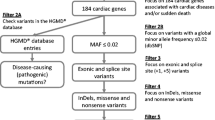
Abstract
Sudden unexplained death (SUD) constitutes a considerable portion of unexpected sudden death in the young. Molecular autopsy has proved to be an efficient diagnostic tool in the multidisciplinary management of SUD. Yet, many cases remain undiagnosed using the widely adopted targeted genetic screening strategies. Here, we investigated the genetic substrates of a young SUD cohort (18–40 years old) from China using whole-exome sequencing (WES), with the primary aim to identify novel SUD susceptibility genes. Within 255 previously acknowledged SUD-associated genes, 21 variants with likely functional effects (pathogenic/likely pathogenic) were identified in 51.9% of the SUD cases. More importantly, a set of 33 candidate genes associated with myopathy were identified to be novel susceptibility genes for SUD. Comparative analysis of the cumulative PHRED-scaled CADD score and polygenetic burden score showed that the amount and deleteriousness of variants in the 255 SUD-associated genes and the 33 candidate genes identified by this study were significantly higher compared with 289 randomly selected genes. A significantly higher genetic burden of rare variants (MAF < 0.1%) in the 33 candidate genes also highlighted putative roles of these genes in SUD. After incorporating these novel genes, the genetic testing yields of the current SUD cohort elevated from 51.9 to 66.7%. Our study expands understanding of the genetic variants underlying SUD and presents insights that improve the utility of genetic screenings.



Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the first or corresponding authors upon reasonable request.
References
Lucena JS (2019) Sudden cardiac death. Forensic Sci Res 4:199–201
Couper K, Putt O, Field R et al (2020) Incidence of sudden cardiac death in the young: a systematic review. BMJ Open 10:e040815
Morentin B, Suárez-Mier MP, Aguilera B (2003) Sudden unexplained death among persons 1–35 years old. Forensic Sci Int 135:213–217
Puranik R, Chow CK, Duflou JA et al (2005) Sudden death in the young. Heart Rhythm 2:1277–1282
Winkel BG, Holst AG, Theilade J et al (2011) Nationwide study of sudden cardiac death in persons aged 1–35 years. Eur Heart J 32:983–990
Doolan A, Semsarian C, Langlois N (2004) Causes of sudden cardiac death in young Australians. Med J Aust 180:110–112
Morris VB, Keelan T, Leen E et al (2009) Sudden cardiac death in the young: a 1-year post-mortem analysis in the Republic of Ireland. Ir J Med Sci 178:257–261
van der Werf C, van Langen IM, Wilde AA (2010) Sudden death in the young: what do we know about it and how to prevent? Circ Arrhythm Electrophysiol 3:96–104
Raju H, Parsons S, Thompson TN et al (2019) Insights into sudden cardiac death: exploring the potential relevance of non-diagnostic autopsy findings. Eur Heart J 40:831–838
Rizzo S, Carturan E, De Gaspari M et al (2019) Update on cardiomyopathies and sudden cardiac death. Forensic Sci Res 4:202–210
Ackerman M, Atkins DL, Triedman JK (2016) Sudden cardiac death in the young. Circulation 133:1006–1026
Brion M, Blanco-Verea A, Sobrino B et al (2014) Next generation sequencing challenges in the analysis of cardiac sudden death due to arrhythmogenic disorders. Electrophoresis 35:3111–3116
Farrugia A, Keyser C, Hollard C et al (2015) Targeted next generation sequencing application in cardiac channelopathies: analysis of a cohort of autopsy-negative sudden unexplained deaths. Forensic Sci Int 254:5–11
Hertz CL, Christiansen SL, Ferrero-Miliani L et al (2015) Next-generation sequencing of 34 genes in sudden unexplained death victims in forensics and in patients with channelopathic cardiac diseases. Int J Legal Med 129:793–800
Hata Y, Kinoshita K, Mizumaki K et al (2016) Postmortem genetic analysis of sudden unexplained death syndrome under 50 years of age: a next-generation sequencing study. Heart Rhythm 13:1544–1551
Christiansen SL, Hertz CL, Ferrero-Miliani L et al (2016) Genetic investigation of 100 heart genes in sudden unexplained death victims in a forensic setting. Eur J Hum Genet 24:1797–1802
Anderson JH, Tester DJ, Will ML et al (2016) Whole-exome molecular autopsy after exertion-related sudden unexplained death in the young. Circ Cardiovasc Genet 9:259–265
Neubauer J, Lecca MR, Russo G et al (2018) Exome analysis in 34 sudden unexplained death (SUD) victims mainly identified variants in channelopathy-associated genes. Int J Legal Med 132:1057–1065
