Abstract
Purpose
Despite safety concerns, β2-sympathomimetics are still widely used as tocolytic agents. β-Blockers in turn are used to treat vasculo-proliferative diseases of the newborn such as retinopathy of prematurity (ROP), which may lead to visual impairment and blindness. The scope of this study was to investigate whether antenatal exposure to the β2-sympathomimetic fenoterol contributes to the development of ROP.
Methods
For this single-center retrospective case–control study of prospectively collected clinical data, all infants born before 32 weeks of gestation between 2001 and 2012 were included. The association of prenatal exposure to fenoterol and the development of ROP were analyzed by multivariate logistic regression.
Results
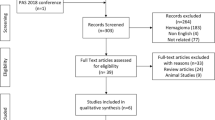
n = 1134 infants < 32 weeks of gestation were screened for eligibility, out of which n = 722 met the inclusion criteria. Exposure to fenoterol (n = 505) was not associated with a higher rate of ROP (OR 0.721, 95% CI 0.463–1.122). Further, duration of exposure (days) did not alter the incidence of ROP (OR 1.001, 95% CI 0.986–1.016). Frequency distribution of different ROP stages and the need for therapeutic intervention was also not affected by prenatal exposure to fenoterol. Risk factors for the development of ROP like low birth weight, low gestational age, prolonged respiratory support and multiple gestation were confirmed in our large study cohort.
Conclusion
β2-Sympathomimetic tocolysis does not increase the rate of ROP in premature infants born < 32 weeks of gestation. Our results render fenoterol a safe tocolytic agent regarding neonatal ROP development.

Similar content being viewed by others
References
Murray CJL, Vos T, Lozano R et al (2012) Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: a systematic analysis for the global burden of disease study 2010. Lancet 380:2197–2223. https://doi.org/10.1016/S0140-6736(12)61689-4
Haas DM, Caldwell DM, Kirkpatrick P et al (2012) Tocolytic therapy for preterm delivery: systematic review and network meta-analysis. BMJ 345:e6226. https://doi.org/10.1136/bmj.e6226
Haas DM, Imperiale TF, Kirkpatrick PR et al (2009) Tocolytic therapy: a meta-analysis and decision analysis. Obstet Gynecol 113:585–594. https://doi.org/10.1097/AOG.0b013e318199924a
de Heus R, Mol BW, Erwich J-JHM et al (2009) Adverse drug reactions to tocolytic treatment for preterm labour: prospective cohort study. BMJ 338:b744–b744. https://doi.org/10.1136/bmj.b744
Neilson JP, West HM, Dowswell T (2014) Betamimetics for inhibiting preterm labour. Cochrane Database Syst Rev 6: CD004352. https://doi.org/10.1002/14651858.CD004352.pub3
Tolsma KW, Allred EN, Chen ML et al (2011) Neonatal bacteremia and retinopathy of prematurity: the ELGAN study. Arch Ophthalmol 129:1555–1563. https://doi.org/10.1001/archophthalmol.2011.319
Kuon RJ, Hudalla H, Seitz C et al (2015) Impaired neonatal outcome after emergency cerclage adds controversy to prolongation of pregnancy. PLoS ONE 10:e0129104. https://doi.org/10.1371/journal.pone.0129104
Dammann O, Brinkhaus M-J, Bartels DB et al (2009) Immaturity, perinatal inflammation, and retinopathy of prematurity: a multi-hit hypothesis. Early Hum Dev 85:325–329. https://doi.org/10.1016/j.earlhumdev.2008.12.010
Hartnett ME, Penn JS (2012) Mechanisms and management of retinopathy of prematurity. N Engl J Med 367:2515–2526. https://doi.org/10.1056/NEJMra1208129
Aiello LP, Pierce EA, Foley ED et al (1995) Suppression of retinal neovascularization in vivo by inhibition of vascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc Natl Acad Sci 92:10457–10461. https://doi.org/10.1073/pnas.92.23.10457
International committee for the classification of retinopathy of prematurity (2005) The international classification of retinopathy of prematurity revisited. Arch Ophthalmol 123:991–999. https://doi.org/10.1001/archopht.123.7.991
Lundgren P, Kistner A, Andersson EM et al (2014) Low birth weight is a risk factor for severe retinopathy of prematurity depending on gestational age. PLoS ONE 9:e109460. https://doi.org/10.1371/journal.pone.0109460
Razak A, Faden M (2019) Association of small for gestational age with retinopathy of prematurity: a systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed fetal neonatal-2019-316976. https://doi.org/10.1136/archdischild-2019-316976
York JR, Landers S, Kirby RS et al (2004) Arterial oxygen fluctuation and retinopathy of prematurity in very-low-birth-weight infants. J Perinatol 24:82–87. https://doi.org/10.1038/sj.jp.7211040
Askie LM, Darlow BA, Finer N et al (2018) Association between oxygen saturation targeting and death or disability in extremely preterm infants in the neonatal oxygenation prospective meta-analysis collaboration. JAMA 319:2190–2201. https://doi.org/10.1001/jama.2018.5725
Slidsborg C, Jensen A, Forman JL et al (2016) Neonatal risk factors for treatment-demanding retinopathy of prematurity: a danish national study. Ophthalmology 123:796–803. https://doi.org/10.1016/j.ophtha.2015.12.019
Natarajan G, Shankaran S, Nolen TL et al (2019) Neurodevelopmental outcomes of preterm infants with retinopathy of prematurity by treatment. Pediatrics 144:e20183537. https://doi.org/10.1542/peds.2018-3537
