Abstract
Purpose
Epidemiological and intervention studies show that milk consumption in childhood and during adolescence is related to higher bone mineral density. Milk and milk products prevent the bone loss in pre- and postmenopausal women. Apart from calcium, there are other biologically active compounds in milk such as bioactive peptides which may play a role in promoting bone health. Casein is the major protein in milk which has also been reported to have numerous biological active peptides within it. The hypothesis of the present study was to identify the key peptides behind osteoanabolic nature of the milk protein, which further can be used to prepare functional foods to alleviate bone diseases like osteoporosis. Hence, this study was carried out to investigate osteogenic nature of four novel bioactive peptides [PEP1 (EDVPSER), PEP2 (NAVPITPTL), PEP3 (VLPVPQK) and PEP4 (HPHPHLSF)] derived from buffalo casein by in vitro osteoblast differentiation model.
Methods
Calvaria cells were isolated from 3-day-old rat pups, cultured under in vitro conditions till confluence and further used for experiments. Calvarial osteoblast cells were cultured in the presence or absence of peptides including positive controls up to 21 days. Effect of peptides was checked at regular intervals by quantifying osteoblast differentiation marker genes (ALP, OCN and COL-1) expression, alkaline phosphatase activity, osteocalcin level in culture supernatants, mineral deposition by alizarin red staining and caspase-3 and 9 assays.
Results
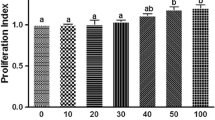
The osteoblast differentiation marker genes (ALP, OCN and COL-1) expression was significantly [(p < 0.01) (p < 0.001)] up-regulated in the presence of these peptides. The peptides also significantly induced alkaline phosphatase activity, osteocalcin level and mineral deposition in comparison with the control. It was also observed that all the four peptides did not show any cytotoxic effect during 21-day treatment period.
Conclusion
All peptides enhanced osteoblast differentiation along with the positive controls. These results hold an immense scope to use peptides as preventive measure for reducing incidence of osteoporosis. These peptides can also be used as drugs and can be utilized as functional ingredients in functional foods preparation for osteoporosis therapy, but in vivo studies are required for further confirmation.








Similar content being viewed by others
References
Leslie WD, Adler RA, Fuleihan GEH, Hodsman A, Kendler DL, McClung M, Miller PD, Watts NB (2006) International society for clinical densitometry. Application of the 1994 WHO classification to populations other than postmenopausal Caucasian women: the 2005 ISCD Official Positions. J Clin Densitom 9(1):22–30
Riggs BL, Khosla S, Melton LJ (2001) The type I/type II model for involutional osteoporosis. Academic, San Diego, pp 49–58
Musumecia G, Loretoa C, Clementib G, Fiorec CE, Martineza G (2011) An in vivo experimental study on osteopenia in diabetic rats. Acta Histochem 113:619–625
Castrogiovanni P, Trovato FM, Szychlinska MA, Nsir H, Imbesi R, Musumecia G (2016) The importance of physical activity in osteoporosis. From the molecular pathways to the clinical evidence. Histol Histopathol 31(11):1183–1194
Pichler K, Loreto C, Leonardi R, Reuber T, Weinberg AM, Musumecia G (2013) RANKL is downregulated in bone cells by physical activity (treadmill and vibration stimulation training) in rat with glucocorticoid-induced osteoporosis. Histol Histopathol 28(9):1185–1196
Musumeci G, Loreto C, Leonardi R, Castorina S, Giunta S, Carnazza ML, Trovato FM, Pichler K, Weinberg AM (2013) The effects of physical activity on apoptosis and lubricin expression in articular cartilage in rats with glucocorticoid-induced osteoporosis. J Bone Miner Metab 31:274
