Abstract
Background
The recurrent risk of stage I colorectal cancer (CRC) is not clear, and the data regarding appropriate post-operative surveillance schedules in stage I CRC are scarce.
Objectives
We aimed to stratify stage I CRC based on the recurrence risk and evaluate optimal post-operative surveillance durations based on this stratification.
Methods
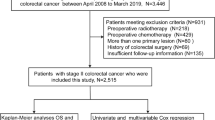
We retrospectively analyzed the cases of 6607 stage I CRC patients from 24 institutions. To assess the patients’ clinicopathological factors that impact recurrence-free survival (RFS), we performed univariate and multivariate analyses using Cox proportional hazards models. We divided the patients into classes based on their numbers of factors that were associated with poor RFI in the multivariate analysis.
Results
Recurrence occurred in 3.9% patients. The multivariate analysis revealed the independent factors for poor RFS: rectal cancer, T2 depth, presence of lymphatic invasion, high level of pre-operative carcinoembryonic antigen, and absence of D2–3 lymphadenectomy. We also divided the patients into three classes based on their numbers of these risk factors; the 3-year and 5-year RFS rates were 99.3% and 99.1% in the no-risk patients, 97.4% and 96.5% in the patients with 1–2 risks, and 92.1% and 90.0% in the patients with 3–5 risks, respectively. In the patients with no risk and in the patients with 1–2 risks after 3 years post-surgery, ≤ 1% recurrence occurred. Thus, post-operative surveillance may be omitted in these populations.
Conclusions
Our new classification properly stratified the recurrence risks of stage I CRC patients, and may help reduce unnecessary post-operative surveillance.



Similar content being viewed by others
References
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, Meester RGS, Barzi A, Jemal A (2017) Colorectal cancer statistics, 2017. CA Cancer J Clin 67(3):177–193
Kattan MW, Hess KR, Amin MB, Lu Y, Moons KG, Gershenwald JE, Gimotty PA, Guinney JH, Halabi S, Lazar AJ, Mahar AL, Patel T, Sargent DJ, Weiser MR, Compton C, members of the AJCC Precision Medicine Core (2016) American Joint Committee on Cancer acceptance criteria for inclusion of risk models for individualized prognosis in the practice of precision medicine. CA Cancer J Clin 66(5):370–374
Hashiguchi Y, Muro K, Saito Y et al (2019) Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Int J Clin Oncol. 25(1):1–42
Kim C, Kim WR, Kim KY, Chon HJ, Beom SH, Kim H, Jung M, Shin SJ, Kim NK, Ahn JB (2018) Predictive nomogram for recurrence of stage I colorectal cancer after curative resection. Clin Colorectal Cancer 17(3):e513–e518
Blumberg D, Paty PB, Picon AI et al (1998) Stage I rectal cancer: identification of high-risk patients. J Am Coll Surg 186(5):574–579 discussion 579-80
Costas-Chavarri A, Nandakumar G, Temin S, Lopes G, Cervantes A, Cruz Correa M, Engineer R, Hamashima C, Ho GF, Huitzil FD, Malekzadeh Moghani M, Sharara AI, Stern MC, Teh C, Vázquez Manjarrez SE, Verjee A, Yantiss R, Shah MA (2019) Treatment of patients with early-stage colorectal cancer: ASCO resource-stratified guideline. J Glob Oncol 5:1–19
Rodriguez-Moranta F, Salo J, Arcusa A et al (2006) Postoperative surveillance in patients with colorectal cancer who have undergone curative resection: a prospective, multicenter, randomized, controlled trial. J Clin Oncol 24(3):386–393
van der Stok EP, Spaander MCW, Grunhagen DJ et al (2017) Surveillance after curative treatment for colorectal cancer. Nat Rev Clin Oncol 14(5):297–315
Sargent D, Sobrero A, Grothey A, O’Connell MJ, Buyse M, Andre T, Zheng Y, Green E, Labianca R, O’Callaghan C, Seitz JF, Francini G, Haller D, Yothers G, Goldberg R, de Gramont A (2009) Evidence for cure by adjuvant therapy in colon cancer: observations based on individual patient data from 20,898 patients on 18 randomized trials. J Clin Oncol 27(6):872–877
