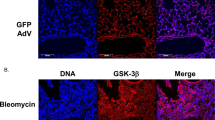
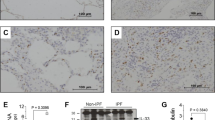
Abstract
Purpose
Fibroproliferation and excess deposition of extracellular matrix (ECM) are the pathologic hallmarks of idiopathic pulmonary fibrosis (IPF), a chronic progressive disorder with high mortality and suboptimal treatment options. Although the etiologic mechanisms responsible for the development and progression of IPF remain unclear, cell-ECM interactions and growth factors are considered important. Cilengitide is a cyclic RGD pentapeptide with anti-angiogenic activity that targets αvβ3, αvβ5 and α5β1, integrins known to mediate cell-ECM interactions and activate the pro-fibrotic growth factor Transforming Growth Factor beta (TGF-β).
Methods
Cilengitide was studied in vitro with the use of NIH/3T3 cells and primary lung fibroblasts, and in vivo in the well-characterized bleomycin-induced lung injury model. The extent of ECM deposition was determined by RT-PCR, Western blot, histologic analysis and hydroxyproline assay of lung tissue. Bronchoalveolar lavage analysis was used to determine cell counts.
Results
Cilengitide treatment of cultured fibroblasts showed decreased adhesion to vitronectin and fibronectin, both integrin-dependent events. Cilengitide also inhibited TGF-β-induced fibronectin gene expression and reduced the accumulation of mRNAs and protein for fibronectin and collagen type I. Both preventive and treatment effects of daily injections of cilengitide (20 mg/kg) failed to inhibit the development of pulmonary fibrosis as determined by histological analysis (Ashcroft scoring), bronchoalveolar lavage (BAL) fluid cell counts, and hydroxyproline content.
Conclusions
Overall, our data suggest that, despite its in vitro activity in fibroblasts, daily injections of cilengitide (20 mg/kg) did not inhibit the development of or ameliorate bleomycin-induced pulmonary fibrosis in mice.




Similar content being viewed by others

References
Thannickal VJ, Henke CA, Horowitz JC, Noble PW, Roman J, Sime PJ, Zhou Y, Wells RG, White ES, Tschumperlin DJ (2014) Matrix biology of idiopathic pulmonary fibrosis: a workshop report of the national heart, lung, and blood institute. Am J Pathol 184(6):1643–1651. https://doi.org/10.1016/j.ajpath.2014.02.003
Watson WH, Ritzenthaler JD, Roman J (2016) Lung extracellular matrix and redox regulation. Redox Biol 8:305–315. https://doi.org/10.1016/j.redox.2016.02.005
Jenkins RG, Su X, Su G, Scotton CJ, Camerer E, Laurent GJ, Davis GE, Chambers RC, Matthay MA, Sheppard D (2006) Ligation of protease-activated receptor 1 enhances alpha(v)beta6 integrin-dependent TGF-beta activation and promotes acute lung injury. J Clin Invest 116(6). https://doi.org/10.1172/JCI27183.1606
Scotton CJ, Krupiczojc MA, Königshoff M, Mercer PF, Lee YC, Kaminski N, Morser J, Post JM, Maher TM, Nicholson AG, Moffatt JD, Laurent GJ, Derian CK, Eickelberg O, Chambers R (2009) Increased local expression of coagulation factor X contributes to the fibrotic response in human and murine lung injury. J Clin Invest 119(9):2550–2563
Henderson NC, Arnold TD, Katamura Y, Giacomini MM, Rodriguez JD, McCarty JH et al (2013) Targeting of αv integrin identifies a core molecular pathway that regulates fibrosis in several organs. Nat Med 19(12):1617–1624. https://doi.org/10.1038/nm.3282
Munger JS, Huang X, Kawakatsu H, Griffiths MJ, Dalton SL, Wu J et al (1999) The integrin αvβ6 binds and activates latent TGFβ1: a mechanism for regulating pulmonary inflammation and fibrosis. Cell 96(3):319–328. https://doi.org/10.1016/S0092-8674(00)80545-0
