Abstract
Background
Cytokine inhibitors, such as soluble tumor necrosis factor receptor II (sTNFRII) and interleukin-1 receptor antagonist (IL-1ra) are possible regulators of proinflammatory cytokine activity. Although previous studies have shown induction of sTNF-RII and IL-1ra by interferon-beta (IFNβ) in patients with relapse-onset forms of multiple sclerosis (MS), to date no studies of these cytokine inhibitors have been performed in patients with essentially progressive forms of MS.
Objective
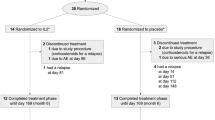
To address the effects of IFNβ on serum levels of sTNF-RII and IL-1ra in a cohort of progressive MS patients and to assess the relationship between levels and changes of sTNF-RII and IL-1ra and clinical and radiological variables. Methods Serial blood samples were obtained from a cohort of 73 patients with progressive MS who participated in a placebo-controlled clinical trial with IFNβ-1b. Serum levels of sTNF-RII and IL-1ra were measured by multiplex enzyme-linked immunosorbent assay at baseline and after 3, 6, 12, and 24 months. EDSS and MSFC scores were recorded at the time of blood sampling, and MR scans were obtained at baseline and after 12 and 24 months.
Results
Treatment with IFNβ was associated with significant increases of sTNF-RII and IL-1ra serum levels during the followup period. A strong correlation at 24 months was observed between levels of sTNF-RII and EDSS scores in the placebo group. Finally, a trend for negative association was found between changes in sTNFRII and percentage change in T2-weighted lesion load at 24 months in the IFNβ treated group.
Conclusions
sTNF-RII and IL-1ra levels are increased in the serum of progressive MS patients during IFNβ therapy and may be one mechanism by which IFNβ mediates its effects in the treatment of MS.
Similar content being viewed by others
References
Aderka D, Engelmann H, Maor Y, Brakebusch C, Wallach D (1992) Stabilization of the bioactivity of tumor necrosis factor by its soluble receptors. J Exp Med 175:323–329
Aymerich FX, Sobrevilla P, Montseny E, Gili J, Rovira A (2001) Fuzzy approach to lesion load measurement in T1-weighted images. Mult Scler 7:S87
Coclet-Ninin J, Dayer JM, Burger D (1997) Interferon-beta not only inhibits interleukin-1beta and tumor necrosis factor-alpha but stimulates interleukin- 1 receptor antagonist production in human peripheral blood mononuclear cells. Eur Cytokine Netw 8:345–349
Cutter GR, Baier ML, Rudick RA, Cookfair DL, Fischer JS, Petkau J, Syndulko K, Weinshenker BG, Antel JP, Confavreux C, Ellison GW, Lublin F, Miller AE, Rao SM, Reingold S, Thompson A, Willoughby E (1999) Development of a multiple sclerosis functional composite as a clinical trial outcome measure. Brain 122:871–882
Dripps DJ, Brandhuber BJ, Thompson RC, Eisenberg SP (1991) Interleukin-1 (IL-1) receptor antagonist binds to the 80-kDa IL-1 receptor but does not initiate, IL-1 signal transduction. J Biol Chem 266:10331–10336
Duran I, Martinez-Caceres EM, Rio J, Barbera N, Marzo ME, Montalban X (1999) Immunological profile of patients with primary progressive multiple sclerosis. Expression of adhesion molecules. Brain 122:2297–2307
Engelmann H, Aderka D, Rubinstein M, Rotman D, Wallach D (1989) A tumor necrosis factor-binding protein purified to homogeneity from human urine protects cells from tumor necrosis factor toxicity. J Biol Chem 264:11974–11980
Granowitz EV, Clarck BD, Mancilla C, Dinarello CA (1991) Interleukin-1 receptor antagonist competitively inhibits the binding of interleukin-1 to the type II interleukin receptor. J Biol Chem 266:14147–14150
Jacobs LD, Cookfair DL, Rudick RA, Herndon RM, Richert JR, Salazar AM, Fischer JS, Goodkin DE, Granger CV, Simon JH, Alam JJ, Bartoszak DM, Bourdette DN, Braiman J, Brownscheidle CM, Coats ME, Cohan SL, Dougherty DS, Kinkel RP, Mass MK, Munschauer FE 3rd, Priore RL, Pullicino PM, Scherokman BJ, Whitham RH, et al. (1996) Intramuscular interferon beta-1a for disease progression in relapsing multiple sclerosis. The Multiple Sclerosis Collaborative Research Group (MSCRG). Ann Neurol 39:285–294
Kurtzke JF (1983) Rating neurological impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33:1444–1452
Laske C, Oschmann P, Tofighi J, Kuehne SB, Diehl H, Bregenzer T, Kraus J, Bauer R, Chatzimanolis N, Kern A, Traupe H, Kaps M (2001) Induction of sTNF-R1 and sTNF-R2 by interferon beta-1b in correlation with clinical and MRI activity. Acta Neurol Scand 103:105–113
