Abstract
Introduction
Non-unions remain a clinical problem and are characterised by the failure to heal after a defined period of time. Current preclinical non-union models apply a wide variety of techniques to diminish intrinsic healing potential deviating from the clinical situation. The aim of this study was to develop and characterise a non-union model in rats using internal plate fixation without the need for additional healing insults, whereby bone healing can be longitudinally assessed using microCT. It was hypothesized that healing/non-unions can be accurately predicted at early time points by microCT.
Materials and methods
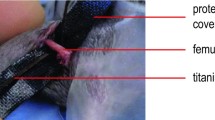
Female, skeletally mature Fischer F344 rats received a 2 mm or 1 mm femoral osteotomy, stabilized with either a 2 mm thick plate or a 1.25 mm thick plate. Healing was monitored by microCT over 14 weeks and histological analysis at euthanasia. The mechanical environment was characterised using finite element (FE) modelling and biomechanical testing.
Results
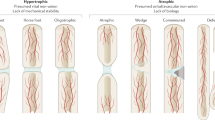
The majority of animals receiving the 2 mm thick plate displayed poor healing responses in both the 2 mm and 1 mm defect size groups. Bone and cartilage formation were markedly improved using the 1.25 mm thick plate. MicroCT could accurately predict bone forming capacity at early time points (3–4 weeks).
Conclusions
The 2 mm thick plating system confers poor healing responses in female Fischer F344 rats, comparable to atrophic non-unions. By reducing plate thickness to increase interfragmentary strain within the defect site healing is improved, leading to borderline healing situations or increased abundance of cartilage tissue present in the defect site with ultimate failure to bridge the defect (hypertrophic non-union). Furthermore, microCT can reliably identify delayed/non-healing animals within 4 weeks, thereby allowing their selective targeting for the testing of novel, clinically relevant treatment strategies in different clinical situations aimed at restoring impaired bone healing.







Similar content being viewed by others
References
Bartnikowski N, Claes LE, Koval L et al (2017) Modulation of fixation stiffness from flexible to stiff in a rat model of bone healing. ActaOrthop 88(2):217–222
Bennett PM, Stewart SK, Dretzke J, Bem D, Penn-Barwell JG (2018) Preclinical therapies to prevent or treat fracture non-union: a systematic review. PLoS ONE 13(8):e0201077
Boursinos LA, Karachalios T, Poultsides L, Malizos KN (2009) Do steroids, conventional non-steroidal anti-inflammatory drugs and selective Cox-2 inhibitors adversely affect fracture healing? J Musculoskelet Neuronal Interact 9(1):44–52
Brouwers JE, van Rietbergen B, Huiskes R (2007) No effects of in vivo micro-CT radiation on structural parameters and bone marrow cells in proximal tibia of wistar rats detected after eight weekly scans. J Orthop Res Off PublOrthop Res Soc 25(10):1325–1332
Claes L, Augat P, Suger G, Wilke HJ (1997) Influence of size and stability of the osteotomy gap on the success of fracture healing. J Orthop Res Off PublOrthop Res Soc 15(4):577–584
Dragomir-Daescu D, Op Den Buijs J, McEligot S et al (2011) Robust QCT/FEA models of proximal femur stiffness and fracture load during a sideways fall on the hip. Ann Biomed Eng 39(2):742–755
Duryea J, Evans C, Glatt V (2018) Image analysis software as a strategy to improve the radiographic determination of fracture healing. J Orthop Trauma 32(9):e354–e358
Einhorn TA, Lane JM (1998) Significant advances have been made in the way surgeons treat fractures. Clin Orthop Relat Res 355(Suppl):S2–3
Ekegren CL, Edwards ER, de Steiger R, Gabbe BJ (2018) Incidence, costs and predictors of non-union, delayed union and mal-union following long bone fracture. Int J Environ Res Public Health 15(12):284
Epari DR, Schell H, Bail HJ, Duda GN (2006) Instability prolongs the chondral phase during bone healing in sheep. Bone 38(6):864–870
Fisher JS, Kazam JJ, Fufa D, Bartolotta RJ (2018) Radiologic evaluation of fracture healing. SkeletRadiol 48:349
Garcia P, Histing T, Holstein JH et al (2013) Rodent animal models of delayed bone healing and non-union formation: a comprehensive review. Eur Cells Mater 26:1–12 (Discussion 12-14)