Sarrazy V, Koehler A, Chow ML, Zimina E, Li CX, Kato H, Caldarone CA, Hinz B (2014) Integrins αvβ5 and αvβ3 promote latent TGF-β1 activation by human cardiac fibroblast contraction. Cardiovasc Res 102(3):407–417. https://doi.org/10.1093/cvr/cvu053
Asano Y, Ihn H, Yamane K, Jinnin M, Tamaki K (2006) Increased expression of integrin αvβ5 induces the myofibroblastic differentiation of dermal fibroblasts. Am J Pathol 168(2):499–510. https://doi.org/10.2353/ajpath.2006.041306
Asano Y, Ihn H, Yamane K, Jinnin M, Mimura Y, Tamaki K (2005) Increased expression of integrin α v β 3 contributes to the establishment of autocrine TGF-β signaling in scleroderma fibroblasts. J Immunol 175(11):7708–7718. https://doi.org/10.4049/jimmunol.175.11.7708
Mas-Moruno C, Rechenmacher F, Kessler H (2010) Cilengitide: the first anti-angiogenic small molecule drug candidate. Design, synthesis and clinical evaluation. Anti Cancer Agents Med Chem 10(10):753–768. https://doi.org/10.2174/187152010794728639
Nabors LB, Mikkelsen T, Rosenfeld SS, Hochberg F, Akella NS, Fisher JD et al (2007) Phase I and correlative biology study of cilengitide in patients with recurrent malignant glioma. J Clin Oncol 25(13):1651–1657. https://doi.org/10.1200/JCO.2006.06.6514
Li C, Flynn RS, Grider JR, Murthy KS, Kellum JM, Akbari H, Kuemmerle JF (2013) Increased activation of latent TGF-β1 by αVβ3 in human Crohn’s disease and fibrosis in TNBS colitis can be prevented by cilengitide. Inflamm Bowel Dis 19(13):2829–2839. https://doi.org/10.1097/MIB.0b013e3182a8452e16
Patsenker E, Popov Y, Stickel F, Schneider V, Ledermann M, Sägesser H, Niedobitek G, Goodman SL, Schuppan D (2009) Pharmacological inhibition of integrin alphavbeta3 aggravates experimental liver fibrosis and suppresses hepatic angiogenesis. Hepatology 50(5):1501–1511. https://doi.org/10.1002/hep.23144
Michaelson JE, Ritzenthaler JD, Roman J (2002) Regulation of serum-induced fibronectin expression by protein kinases, cytoskeletal integrity, and CREB. Am J Physiol Lung Cell Mol Physiol 282:291–301. https://doi.org/10.1152/ajplung.00445.2000
Sueblinvong V, Neujahr DC, Mills ST, Roser-Page S, Ritzenthaler JD, Guidot D, Rojas M, Roman J (2012) Predisposition for disrepair in the aged lung. Am J Med Sci 344(1):41–51. https://doi.org/10.1097/MAJ.0b013e318234c132
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Ashcroft T, Simpson JM, Timbrell V (1988) Simple method of estimating severity of pulmonary fibrosis on a numerical scale. J Clin Pathol 41(4):467–470. https://doi.org/10.1136/jcp.41.4.467
Pohl WR, Conlan MG, Thompson AB, Ertl RF, Romberger DJ, Mosher DF, Rennard SI (1991) Vitronectin in bronchoalveolar lavage fluid is increased in patients with interstitial lung disease. Am Rev Respir Dis 143(6):1369–1375. https://doi.org/10.1164/ajrccm/143.6.1369
Roberts MS, Woods AJ, Shaw PE, Norman JC (2003) ERK1 associates with αvβ3 integrin and regulates cell spreading on vitronectin. J Biol Chem 278(3):1975–1985. https://doi.org/10.1074/jbc.M208607200
Hermann P, Armant M, Brown E, Rubio M, Ishihara H, Ulrich D, Caspary RG et al (1999) The vitronectin receptor and its associated CD47 molecule mediates proinflammatory cytokine synthesis in human monocytes by interaction with soluble CD23. J Cell Biol 144(4):767–775. https://doi.org/10.1083/jcb.144.4.767
Ignotzs RA, Massague J (1986) Transforming growth factor stimulates the expression of fibronectin and collagen and their incorporation into the extracellular atrix. J Biol Chem 261(9):4337–4345
Hocevar BA, Brown TL, Howe PH (1999) TGF-β induces fibronectin synthesis through a c-Jun N-terminal kinase-dependent, Smad4-independent pathway. EMBO J 18(5):1345–1356