Solebo AL, Teoh L, Rahi J (2017) Epidemiology of blindness in children. Arch Dis Child 102:853–857. https://doi.org/10.1136/archdischild-2016-310532
Sankar MJ, Sankar J, Chandra P (2018) Anti-vascular endothelial growth factor (VEGF) drugs for treatment of retinopathy of prematurity. Cochrane Database Syst Rev 1:CD009734. https://doi.org/10.1002/14651858.CD009734.pub3
Kaempfen S, Neumann RP, Jost K, Schulzke SM (2018) Beta-blockers for prevention and treatment of retinopathy of prematurity in preterm infants. Cochrane Database Syst Rev 3: CD011893. https://doi.org/10.1002/14651858.CD011893.pub2
Zhang L, Mai H-M, Zheng J et al (2014) Propranolol inhibits angiogenesis via down-regulating the expression of vascular endothelial growth factor in hemangioma derived stem cell. Int J Clin Exp Pathol 7:48–55
Novoa M, Baselga E, Beltran S, et al (2018) Interventions for infantile haemangiomas of the skin. Cochrane Database Syst Rev 4: CD006545. https://doi.org/10.1002/14651858.CD006545.pub3
Léauté-Labrèze C, Harper JI, Hoeger PH (2017) Infantile haemangioma. Lancet 390:85–94. https://doi.org/10.1016/S0140-6736(16)00645-0
Garg J, Feng Y-X, Jansen SR et al (2017) Catecholamines facilitate VEGF-dependent angiogenesis via β2-adrenoceptor-induced Epac1 and PKA activation. Oncotarget 8:44732–44748. https://doi.org/10.18632/oncotarget.17267
Mayer M, Minichmayr A, Klement F et al (2013) Tocolysis with the β-2-sympathomimetic hexoprenaline increases occurrence of infantile haemangioma in preterm infants. Arch Dis Child Fetal Neonatal Ed 98:F108–F111. https://doi.org/10.1136/archdischild-2011-301030
Hudalla H, Karmen C, Bruckner T et al (2018) Tocolysis with the β2-sympathomimetic fenoterol does not increase the occurrence of infantile hemangioma in preterm and term infants. Arch Gynecol Obstet 10:S2–7. https://doi.org/10.1007/s00404-018-4830-5
Boghossian NS, Saha S, Bell EF et al (2019) Birth weight discordance in very low birth weight twins: mortality, morbidity, and neurodevelopment. J Perinatol 206:10. https://doi.org/10.1038/s41372-019-0427-5
Gur Z, Tsumi E, Wainstock T et al (2018) Association between delivery of small-for-gestational age neonate and long-term pediatric ophthalmic morbidity. Arch Gynecol Obstet 298:1095–1099. https://doi.org/10.1007/s00404-018-4901-7
Ricard CA, Dammann CEL, Dammann O (2017) Screening tool for early postnatal prediction of retinopathy of prematurity in preterm newborns (STEP-ROP). Neonatology 112:130–136. https://doi.org/10.1159/000464459
Cayabyab R, Ramanathan R (2016) Retinopathy of prematurity: therapeutic strategies based on pathophysiology. Neonatology 109:369–376. https://doi.org/10.1159/000444901
Bizzarro MJ, Hussain N, Jonsson B et al (2006) Genetic susceptibility to retinopathy of prematurity. Pediatrics 118:1858–1863. https://doi.org/10.1542/peds.2006-1088
Husain SM, Sinha AK, Bunce C et al (2013) Relationships between maternal ethnicity, gestational age, birth weight, weight gain, and severe retinopathy of prematurity. J Pediatr 163:67–72. https://doi.org/10.1016/j.jpeds.2012.12.038
Mohamed S, Schaa K, Cooper ME et al (2009) Genetic contributions to the development of retinopathy of prematurity. Pediatr Res 65:193–197. https://doi.org/10.1203/PDR.0b013e31818d1dbd
Movsas TZ, Spitzer AR, Gewolb IH (2015) Trisomy 21 and risk of retinopathy of prematurity. Pediatrics 136:e441–e447. https://doi.org/10.1542/peds.2015-0623
Palmer EA, Flynn JT, Hardy RJ et al (1991) Incidence and early course of retinopathy of prematurity. The cryotherapy for retinopathy of prematurity cooperative group. Ophthalmology 98:1628–1640. https://doi.org/10.1016/s0161-6420(91)32074-8
Léauté-Labrèze C, Dumas de la Roque E, Hubiche T et al (2008) Propranolol for severe hemangiomas of infancy. N Engl J Med 358:2649–2651. https://doi.org/10.1056/NEJMc0708819
Bührer C, Bassler D (2015) Oral propranolol: a new treatment for infants with retinopathy of prematurity? Neonatology 108:49–52. https://doi.org/10.1159/000381659
Praveen V, Vidavalur R, Rosenkrantz TS, Hussain N (2009) Infantile hemangiomas and retinopathy of prematurity: possible association. Pediatrics 123:e484–e489. https://doi.org/10.1542/peds.2007-0803
Funding
No external or internal funding was received for this project.
Author information
Authors and Affiliations
Contributions
HH: protocol/project development, data collection or management, data analysis, and manuscript writing/editing. TB: data collection or management and data analysis. JP: manuscript editing. TS: manuscript editing. RJK: protocol/project development, data collection or management, data analysis, and manuscript writing/editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest.
Ethical approval
The study was approved by the Heidelberg University institutional Review Board (IRB, S-094/2013). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
The institutional ethics committee waived informed consent due to the retrospective nature of the study, and pseudonymization of data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hudalla, H., Bruckner, T., Pöschl, J. et al. Antenatal exposure to fenoterol is not associated with the development of retinopathy of prematurity in infants born before 32 weeks of gestation. Arch Gynecol Obstet 301, 687–692 (2020). https://doi.org/10.1007/s00404-020-05463-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-020-05463-z