interferon beta-1a. J Neurol Sci 216:113–118
Acknowledgment
This research was funded by Bayer Pharma AG.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pozzilli, C., Schweikert, B., Ecari, U. et al. Quality of life and depression in multiple sclerosis patients: longitudinal results of the BetaPlus study. J Neurol 259, 2319–2328 (2012). https://doi.org/10.1007/s00415-012-6492-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-012-6492-8