Kim S, Bakre M, Yin H, Varner JA (2002) Inhibition of endothelial cell survival and angiogenesis by protein kinase A. J Clin Invest 110(7):933–941. https://doi.org/10.1172/JCI0214268
Oku H, Shimizu T, Kawabata T, Nagira M, Hikita I, Ueyama A, Matsushima S, Torii M, Arimura A (2008) Antifibrotic action of pirfenidone and prednisolone: different effects on pulmonary cytokines and growth factors in bleomycin-induced murine pulmonary fibrosis. Eur J Pharmacol 590:400–408. https://doi.org/10.1016/j.ejphar.2008.06.046
Wollin L, Maillet I, Quesniaux V, Holweg A, Ryffel B (2014) Antifibrotic and anti-inflammatory activity of the tyrosine kinase inhibitor nintedanib in experimental models of lung fibrosiss. J Pharmacol Exp Ther 349(2):209–220. https://doi.org/10.1124/jpet.113.208223
Eskens FALM, Dumez H, Hoekstra R, Perschl A, Brindley C, Böttcher S, Wynendaele W, Drevs J, Verweij J, van Oosterom AT (2003) Phase I and pharmacokinetic study of continuous twice weekly intravenous administration of Cilengitide (EMD 121974), a novel inhibitor of the integrins αvβ3 and αvβ5 in patients with advanced solid tumours. Eur J Cancer 39(7):917–926. https://doi.org/10.1016/S0959-8049(03)00057-1
Stupp R, Hegi ME, Gorlia T, Erridge SC, Perry J, Hong YK et al (2014) Cilengitide combined with standard treatment for patients with newly diagnosed glioblastoma with methylated MGMT promoter (CENTRIC EORTC 26071-22072 study): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol 15(10):1100–1108. https://doi.org/10.1016/S1470-2045(14)70379-1
Kim YH, Lee JK, Kim B, DeWitt JP, Lee JE, Han JH, Kim SK, Oh CW, Kim CY (2013) Combination therapy of cilengitide with belotecan against experimental glioblastoma. Int J Cancer 133(3):749–756. https://doi.org/10.1002/ijc.28058 Epub 2013 Feb 27
Bon H, Hales P, Lumb S, Holdsworth G, Johnson T, Qureshi O, Twomey BM (2019) Spontaneous extracellular matrix accumulation in a human in vitro model of renal fibrosis is mediated by αV integrins. Nephron 142(4):328–350. https://doi.org/10.1159/000499506
Tucci M, Stucci S, Silvestris F (2014) Does cilengitide deserve another chance? Lancet Oncol 15(13):e584–e585. https://doi.org/10.1016/S1470-2045(14)70462-0
Luzina IG, Todd NW, Nacu N, Lockatell V, Choi J, Hummers LK, Atamas SP (2009) Regulation of pulmonary inflammation and fibrosis through expression of integrins αVβ3 and αVβ5 on pulmonary T lymphocytes. Arthritis Rheum 60(5):1530–1539. https://doi.org/10.1002/art.24435
Funding
This work was supported by a Grant from the Department of Veterans Affairs grant (BX000216) to JR.
Author information
Authors and Affiliations
Contributions
Concept and design: MZ, ETG, JDR, JR; Analysis and interpretation: MZ, ETG, JDR, JR; Drafting manuscript and important intellectual input: MZ, JDR, JR.
Corresponding author
Ethics declarations
Conflict of interest
Jesse Roman receives support for research related to clinical trials from industry (e.g., Genentech, Boehringer Ingelheim, Galapagos). He also serves as consultant on DSMB and steering committees for Genentech, Novartis and Boehringer Ingelheim. All other authors, no potential conflict of interest was reported.
Ethical Approval
All animal procedures were approved by the University of Louisville’s Institutional Animal Care and Use Committee.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ritzenthaler, J.D., Zhang, M., Torres-Gonzalez, E. et al. The Integrin Inhibitor Cilengitide and Bleomycin-Induced Pulmonary Fibrosis. Lung 198, 947–955 (2020). https://doi.org/10.1007/s00408-020-00400-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-020-00400-y