Rieckmann P, Albrecht M, Kitze B, Weber T, Tumani H, Broocks A, Luer W, Helwig A, Poser S (1995) Tumor necrosis factor-alpha messenger RNA expression in patients with relapsing-remitting multiple sclerosis is associated with disease activity. Ann Neurol 37:82–88
Liu J, Marino MW, Wong G, Grail D, Dunn A, Bettadapura J, Slavin AJ, Old L, Bernard CC (1998) TNF is a potent antiinflammatory cytokine in autoimmunemediated demyelination. Nat Med 4:78–83
Lublin FD, Reingold SC (1996) Defining the clinical course of multiple sclerosis: results of an international survey. Neurology 46:907–911
Mohler KM, Torrance DS, Smith CA, Goodwin RG, Stremler KE, Fung VP, Madani H, Widmer MB (1993) Soluble tumor necrosis factor (TNF) receptors are effective therapeutic agents in lethal endotoxemia and function simultaneously as both TNF carriers and TNF antagonists. J Immunol 151:1548–1561
Molyneux PD, Miller DH, Filippi M, Yousry TA, Radu EW, Ader HJ, Barkhof F (1999) Visual analysis of serial T2-weighted MRI in multiple sclerosis: intra- and interobserver reproducibility. Neuroradiology 41:882–888
Montalban X (2004) Overview of European pilot study of interferon β-1b in primary progressive multiple sclerosis. Mult Scler 10:S62–S64
Nicoletti F, Patti F, Di Marco R, Zaccone P, Nicoletti A, Meroni P, Reggio A (1996) Circulating serum levels of IL-1ra in patients with relapsing-remitting multiple sclerosis are normal during remission phases but significantly increased either during exacerbations or in response to IFNbeta treatment. Cytokine 8:395–400
Perini P, Tiberio M, Sivieri S, Facchinetti A, Biasi G, Gallo P (2000) Interleukin-1 receptor antagonist, soluble tumor necrosis factor-a receptor type I and II, and soluble E-selectin serum levels in multiple sclerosis patients receiving weekly intramuscular injections of interferon- b1a. Eur Cytokine Netw 11:81–86
Porteu T, Nathan C (1990) Shedding of tumor necrosis factor receptors by activated human neutrophils. J Exp Med 172:599–607
PRISMS (Prevention of Relapses and Disability by Interferon beta-1a Subcutaneously in Multiple Sclerosis) Study Group (1998) Randomised double-blind placebo-controlled study of interferon beta-1a in relapsing/remitting multiple sclerosis. Lancet 352:1498–1504
Rieckmann P, Kruse N, Nagelkerken L, Beckmann K, Miller D, Polman C, Dahlke F, Toyka KV, Hartung HP, Sturzebecher S (2005) Soluble vascular cell adhesion molecule (VCAM) is associated with treatment effects of Interferon beta-1b in patients with Secondary Progressive Multiple Sclerosis. J Neurol 252:526–533
Rio J, Nos C, Tintore M, Borras C, Galan I, Comabella M, Montalban X (2002) Assessment of different treatment failure criteria in a cohort of relapsingremitting multiple sclerosis patients treated with interferon beta: implications for clinical trials. Ann Neurol 52:400–406
Seckinger P, Isaaz S, Dayer JM (1989) Purification and biologic characterization of a specific tumor necrosis factor alpha inhibitor. J Biol Chem 264:11966–11973
Siebert H, Bruck W (2003) The role of cytokines and adhesion molecules in axon degeneration after peripheral nerve axotomy: a study in different knockout mice. Brain Res 960:152–156
The Interferon β Multiple Sclerosis Study Group (1993) Interferon beta-1b is effective in relapsing-remitting multiple sclerosis. I. Clinical results of a multicenter, randomized, double-blind, placebo-controlled trial. Neurology 43:655–661
Thompson AJ, Polman C, Miller CH, McDonald WI, Brochet B, Filippi M, Montalban X, De Sa J (1997) Primary progressive multiple sclerosis. Brain 120:1085–1096
van Oosten BW, Barkhof F, Truyen L, Boringa JB, Bertelsmann FW, von Blomberg BM, Woody JN, Hartung HP, Polman CH (1996) Increased MRI activity and immune activation in two multiple sclerosis patients treated with the monoclonal anti-tumor necrosis factor antibody cA2. Neurology 47:1531–1534
The Lerencept Multiple Sclerosis Study Group (1999) TNF neutralization in MS: results of a randomized, placebo-controlled multicenter study. The Lenercept Multiple Sclerosis Study Group and The University of British Columbia MS/MRI Analysis Group. Neurology 53:457–465
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Comabella, M., Julià, E., Tintoré, M. et al. Induction of serum soluble tumor necrosis factor receptor II (sTNF-RII) and interleukin-1 receptor antagonist (IL-1ra) by interferon beta-1b in patients with progressive multiple sclerosis. J Neurol 255, 1136–1141 (2008). https://doi.org/10.1007/s00415-008-0855-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-008-0855-1