Andersen JD, Jacobsen SB, Trudso LC et al (2019) Whole genome and transcriptome sequencing of post-mortem cardiac tissues from sudden cardiac death victims identifies a gene regulatory variant in NEXN. Int J Legal Med 133:1699–1709
Larsen MK, Christiansen SL, Hertz CL et al (2020) Targeted molecular genetic testing in young sudden cardiac death victims from Western Denmark. Int J Legal Med 134:111–121
Moon RY, Horne RSC, Hauck FR (2007) Sudden infant death syndrome. Lancet 370:1578–1587
Zheng J, Zheng D, Su T et al (2018) Sudden unexplained nocturnal death syndrome: the hundred years’ enigma. J Am Heart Assoc 7:e007837
Markwerth P, Bajanowski T, Tzimas I et al (2021) Sudden cardiac death-update. Int J Legal Med 135:483–495
Li H (2013) Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM. ArXiv 1303.3997. https://doi.org/10.48550/arXiv.1303.3997
Freed D, Aldana R, Weber JA et al (2017) The Sentieon Genomics tools—a fast and accurate solution to variant calling from next-generation sequence data. bioRxiv 115717v2. https://doi.org/10.1101/115717
Wang K, Li M, Hakonarson H (2010) ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 38:e164
Talevich E, Shain AH, Botton T et al (2016) CNVkit: genome-wide copy number detection and visualization from targeted DNA sequencing. PLoS Comput Biol 12:e1004873
Neubauer J, Wang S, Russo G et al (2021) Re-evaluation of single nucleotide variants and identification of structural variants in a cohort of 45 sudden unexplained death cases. Int J Legal Med 135:1341–1349
Karczewski KJ, Francioli LC, Tiao G et al (2020) The mutational constraint spectrum quantified from variation in 141,456 humans. Nature 581:434–443
Kopanos C, Tsiolkas V, Kouris A et al (2019) VarSome: the human genomic variant search engine. Bioinformatics 35:1978–1980
Richards S, Aziz N, Bale S et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–424
Neubauer J, Kissel CK, Bolliger SA et al (2022) Benefits and outcomes of a new multidisciplinary approach for the management and financing of sudden unexplained death cases in a forensic setting in Switzerland. Forensic Sci Int 334:111240
Rentzsch P, Schubach M, Shendure J et al (2021) CADD-Splice-improving genome-wide variant effect prediction using deep learning-derived splice scores. Genome Med 13:31
Zhou Y, Zhou B, Pache L et al (2019) Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat Commun 10:1523
Christiansen SL, Andersen JD, Themudo GE et al (2021) Genetic investigations of 100 inherited cardiac disease-related genes in deceased individuals with schizophrenia. Int J Legal Med 135:1395–1405
Guo MH, Plummer L, Chan YM et al (2018) Burden testing of rare variants identified through exome sequencing via publicly available control data. Am J Hum Genet 103:522–534
Salfati EL, Spencer EG, Topol SE et al (2019) Re-analysis of whole-exome sequencing data uncovers novel diagnostic variants and improves molecular diagnostic yields for sudden death and idiopathic diseases. Genome Med 11:83
Modena M, Castiglione V, Aretini P et al (2020) Unveiling a sudden unexplained death case by whole exome sequencing and bioinformatic analysis. Mol Genet Genomic Med 8:e1182
Schön U, Holzer A, Laner A et al (2021) HPO-driven virtual gene panel: a new efficient approach in molecular autopsy of sudden unexplained death. BMC Med Genomics 14:94
Kong T, Feulefack J, Ruether K et al (2017) Ethnic differences in genetic ion channelopathies associated with sudden cardiac death: a systematic review and meta-analysis. Ann Clin Lab Sci 47:481–490
Wang C, Duan S, Lv G et al (2015) Using whole exome sequencing and bioformatics in the molecular autopsy of a sudden unexplained death syndrome (SUDS) case. Forensic Sci Int 257:e20–e25
Lin Y, He S, Liao Z et al (2018) Whole exome sequencing identified a pathogenic mutation in RYR2 in a Chinese family with unexplained sudden death. J Electrocardiol 51:309–315
Huikuri HV, Castellanos A, Myerburg RJ (2001) Sudden death due to cardiac arrhythmias. N Engl J Med 345:1473–1482
Saenen JB, Van Craenenbroeck EM, Proost D et al (2015) Genetics of sudden cardiac death in the young. Clin Genet 88:101–113
Weese-Mayer DE, Silvestri JM, Marazita ML et al (1993) Congenital central hypoventilation syndrome: inheritance and relation to sudden infant death syndrome. Am J Med Genet 47:360–367