Owen TA, Aronow M, Shalhoub V et al (1990) Progressive development of the rat osteoblast phenotype invitro: reciprocal relationships in expression of genes associated with osteoblast proliferation and differentiation during formation of the bone extracellular matrix. J Cell Physiol 143(3):420–430
Dworetzky SI, Fey EG, Penman S, Lian JB, Stein JL, Stein GS (1990) Progressive changes in the protein composition of the nuclear matrix during rat osteoblast differentiation. Proc Natl Acad Sci USA 87:4605–4609
Beck JGR, Zerler B, Moran E (2001) Gene array analysis of osteoblast differentiation. Cell Growth Differ 12(2):61–83
Moore MAS (2005) Converging pathways in leukemogenesis and stem cell self-renewal. Exp Hematol 33:719–737
Stein GS, Lian JB, Owen TA (1990) Relationship of cell growth to the regulation of tissue-specific gene expression during osteoblast differentiation. FASEB J 4:3111–3312
Weinreb M, Shinar D, Rodan GA (1990) Different pattern of alkaline phosphatase, osteopontin, and osteocalcin expression in developing rat bone visualized by in situ hybridization. J Bone Miner Res 5:831–842
Orimo H (2010) The mechanism of mineralization and the role of alkaline phosphatase in health and disease. J Nippon Med Sch 77:4–12
Franceschi RT, Iyer BS (1992) Relationship between collagen synthesis and expression of the osteoblast phenotype in MC3T3-E1 cells. J Bone Miner Res 7:235–246
Huttunen MM, Pekkinen M, Ahlstrom MEB, Lamberg-Allarddt CJE (2008) Long-term effects of tripeptide Ile-Pro-Pro on osteoblast differentiation in vitro. J Nutr Biochem 19:708–715
Kim HK, Kim JH, Park DS, Park KS, Kang SS, Lee JS, Jeonga MH, Yoon TR (2012) Osteogenesis induced by a bone forming peptide from the prodomain region of BMP-7. Biomaterials 33:7057–7063
Liu J, Zhang B, Song S, Ma M, Si S et al (2014) Bovine collagen peptides compounds promote the proliferation and differentiation of MC3T3-E1 pre-osteoblasts. PLoS ONE 9(6):e99920. doi:10.1371/journal.pone.0099920
Fukushima N, Hiraoka K, Shirachi I, Kojima M, Nagata K (2010) Isolation and characterization of a novel peptide, osteoblast activating peptide (OBAP), associated with osteoblast differentiation and bone formation. Biochem Biophys Res Commun 400:157–163
Fukushima N, Hanada R, Teranishi H et al (2005) Ghrelin directly regulates bone formation. J Bone Min Res 20(5):790–798
Widyowati R (2001) Alkaline phosphatase activity of Graptophyllum pictum and Sphilanthes acmella fractions against MC3T3-E1 cells as marker of osteoblast differentiation cells. Int J Pharm Pharm Sci 3(1):34–37
FitzGerald RJ, Meisel H (2000) Milk protein-derived peptide inhibitors of angiotensin-converting enzyme. Br J Nutr 84(1):S33–S37
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction. Anal Biochem 162:156–159
Haque E, Chand R, Kapila S (2009) Biofunctional properties of bioactive peptides of milk origin. Food Rev Int 25(1):28–43
Shimizu M (2004) Food-derived peptides and intestinal functions. BioFactors 21:43–47
Chee WSS, Suriah AR, Chan SP, Zaitun Y, Chan YM (2003) The effect of milk supplementation on bone mineral density in postmenopausal Chinese women in Malaysia. Osteoporos Int 14:828–834
Matkovic V, Landoll JD, Badenhop-Stevens NE et al (2004) Nutrition influences skeletal development from childhood to adulthood: a study of hip, spine and forearm in adolescent females. J Nutr 134:7015–7055
Behera PS, Kumar R, Sandeep IVR, Kapila R, Dang AK, Kapila S (2013) Casein hydrolysates enhances osteoblast proliferation and differentiation in mouse bone marrow culture. Food Biosci 2:24–30
Reddi S, Shanmugam VP, Kapila S, Kapila R (2016) Identification of buffalo casein derived bioactive peptides with osteoblast proliferation activity. Eur Food Res Technol. doi:10.1007/s00217-016-2710-4
Gregory CA, Gunn WG, Peister A, Prockop DJ (2004) An Alizarin red-based assay of mineralization by adherent cells in culture: comparison with cetylpyridinium chloride extraction. Anal Biochem 329(1):77–84