Hashiguchi Y, Hase K, Ueno H, Mochizuki H, Shinto E, Yamamoto J (2011) Optimal margins and lymphadenectomy in colonic cancer surgery. Br J Surg 98(8):1171–1178
Benson AB 3rd, Schrag D, Somerfield MR et al (2004) American Society of Clinical Oncology recommendations on adjuvant chemotherapy for stage II colon cancer. J Clin Oncol 22(16):3408–3419
Grothey A, Sobrero AF, Shields AF, Yoshino T, Paul J, Taieb J, Souglakos J, Shi Q, Kerr R, Labianca R, Meyerhardt JA, Vernerey D, Yamanaka T, Boukovinas I, Meyers JP, Renfro LA, Niedzwiecki D, Watanabe T, Torri V, Saunders M, Sargent DJ, Andre T, Iveson T (2018) Duration of adjuvant chemotherapy for stage III colon cancer. N Engl J Med 378(13):1177–1188
Salazar R, Roepman P, Capella G, Moreno V, Simon I, Dreezen C, Lopez-Doriga A, Santos C, Marijnen C, Westerga J, Bruin S, Kerr D, Kuppen P, van de Velde C, Morreau H, van Velthuysen L, Glas AM, van’t Veer LJ, Tollenaar R (2011) Gene expression signature to improve prognosis prediction of stage II and III colorectal cancer. J Clin Oncol 29(1):17–24
Sinicrope FA, Shi Q, Smyrk TC, Thibodeau SN, Dienstmann R, Guinney J, Bot BM, Tejpar S, Delorenzi M, Goldberg RM, Mahoney M, Sargent DJ, Alberts SR (2015) Molecular markers identify subtypes of stage III colon cancer associated with patient outcomes. Gastroenterology 148(1):88–99
Teloken PE, Ransom D, Faragher I, Jones I, Gibbs P, Platell C (2016) Recurrence in patients with stage I colorectal cancer. ANZ J Surg 86(1–2):49–53
Chok KS, Law WL (2007) Prognostic factors affecting survival and recurrence of patients with pT1 and pT2 colorectal cancer. World J Surg 31(7):1485–1490
Mroczkowski P, Schmidt U, Sahm M, Gastinger I, Lippert H, Kube R (2012) Prognostic factors assessed for 15,096 patients with colon cancer in stages I and II. World J Surg 36(7):1693–1698
Kang SI, Kim DW, Kwak Y, Lee HS, Kim MH, Kim MJ, Oh HK, Kang SB (2018) The prognostic implications of primary tumor location on recurrence in early-stage colorectal cancer with no associated risk factors. Int J Color Dis 33(6):719–726
Bertelsen CA, Neuenschwander AU, Jansen JE, Tenma JR, Wilhelmsen M, Kirkegaard-Klitbo A, Iversen ER, Bols B, Ingeholm P, Rasmussen LA, Jepsen LV, Born PW, Kristensen B, Kleif J (2019) 5-year outcome after complete mesocolic excision for right-sided colon cancer: a population-based cohort study. Lancet Oncol 20(11):1556–1565
Primrose JN, Perera R, Gray A, Rose P, Fuller A, Corkhill A, George S, Mant D, FACS Trial Investigators (2014) Effect of 3 to 5 years of scheduled CEA and CT follow-up to detect recurrence of colorectal cancer: the FACS randomized clinical trial. JAMA 311(3):263–270
Snyder RA, Hu CY, Cuddy A, Francescatti AB, Schumacher JR, van Loon K, You YN, Kozower BD, Greenberg CC, Schrag D, Venook A, McKellar D, Winchester DP, Chang GJ, for the Alliance for Clinical Trials in Oncology Network Cancer Surveillance Optimization Working Group (2018) Association between intensity of posttreatment surveillance testing and detection of recurrence in patients with colorectal cancer. JAMA 319(20):2104–2115
Benson AB, Venook AP, Al-Hawary MM et al (2018) NCCN guidelines insights: colon cancer, version 2.2018. J Natl Compr Cancer Netw 16(4):359–369
Steele SR, Chang GJ, Hendren S, Weiser M, Irani J, Buie WD, Rafferty JF (2015) Practice guideline for the surveillance of patients after curative treatment of colon and rectal cancer. Dis Colon Rectum 58(8):713–725
Schmoll HJ, Van Cutsem E, Stein A et al (2012) ESMO consensus guidelines for management of patients with colon and rectal cancer. A personalized approach to clinical decision making. Ann Oncol 23(10):2479–2516
