Abstract
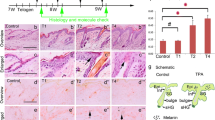
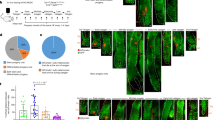
Melanocyte stem cells (McSCs) are localized in the bulge region of hair follicles and supply melanocytes, which determine hair color by synthesizing melanin. Ectopic differentiation of McSCs, which are usually undifferentiated in the bulge region, causes depletion of McSCs and results in hair graying. Therefore, to prevent hair graying, it is essential to maintain McSCs in the bulge region, but the mechanism of McSC maintenance remains unclear. To address this issue, we investigated the role of CXCL12, a chemokine which was previously suggested to induce migration of melanocyte lineage cells, as a niche component of McSCs. Immunohistological analysis revealed that CXCL12 was highly expressed in the bulge region of human hair follicles. CXCL12 mRNA expression level was significantly lower in white hairs plucked from human scalps than in black hairs. CXCL12 attracted the migration of early-passage normal human epidermal melanocytes (eNHEMs), an in vitro model of McSCs, which had characteristics of immature melanocyte precursors. We also found that CXCL12 suppressed their differentiation. These results suggest that CXCL12 regulates differentiation of McSCs as well as their proper localization, and maintaining McSCs by regulating CXCL12 expression level in the bulge region may be a key to preventing hair graying.




Similar content being viewed by others
References
Avniel S, Arik Z, Maly A, Sagie A, Basst HB, Yahana MD, Weiss ID, Pal B, Wald O, Ad-El D, Fujii N, Arenzana-Seisdedos F, Jung S, Galun E, Gur E, Peled A (2006) Involvement of the CXCL12/CXCR4 pathway in the recovery of skin following burns. J Invest Dermatol 126:468–476
Belmadani A, Jung H, Ren D, Miller RJ (2009) The chemokine SDF-1/CXCL12 regulates the migration of melanocyte progenitors in mouse hair follicles. Differentiation 77:395–411
Brizzi MF, Tarone G, Defilippi P (2012) Extracellular matrix, integrins, and growth factors as tailors of the stem cell niche. Curr Opin Cell Biol 24:645–651
Choi HR, Byun SY, Kwon SH, Park KC (2015) Niche interactions in epidermal stem cells. World J Stem Cells 7:495–501
Choi YJ, Yoon TJ, Lee YH (2008) Changing expression of the genes related to human hair graying. Eur J Dermatol 18:397–399
Gattazzo F, Urciuolo A, Bonaldo P (2014) Extracellular matrix: a dynamic microenvironment for stem cell niche. Biochim Biophys Acta 1840:2506–2519
Ghosh MC, Collins GD, Vandanmagsar B, Patel K, Brill M, Carter A, Lustig A, Becker KG, Wood WW III, Emeche CD, French AD, O’Connell MP, Xu M, Weeraratna AT, Taub DD (2009) Activation of Wnt5A signaling is required for CXC chemokine ligand 12-mediated T-cell migration. Blood 114:1366–1373
Hachiya A, Sriwiriyanont P, Kobayashi T, Nagasawa A, Yoshida H, Ohuchi A, Kitahara T, Visscher MO, Takema Y, Tsuboi R, Boissy RE (2009) Stem cell factor-KIT signalling plays a pivotal role in regulating pigmentation in mammalian hair. J Pathol 218:30–39
Inomata K, Aoto T, Binh NT, Okamoto N, Tanimura S, Wakayama T, Iseki S, Hara E, Masunaga T, Shimizu H, Nishimura EK (2009) Genotoxic stress abrogates renewal of melanocyte stem cells by triggering their differentiation. Cell 137:1088–1099
Keogh EV, Walsh RJ (1965) Rate of greying of human hair. Nature 207:877–878