Yoon K, Buenaga R, Rodan GA (1987) Tissue specificity and developmental expression of rat osteopontin. Biochem Biophys Res Commun 148:1129–1136
Strauss PG, Closs EI, Schmidt J, Erfle V (1990) Gene expression during osteogenic differentiation in mandibular condyles in vitro. J Cell Biol 110:1369–1378
Patti A, Gennari L, Merlotti D, Dotta F, Nuti R (2013) Endocrine actions of osteocalcin. Int J Endocrinol 2013:846480. doi:10.1155/2013/846480
Hauschka PV, Lian JB, Cole DE, Gundberg CM (1989) Osteocalcin and matrix Gla protein: vitamin K-dependent proteins in bone. Physiol Rev 69:990–1047
Yamaguchi A, Katagiri T, Ikeda T et al (1991) Recombinant human bone morphogenetic protein-2 stimulates osteoblastic maturation and inhibits myogenic differentiation in vitro. J Cell Biol 113:681–687
Koh ET, Torabinejad M, PittFord TR, Brady K, McDonald F (1997) Mineral trioxide aggregate stimulates a biological response in human osteoblasts. J Biomed Mater Res 37:432–439
Spreafico A, Frediani B, Capperucci C et al (2006) Osteogenic growth peptide effects on primary human osteoblast cultures: potential relevance for the treatment of glucocorticoid-induced osteoporosis. J Cell Biochem 98:1007–1020
Kostenuik PJ et al (1999) Skeletal unloading causes resistance of osteoprogenitor cells to parathyroid hormone and to insulin-like growth factor-I. J Bone Miner Res 14:21
Kasperk C et al (1995) Human bone cell phenotypes differ depending on their skeletal site of origin. J Clin Endocrinol Metab 80(8):2511–2517
Huttunen MM, Pekkinen M, Ahlstrom MEB, Lamberg-Allarddt CJE (2007) Effects of bioactive peptides isoleucine–proline–proline (IPP), valine–proline–proline (VPP) and leucine-lysine-proline (LKP) on gene expression of osteoblasts differentiated from human mesenchymal stem cell. Br J Nutr 98:780–788
Choi YJ, Lee JY, Chung CP, Park YJ (2012) Enhanced osteogenesis by collagen-binding peptide from bone sialoprotein in vitro and in vivo. J Biomed Mater Res Part A 101(2):547–554
Wolf BB, Green DR (1999) Suicidal tendencies: apoptotic cell death by caspase family proteinases. J Biol Chem 274:20049–20052
Bump NJ, Hackett M, Hugunin M et al (1995) Inhibition of ICE family proteases by baculovirus antiapoptotic protein p35. Science 269:1885–1888
Nicholson DW, Thornberry NA (1997) Caspases: killer proteases. Trends Biochem Sci 22:299–306
Plotkin LI, Robert S, Weinstein A et al (1999) Prevention of osteocyte and osteoblast apoptosis by bisphosphonates and calcitonin. J Clin Investig 104:1363–1374
Chua CC, Chua BHL, Chen Z, Landy C, Hamdy RC (2003) Dexamethasone induces caspase activation in murine osteoblastic MC3T3-E1 cells. Biochim Biophys Acta 1642:79–85
Kim SW, Her SJ, Park SJ et al (2005) Ghrelin stimulates proliferation and differentiation and inhibits apoptosis in osteoblastic MC3T3-E1 cells. Bone 37:359–369
Tang S, Xie H, Yuan L et al (2007) Apelin stimulates proliferation and suppresses apoptosis of mouse osteoblastic cell line MC3T3-E1 via JNK and PI3-K/Akt signaling pathways. Peptides 28:708–718
Navarrete R, Hyzy SL, Almaguer-Flores A, Mauth C, Gemperli AC, Boyan BD, Schwartz Z (2013) Amelogenin peptide extract increases differentiation and angiogenic and local factor production and inhibits apoptosis in human osteoblasts. Biomaterials. doi:10.5402/2013/347318
Acknowledgements
The authors are grateful to the Director, ICAR-National Dairy Research Institute, Karnal, for providing funding and laboratory facilities to carry out this piece of work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Reddi, S., Shanmugam, V.P., Tanedjeu, K.S. et al. Effect of buffalo casein-derived novel bioactive peptides on osteoblast differentiation. Eur J Nutr 57, 593–605 (2018). https://doi.org/10.1007/s00394-016-1346-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-016-1346-2