Gronli JO, Santucci BA, Leurgans SE et al (2008) Congenital central hypoventilation syndrome: PHOX2B genotype determines risk for sudden death. Pediatr Pulmonol 43:77–86
Liebrechts-Akkerman G, Liu F, Lao O et al (2014) PHOX2B polyalanine repeat length is associated with sudden infant death syndrome and unclassified sudden infant death in the Dutch population. Int J Legal Med 128:621–629
Ventura F, Barranco R, Bachetti T et al (2018) Medico-legal investigation in an explicable case of congenital central hypoventilation syndrome due to a rare variant of the PHOX2B gene. J Forensic Leg Med 58:1–5
Bachetti T, Bagnasco S, Piumelli R et al (2021) A common 3′UTR variant of the PHOX2B gene is associated with infant life-threatening and sudden death events in the Italian population. Front Neurol 12:642735
Hino A, Terada J, Kasai H et al (2020) Adult cases of late-onset congenital central hypoventilation syndrome and paired-like homeobox 2B-mutation carriers: an additional case report and pooled analysis. J Clin Sleep Med 16:1891–1900
Lopez-Sainz A, Dominguez F, Lopes LR et al (2020) Clinical features and natural history of PRKAG2 variant cardiac glycogenosis. J Am Coll Cardiol 76:186–197
Burchell A, Busuttil A, Bell J et al (1989) Hepatic microsomal glucose-6-phosphatase system and sudden infant death syndrome. Lancet 334:291–294
Forsyth L, Scott HM, Howatson A et al (2007) Genetic variation in hepatic glucose-6-phosphatase system genes in cases of sudden infant death syndrome. J Pathol 212:112–120
Miani D, Taylor M, Mestroni L et al (2012) Sudden death associated with Danon disease in women. Am J Cardiol 109:406–411
Hu D, Hu D, Liu L et al (2020) Identification, clinical manifestation and structural mechanisms of mutations in AMPK associated cardiac glycogen storage disease. EBioMedicine 54:102723
Arbustini E, Di Toro A, Giuliani L et al (2018) Cardiac phenotypes in hereditary muscle disorders: JACC state-of-the-art review. J Am Coll Cardiol 72:2485–2506
Karmakar S, Sankhla A, Katiyar V (2021) Supramolecular organization of cytochrome-C into quantum-dot decorated macromolecular network under pH and thermal stress. Int J Biol Macromol 193:1623–1634
Acknowledgements
We thank Dr. Cordula Haas and Dr. Jacqueline Neubauer from the Zurich Institute of Forensic Medicine (University of Zurich, Switzerland) for their guidance on data interpretation. We also feel grateful to Dr. Fanqian Yin from the Kunming Institute of Zoology (Chinese Academy of Sciences, China) for bioinformatics support.
Funding
This study was supported by the National Science Foundation of China (82070285, 82101971 and 82322033), the National Postdoctoral Science Foundation of China (2021M700815), and the Student Scientific Research Program of Shanghai Medical College, Fudan University (QF2203). The authors express their gratitude to the foundations that supported this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Informed consent was obtained from relatives of all individual participants included in the study before sample collection. The use of collected human samples for research purpose was approved by the Institutional Review Board at the School of Basic Medical Sciences, Fudan University (approval No.: 2021-C017). All procedures performed in studies were in accordance with the local ethical standards and principles outlined in the Declaration of Helsinki.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information

ESM 1
Top 60 pathways and processes clusters identified by enrichment analysis. The p values are calculated based on the cumulative hypergeometric distribution. (PNG 905 kb)

ESM 2
Canonical pathway, KEGG pathway and GO biological processes identified by enrichment analysis for the 20 myopathy-associated genes. (PNG 1647 kb)

ESM 3
Significant differences of polygenetic burden scores (the black dots) observed among groups (***p < 0.001). In addition, the amount of rare variants within exonic or splicing region in Group S (374) and Group C (266) was higher compared with Group R (208). (PNG 57 kb)
ESM 4
(XLSX 11 kb)
ESM 5
(XLSX 10 kb)
ESM 6
(XLSX 20 kb)
ESM 7
(XLSX 10 kb)
ESM 8
(XLSX 11 kb)
ESM 9
(XLSX 11 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, S., Chen, Y., Du, J. et al. Post-mortem genetic analysis of sudden unexplained death in a young cohort: a whole-exome sequencing study. Int J Legal Med 137, 1661–1670 (2023). https://doi.org/10.1007/s00414-023-03075-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-023-03075-1