Glatt V, Bartnikowski N, Quirk N, Schuetz M, Evans C (2016) Reverse dynamization: influence of fixator stiffness on the mode and efficiency of large-bone-defect healing at different doses of rhBMP-2. J Bone Joint Surg. 98(8):677–687 (American volume)
Glatt V, Evans CH, Stoddart MJ (2019) Regenerative rehabilitation: The role of mechanotransduction in orthopaedic regenerative medicine. J Orthop Res OffPublOrthop Res Soc 37(6):1263–1269
Glatt V, Tepic S, Evans C (2016) Reverse dynamization: a novel approach to bone healing. J Am AcadOrthopSurg 24(7):e60-61
Goodship AE, Kenwright J (1985) The influence of induced micromovement upon the healing of experimental tibial fractures. J Bone Joint Surg 67(4):650–655 (British volume)
Klinck RJ, Campbell GM, Boyd SK (2008) Radiation effects on bone architecture in mice and rats resulting from in vivo micro-computed tomography scanning. Med EngPhys 30(7):888–895
Mills LA, Simpson AH (2012) In vivo models of bone repair. J Bone Joint Surg 94(7):865–874 (British volume)
Muller J, Schenk R, Willenegger H (1968) Experimental studies on the development of reactive pseudarthroses on the canine radius. Helvetica chirurgicaacta 35(1):301–308
Pearson RG, Clement RG, Edwards KL, Scammell BE (2016) Do smokers have greater risk of delayed and non-union after fracture, osteotomy and arthrodesis? A systematic review with meta-analysis. BMJ open 6(11):e010303
Perren SM, Rahn BA (1980) Biomechanics of fracture healing. Can J Surg. Journal canadien de chirurgie 23(3):228–232
Pountos I, Georgouli T, Blokhuis TJ, Pape HC, Giannoudis PV (2008) Pharmacological agents and impairment of fracture healing: what is the evidence? Injury 39(4):384–394
Rontgen V, Blakytny R, Matthys R et al (2010) Fracture healing in mice under controlled rigid and flexible conditions using an adjustable external fixator. J Orthop Res Off PublOrthop Res Soc 28(11):1456–1462
Salih S, Blakey C, Chan D et al (2015) The callus fracture sign: a radiological predictor of progression to hypertrophic non-union in diaphysealtibial fractures. Strateg Trauma Limb Reconstr (Online) 10(3):149–153
Santolini E, West R, Giannoudis PV (2015) Risk factors for long bone fracture non-union: a stratification approach based on the level of the existing scientific evidence. Injury 46(Suppl 8):S8-s19
Schwarz C, Ott CE, Wulsten D et al (2018) The interaction of BMP2-induced defect healing in rat and fixator stiffness modulates matrix alignment and contraction. JBMR Plus 2(3):174–186
Vicenti G, Carrozzo M, Caiaffa V et al (2019) The impact of the third fragment features on the healing of femoral shaft fractures managed with intramedullary nailing: a radiological study. IntOrthop 43(1):193–200
Wehner T, Wolfram U, Henzler T, Niemeyer F, Claes L, Simon U (2010) Internal forces and moments in the femur of the rat during gait. J Biomech 43(13):2473–2479
Woo SL, Simon BR, Akeson WH, Gomez MA, Seguchi Y (1983) A new approach to the design of internal fixation plates. J Biomed Mater Res 17(3):427–439
Zeckey C, Mommsen P, Andruszkow H et al (2011) The aseptic femoral and tibial shaft non-union in healthy patients—an analysis of the health-related quality of life and the socioeconomic outcome. Open Orthop J 5:193–197
Acknowledgements
The authors would like to thank Dr Dirk Nehrbass, Ivan Zderic, Dr Peter Varga and all the members of the Preclinical Facility at ARI Davos for their help with this study. Prof Martin Stoddart is also acknowledged for helpful comments on the manuscript.
Funding
This research was funded by the AO Foundation.
Author information
Authors and Affiliations
Contributions
MHi, MHe, WAL, FG, DG, DM, SZ and KT conducted experiments, acquired data and performed data analysis. MHi, SV, MA, SZ and KT designed the study. MHi, SZ and KT wrote the manuscript. MA and SV critically revised the manuscript. All authors have read and approve the submitted version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hildebrand, M., Herrmann, M., Gieling, F. et al. Development and characterization of a predictive microCT-based non-union model in Fischer F344 rats. Arch Orthop Trauma Surg 142, 579–590 (2022). https://doi.org/10.1007/s00402-020-03680-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-020-03680-4