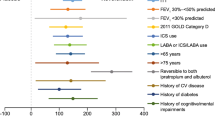
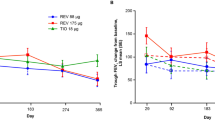
Abstract
Purpose
There are limited studies on the use of bronchodilators for the treatment of bronchiectasis. This study investigated the efficacy of tiotropium in patients with bronchiectasis and airflow limitation.
Methods
This study was a prospective cohort study, including 169 patients with bronchiectasis and airflow limitation from 2015 to 2019. The clinical outcomes observed in our study were the effect of tiotropium on the frequency of moderate exacerbations, the time to the first severe exacerbation, and the annual decline in FEV1.
Results
After 12 months, the annual decline in the FEV1 after bronchodilator use was 27.08 ml or 42.9 ml per year in the group with or without tiotropium, respectively. Treatment with tiotropium was associated with a decreased risk of moderate exacerbation of bronchiectasis (Adjusted RR 0.618 95% CI 0.493–0.774; P < 0.005). The time to the first severe acute exacerbation of bronchiectasis in the tiotropium group was longer than the non-tiotropium group (Adjusted HR 0.333 95% CI 0.219–0.506; P < 0.001).
Conclusion
In conclusion, prospective cohort study showed that tiotropium effectively ameliorated the annual decline in the FEV1, with a lower-risk rate of moderate exacerbations and prolonging the time to the first-time severe exacerbation in patients with bronchiectasis and airflow limitation.

Similar content being viewed by others
Abbreviations
- COPD:
-
Chronic obstructive pulmonary disease
- LABA:
-
Long-acting β2 agonist
- LAMA:
-
Long-acting muscarinic antagonist
- BMI:
-
Body Mass Index
- FEV1:
-
Forced expiratory volume in 1 s
- FVC:
-
Forced vital capacity
- RR:
-
Relative risk
References
Polverino E, Goeminne PC, McDonnell MJ et al (2017) European respiratory society guidelines for the management of adult bronchiectasis. Eur Respir J 50(3):1700629. https://doi.org/10.1183/13993003.00629-2017
Cole PJ (1986) Inflammation: a two-edged sword–the model of bronchiectasis. Eur J Respir Dis Suppl 147:6–15
Guan WJ, Yuan JJ, Gao YH et al (2016) Impulse oscillometry and spirometry small airway parameters in mild to moderate bronchiectasis. Respir Care 61(11):1513–1522. https://doi.org/10.4187/respcare.04710
Guan WJ, Yuan JJ, Huang Y, Li HM, Chen RC, Zhong NS (2017) Residual volume/total lung capacity ratio confers limited additive significance to lung clearance index for assessment of adults with bronchiectasis. PLoS One 12(9):e0183779. https://doi.org/10.1371/journal.pone.0183779
Jeong HJ, Lee H, Carriere KC, Kim JH, Han JH, Shin B, Jeong BH, Koh WJ, Kwon OJ, Park HY (2016) Effects of long-term bronchodilators in bronchiectasis patients with airflow limitation based on bronchodilator response at baseline. Int J Chron Obstruct Pulmon Dis 7(11):2757–2764. https://doi.org/10.2147/COPD.S115581
Lee SY, Lee JS, Lee SW, Oh YM (2021) Effects of treatment with long-acting muscarinic antagonists (LAMA) and long-acting beta-agonists (LABA) on lung function improvement in patients with bronchiectasis: an observational study. J Thorac Dis 13(1):169–177. https://doi.org/10.21037/jtd-20-1282
Martínez-García MÁ, Oscullo G, García-Ortega A, Matera MG, Rogliani P, Cazzola M (2022) Rationale and clinical use of bronchodilators in adults with bronchiectasis. Drugs 82(1):1–13. https://doi.org/10.1007/s40265-021-01646-3
Hill AT et al (2019) British thoracic society guideline for bronchiectasis in adults. Thorax 74(Suppl1):169. https://doi.org/10.1136/thoraxjnl2018212463
Polverino E, Goeminne PC, McDonnell MJ, Aliberti S, Marshall SE, Loebinger MR et al (2017) European respiratory society guidelines for the management of adult bronchiectasis. Eur Respir J 50(3):1700629. https://doi.org/10.1183/13993003.00629-2017
Martínez-García MÁ, Máiz L, Olveira C, Girón RM, de la Rosa D, Blanco M et al (2018) Spanish guidelines on treatment of bronchiectasis in adults. Arch Bronconeumol (Engl Ed) 54(2):88–98. https://doi.org/10.1016/j.arbres.2017.07.016
Miller MR, Hankinson J, Brusasco V et al (2005) Standardisation of spirometry. Eur Respir J 26(2):319–338. https://doi.org/10.1183/09031936.05.00034805