Labianca R, Nordlinger B, Beretta GD et al (2013) Early colon cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 24(Suppl 6):vi64–vi72
Tsikitis VL, Malireddy K, Green EA, Christensen B, Whelan R, Hyder J, Marcello P, Larach S, Lauter D, Sargent DJ, Nelson H (2009) Postoperative surveillance recommendations for early stage colon cancer based on results from the clinical outcomes of surgical therapy trial. J Clin Oncol 27(22):3671–3676
Okamura R, Hida K, Nishizaki D, Sugihara K, Sakai Y (2018) Proposal of a stage-specific surveillance strategy for colorectal cancer patients: a retrospective analysis of Japanese large cohort. Eur J Surg Oncol 44(4):449–455
Hutchins G, Southward K, Handley K, Magill L, Beaumont C, Stahlschmidt J, Richman S, Chambers P, Seymour M, Kerr D, Gray R, Quirke P (2011) Value of mismatch repair, KRAS, and BRAF mutations in predicting recurrence and benefits from chemotherapy in colorectal cancer. J Clin Oncol 29(10):1261–1270
Taieb J, Le Malicot K, Shi Q et al (2017) Prognostic value of BRAF and KRAS mutations in MSI and MSS stage III colon cancer. J Natl Cancer Inst 109(5):djw272
Petrelli F, Tomasello G, Borgonovo K, Ghidini M, Turati L, Dallera P, Passalacqua R, Sgroi G, Barni S (2017) Prognostic survival associated with left-sided vs right-sided colon cancer: a systematic review and meta-analysis. JAMA Oncol 3(2):211–219
Mou S, Soetikno R, Shimoda T, Rouse R, Kaltenbach T (2013) Pathologic predictive factors for lymph node metastasis in submucosal invasive (T1) colorectal cancer: a systematic review and meta-analysis. Surg Endosc 27(8):2692–2703
Acknowledgments
The authors thank the members of the Japanese Study Group for Postoperative Follow-up of CRC: Akio Shiomi (Shizuoka Cancer Center); Heita Ozawa (Tochigi Cancer Center); Hideki Ueno (National Defense Medical College); Hitoshi Kameyama (Niigata University); Ichiro Takemasa (Sapporo Medical University); Keiichi Takahashi (Tokyo Metropolitan Cancer and Infectious diseases Center Komagome Hospital); Kenichi Hakamada (Hirosaki University); Koji Okabayashi (Keio University); Koji Komori (Aichi Cancer Center Hospital); Masahiko Watanabe (Kitasato University); Masayuki Ohue (Osaka Medical Center for Cancer and Cardiovascular Diseases); Michio Itabashi (Tokyo Women’s Medical University); Naohiro Tomita (Hyogo College of Medicine); Shingo Noura (Osaka International Cancer Institute); Tadahiko Masaki (Kyorin University); Tomomichi Kiyomatsu (National Center for Global Health and Medicine); Tsunekazu Hanai (Fujita Health University); Yasukimi Takii (Niigata Cancer Center Hospital); Yoshiharu Sakai (Kyoto University); Yoshito Akagi (Kurume University); Yukihide Kanemitsu (National Cancer Center Hospital); and Yusuke Kinugasa (Tokyo Medical and Dental University). All the authors have read the manuscript and have approved this submission.
Author information
Authors and Affiliations
Contributions
Study conception, study design, data analysis, and manuscript drafting were done by T.O.
Data acquisition was done by T. O, T. H, T. T, K.N., and K.M.
Manuscript editing and critical revise were done by Y. H, S. I, T. H, T. T, K. N, S. Y, K.S., and K.M
Corresponding author
Ethics declarations
The study was approved by the Central Institutional Review Board (Tokyo Medical and Dental University) and local ethics committee (Teikyo University ethics committee No. 19-127).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ozawa, T., Hashiguchi, Y., Ishihara, S. et al. Proposal for a post-operative surveillance strategy for stage I colorectal cancer patients based on a novel recurrence risk stratification: a multicenter retrospective study. Int J Colorectal Dis 36, 67–74 (2021). https://doi.org/10.1007/s00384-020-03737-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-020-03737-1