Lee E, Han J, Kim K, Choi H, Cho EG, Lee TR (2013) CXCR7 mediates SDF1-induced melanocyte migration. Pigment Cell Melanoma Res 26:58–66
Millar SE (2002) Molecular mechanisms regulating hair follicle development. J Invest Dermatol 118:216–225
Nishimura EK, Jordan SA, Oshima H, Yoshida H, Osawa M, Moriyama M, Jackson IJ, Barrandon Y, Miyachi Y, Nishikawa S (2002) Dominant role of the niche in melanocyte stem-cell fate determination. Nature 416:854–860
Nishimura EK, Suzuki M, Igras V, Du J, Lonning S, Miyachi Y, Roes J, Beermann F, Fisher DE (2010) Key roles for transforming growth factor beta in melanocyte stem cell maintenance. Cell Stem Cell 6:130–140
Pajonk F, Vlashi E (2013) Characterization of the stem cell niche and its importance in radiobiological response. Semin Radiat Oncol 23:237–241
Panhard S, Lozano I, Loussouarn G (2012) Greying of the human hair: a worldwide survey, revisiting the ‘50’ rule of thumb. Br J Dermatol 167:865–873
Sato A, Yamamoto H, Sakane H, Koyama H, Kikuchi A (2010) Wnt5a regulates distinct signalling pathways by binding to Frizzled2. EMBO J 29:41–54
Sugiyama T, Kohara H, Noda M, Nagasawa T (2006) Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity 25:977–988
Tanimura S, Tadokoro Y, Inomata K, Binh NT, Nishie W, Yamazaki S, Nakauchi H, Tanaka Y, McMillan JR, Sawamura D, Yancey K, Shimizu H, Nishimura EK (2011) Hair follicle stem cells provide a functional niche for melanocyte stem cells. Cell Stem Cell 8:177–187
Yamada T, Hasegawa S, Inoue Y, Date Y, Arima M, Yagami A, Iwata Y, Takahashi M, Yamamoto N, Mizutani H, Nakata S, Matsunaga K, Akamatsu H (2014) Accelerated differentiation of melanocyte stem cells contributes to the formation of hyperpigmented maculae. Exp Dermatol 23:652–658
Yamada T, Hasegawa S, Inoue Y, Date Y, Yamamoto N, Mizutani H, Nakata S, Matsunaga K, Akamatsu H (2013) Wnt/β-catenin and kit signaling sequentially regulate melanocyte stem cell differentiation in UVB-induced epidermal pigmentation. J Invest Dermatol 133:2753–2762
Yamauchi A, Hadjur C, Takahashi T, Suzuki I, Hirose K, Mahe YF (2013) Human skin melanocyte migration towards stromal cell-derived factor-1α demonstrated by optical real-time cell mobility assay: modulation of their chemotactic ability by α-melanocyte-stimulating hormone. Exp Dermatol 22:664–667
Zhang H, Zhu NX, Huang K, Cai BZ, Zeng Y, Xu YM, Liu Y, Yuan YP, Lin CM (2016) iTRAQ-based quantitative proteomic comparison of early- and late-passage human dermal papilla cell secretome in relation to inducing hair follicle regeneration. PLoS One 11:e0167474
Acknowledgements
We are grateful to N. Goshima (Cellisis Co., Ltd., Aichi, Japan) for her support in preparing the manuscript.
Author information
Authors and Affiliations
Contributions
TY and SH designed the research study. TY, MA, YI, TK, SN and NY performed the research. TY, SH, SN, KS and HA carried out the data analysis and wrote the manuscript. All authors critically revised the manuscript and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors state no conflict of interest.
Rights and permissions
About this article
Cite this article
Yamada, T., Hasegawa, S., Hasebe, Y. et al. CXCL12 regulates differentiation of human immature melanocyte precursors as well as their migration. Arch Dermatol Res 311, 55–62 (2019). https://doi.org/10.1007/s00403-018-1880-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-018-1880-2