Tsikrika S, Dimakou K, Papaioannou AI et al (2017) The role of non-invasive modalities for assessing inflammation in patients with non-cystic fibrosis bronchiectasis. Cytokine 99:281–286. https://doi.org/10.1016/j.cyto.2017.08.005
Clofent D, Álvarez A, Traversi L, Culebras M, Loor K, Polverino E (2021) Comorbidities and mortality risk factors for patients with bronchiectasis. Expert Rev Respir Med 15(5):623–634. https://doi.org/10.1080/17476348.2021.1886084
Radovanovic D, Santus P, Blasi F, Sotgiu G, D’Arcangelo F, Simonetta E, Contarini M, Franceschi E, Goeminne PC, Chalmers JD, Aliberti S (2018) A comprehensive approach to lung function in bronchiectasis. Respir Med 145:120–129. https://doi.org/10.1016/j.rmed.2018.10.031
White AJ, O’Brien C, Hill SL, Stockley RA (2005) Exacerbations of COPD diagnosed in primary care: changes in spirometry and relationship to symptoms. COPD 2(4):419–425. https://doi.org/10.1080/15412550500346477
Guan W-j, Gao Y-h, Xu G, Lin Z-y, Tang Y et al (2014) Characterization of lung function impairment in adults with bronchiectasis. PLoS ONE 9(11):e113373. https://doi.org/10.1371/journal.pone.0113373
Håkansson KEJ, Fjaellegaard K, Browatzki A, Dönmez Sin M, Ulrik CS (2021) Inhaled corticosteroid therapy in bronchiectasis is associated with all-cause mortality: a prospective cohort study. Int J Chron Obstruct Pulmon Dis 16(16):2119–2127. https://doi.org/10.2147/COPD.S311236
Vogelmeier C, Hederer B, Glaab T, Schmidt H, Rutten-van Mölken MP, Beeh KM, Rabe KF, Fabbri LM, POET-COPD Investigators (2011) Tiotropium versus salmeterol for the prevention of exacerbations of COPD. N Engl J Med 364(12):1093–1103. https://doi.org/10.1056/NEJMoa1008378
Martinez-Garcia MA, Polverino E, Aksamit T (2018) Bronchiectasis and chronic airway disease: it is not just about asthma and COPD. Chest 154(4):737–739. https://doi.org/10.1016/j.chest.2018.02.024
Lonni S, Chalmers JD, Goeminne PC et al (2015) Etiology of non-cystic fibrosis bronchiectasis in adults and its correlation to disease severity. Ann Am Thorac Soc 12:1764–1770. https://doi.org/10.1513/AnnalsATS.201507-472OC
Jayaram L, Vandal AC, Chang CL, Lewis C, Tong C, Tuffery C, Bell J, Fergusson W, Jeon G, Milne D, Jones S, Karalus N, Hotu S, Wong C (2022) Tiotropium treatment for bronchiectasis: a randomised, placebo-controlled, crossover trial. Eur Respir J 59(6):2102184. https://doi.org/10.1183/13993003.02184-2021
Martinez-Garcia MA, Soler-Cataluna JJ, Perpina-Tordera M et al (2007) Factors associated with lung function decline in adult patients with stable non-cystic fibrosis bronchiectasis. Chest 132:1565–1572. https://doi.org/10.1378/chest.07-0490
Ma Y, Niu Y, Tian G, Wei J, Gao Z (2015) Pulmonary function abnormalities in adult patients with acute exacerbation of bronchiectasis: a retrospective risk factor analysis. Chron Respir Dis 12(3):222–229. https://doi.org/10.1177/1479972315583042
Wang H, Ji XB, Mao B et al (2018) Pseudomonas aeruginosa isolation in patients with non-cystic fibrosis bronchiectasis: a retrospective study. BMJ Open 8(3):e014613. https://doi.org/10.1136/bmjopen-2016-014613
Acknowledgements
None
Funding
None.
Author information
Authors and Affiliations
Contributions
Z-LS and Z-MZ designed the study and Z-LS and H-YZ performed data analyses and wrote the report. H-BP performed investigation and data collection. All authors contributed to the review and revision of the manuscript and have read and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the author(s).
Ethical Approval
This prospective observational study was approved by the Ethics Committee in the Beijing Rehabilitation Hospital, Capital Medical University (No. fd2015-001).
Consent to Participate
All participants signed informed consent.
Consent to Publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shi, ZL., Zhang, HY., Peng, HB. et al. Tiotropium in Patients with Bronchiectasis: A Prospective Cohort Study. Lung 201, 9–15 (2023). https://doi.org/10.1007/s00408-023-00597-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-023-00597-8