Abstract
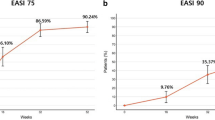
Dupilumab is the first human monoclonal antibody that treats atopic dermatitis (AD) by blocking interleukin 4 (IL-4) and interleukin 13 (IL-13), which can suppress the Th2 inflammatory reaction. Effective treatments for pediatric AD patients are limited; therefore, we aimed to assess the efficacy and safety of dupilumab in pediatric AD patients. Fifteen pediatric patients diagnosed with moderate to severe AD and treated with dupilumab were enrolled in this study. SPSS was used to analyze data and obtain the average values of Eczema Area and Severity Index (EASI), SCORing AD (SCORAD), and Children’s Dermatology Life Quality Index (CDLQI). GRAPHPAD was used to analyze and plot the statistics. The average EASI values were 19.23 ± 3.03 and 1.69 ± 0.54 at baseline and at following up for 6 months after standardized treatment protocol, respectively. The average SCORAD values were 43.27 ± 4.63 and 6.13 ± 1.41 at baseline and at following up for 6 months after standardized treatment protocol, respectively. The average CDLQI value at baseline was 13.53 ± 2.88 and following up for 6 months after standardized treatment protocol was 1.60 ± 0.63. The most frequent adverse event was conjunctivitis. No serious adverse events occurred during the treatment period. Dupilumab could reduce symptoms and improve pruritus in pediatric AD patients, and the frequent adverse events were reversible. It has a definite therapeutic effect on AD; nevertheless, further studies should be conducted to obtain information on its the long-term efficacy and safety.



Similar content being viewed by others
Data availability
The data that support the findings of this study are openly available in Figshare at http://doi.org/10.6084/m9.figshare.17040206.
References
DaVeiga SP (2012) Epidemiology of atopic dermatitis: a review. Allergy Asthma Proc [Internet] 33(3):227–234
Mallol J, Crane J, von Mutius E, Odhiambo J, Keil U, Stewart A (2013) The International study of asthma and allergies in childhood (ISAAC) phase three: a global synthesis. Allergol Immunopathol 41(2):73–85
Atopic Dermatitis Working Group IGCS of D, Yao Xu, Song Z-Q, Li W, Liang Y-S, Zhao Y et al (2021) Guidelines for diagnosis and treatment of atopic dermatitis in china (2020) #. Int J Dermatol Venerol 4(1):1–9
Urban K, Chu S, Giesey RL, Mehrmal S, Uppal P, Nedley N et al (2021) The global, regional, and national burden of atopic dermatitis in 195 countries and territories: an ecological study from the global burden of disease study 2017. JAAD Int 2:12–18
Global Burden of Disease Collaborative Network. Global Burden of Disease Study 2016 (GBD 2016) results; 2017. http://ghdx.healthdata. Accessed 1 May 2020
Tsai TF, Rajagopalan M, Chu CY, Encarnacion L, Gerber RA, Santos Estrella P et al (2019) Burden of atopic dermatitis in Asia. J Dermatol 46(10):825–834
Napolitano M, Di Guida A, Nocerino M, Fabbrocini G, Patruno C (2021) The emerging role of dupilumab in dermatological indications. Expert Opin Biol Ther [Journal Article] 21(11):1461–1471
Dupilumab [Prescribing Information] (2020) Regeneron Pharmaceuticals, Tarrytown. https://www.regeneron.com/sites/default/files/Dupixent_FPI.pdf. Accessed 16 June 2021.
https://www.nmpa.gov.cn/zwfw/sdxx/sdxxyp/yppjfb/20210909162932132.html. Accessed 1 Dec 2021
https://www.cde.org.cn/main/xxgk/listpage/2f78f372d351c6851af7431c7710a731. Accessed 1 Dec 2021
Paller AS, Siegfried EC, Simpson EL, Cork MJ, Lockshin B, Kosloski MP et al (2021) A phase 2, open-label study of single-dose dupilumab in children aged 6 months to <6 years with severe uncontrolled atopic dermatitis: pharmacokinetics, safety and efficacy. J Eur Acad Dermatol 35(2):464–475
Paller AS, Siegfried EC, Thaçi D, Wollenberg A, Cork MJ, Arkwright PD et al (2020) Efficacy and safety of dupilumab with concomitant topical corticosteroids in children 6–11 years old with severe atopic dermatitis: a randomized, double-blinded, placebo-controlled phase 3 trial. J AM Acad Dermatol 83(5):1282–1293
Lewis-Jones MS, Finlay AY (1995) The children’s dermatology life quality index (CDLQI): initial validation and practical use. Br J Dermatol 132(6):942–949
Simpson EL, de Bruin-Weller M, Bansal A, Chen Z, Nelson L, Whalley D et al (2021) Definition of clinically meaningful within-patient changes in POEM and CDLQI in children 6 to 11 years of age with severe atopic dermatitis. Dermatol Ther 11(4):1415–1422
Napolitano M, Ferrillo M, Patruno C, Scalvenzi M, Andrea MD, Fabbrocini G (2021) Efficacy and safety of dupilumab in clinical practice: one year of experience on 165 adult patients from a tertiary referral centre. Dermatol Ther 11(2):355–361
Beck LA, Thaçi D, Hamilton JD, Graham NM, Bieber T, Rocklin R et al (2014) Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. New Engl J Med 371(2):130–139
Armario-Hita JC, Pereyra-Rodriguez J, Silvestre JF, Ruiz-Villaverde R, Valero A, Izu-Belloso R et al (2019) Treatment of moderate-to-severe atopic dermatitiswith dupilumab in real clinical practice: a multicentre, retrospective case series. Br J Dermatol 181(5):1072–1074
Dakin H, Haas C (2020) Successful and rapid clearance of severe, treatment-resistant atopic dermatitis with dupilumab in a3-year-old. Dermatol Ther. https://doi.org/10.1111/dth.14282
Varma BA, Tassavor M, Levitt J (2019) The utility of dupilumab for use in the pediatric population. JAAD Case Rep 5(11):943–944
Acknowledgements
All the authors had access to relevant data and participated in the drafting, review, and approval of this publication. Thanks to the professor Song-Mei Geng, Ying-Ying Dong, Xiao-Peng Wang, Jin-Gang An, Zheng-Xiao Li, and Ding-Wei Zhang for providing information about the patients that they treated. Thanks for the guidance of professor Xiao-Li Li.
Funding
This work was supported by the National Natural Science Foundation of China (no. 30901297); the Doctoral Fund of Youth Scholars of Ministry of Education of China (no. 20090201120074); and Shaanxi Province She Fa (2020SF-177).
Author information
Authors and Affiliations
Contributions
The concept was developed by X-LL. All the authors carried out the search, data extraction, and software using. The disagreement was solved by X-LL. The funding acquisition was finished by X-LL. Statistical analysis and inference was done by DYY and LL. The manuscript was written by DYY, LL and TL. All the authors agreed to the final version for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Ethical approval
This study was conducted in accordance with provisions of the Declaration of Helsinki and International Conference on Harmonization guidelines for Good Clinical Practice. This study protocol was reviewed and approved by the Chinese Ethics Committee of Registering Clinical Trials (the approval number is ChiECRCT2021219).
Informed consent
Written informed consents to participate in the study were obtained from the participants or their parents/legal guardians/next of kin.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, DY., Li, L., Lu, T. et al. Efficacy and safety of dupilumab in pediatric patients with moderate to severe atopic dermatitis: a real-world study. Arch Dermatol Res 315, 467–472 (2023). https://doi.org/10.1007/s00403-022-02380-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-022-02380-w