Abstract
Purpose
Although some biomarkers of hepatic progenitor cells have been reported to be involved in the liver fibrosis in patients with biliary atresia (BA), however, research still shows conflicting results. Therefore, we investigated the effect of Alu-mediated p21 transcriptional regulator (APTR), fibroblast growth factor-inducible 14 (Fn14) and CD133 expressions on liver fibrosis in Indonesian BA patients.
Methods
Nineteen liver samples from BA patients and 9 liver specimens from non-BA controls were obtained. The expressions of APTR, Fn14 and CD133 were analyzed by quantitative real-time polymerase chain reaction (qPCR).
Results
APTR expression was strongly up-regulated (1.5-fold) in liver BA specimens compared to liver controls (ΔCT 3.2 ± 0.6 vs 3.8 ± 0.51; p = 0.028). Moreover, Fn14 and CD133 expressions were similar in the BA and control groups (ΔCT 2.7 ± 1.3 vs. 1.4 ± 1.6, p = 0.07; and 12.0 ± 3.7 vs. 11.78 ± 2.30, p = 0.88, respectively). Intriguingly, CD133 expression was strongly related with the survival of BA patients (p = 0.0061), but not with age at Kasai procedure (p = 0.36) and the presence of cirrhosis (p = 0.77).
Conclusion
We present the first study of aberrant APTR expressions in the liver of BA infants which might contribute to liver fibrogenesis in BA infants.
Similar content being viewed by others
Introduction
Biliary atresia (BA) is characterized by a fibro-obliterative disorder of the intra- and extrahepatic biliary system in infants, resulting in end-stage liver disease if the Roux-en-Y portoenterostomy (Kasai) surgery is not conducted at age of < 90 days [1,2,3]. BA incidence differs between ethnic group, from 1:5–7000 in Asian [3,4,5,6] to about 1 in 15–20,000 in Caucasian patients [7,8,9].
Although some biomarkers of hepatic progenitor cells have been reported to be involved in the liver fibrosis of biliary atresia patients, including fibroblast growth factor-inducible 14 (Fn14) and CD133, however, research still shows conflicting results [10,11,12]. Furthermore, long non-coding RNA (lncRNA) Alu-mediated p21 transcriptional regulator (APTR) was up-regulated in the fibrotic livers of cirrhosis patients and activated hepatic stellate cells (HSCs) [13], however, the role of APTR in liver fibrosis of biliary atresia patients is not known yet. Nevertheless, some common variants’ frequency varies among Asia population [14] and our study revealed the significant association between the SEMA3 rs11766001 effect on Hirschsprung disease and population background [15]. Therefore, we investigated the long non-coding RNA (lncRNA) APTR, Fn14, and CD133 expressions impact on liver fibrosis in Indonesian BA patients.
Materials and methods
Subjects
We recruited the BA patients who underwent Kasai procedure at Dr. Sardjito Hospital, Indonesia [6, 12] from October 2017 to December 2018. BA patients and controls parents signed an informed consent before joining the study. Nineteen liver samples of BA patients were taken during the Kasai procedure, while 9 controls liver samples were collected during the surgery for other diseases as follows: gallbladder hydrops (1), omphalocele (1), internal bleeding (1), liver abscess (1), and choledochal cyst (5). The median age of controls was 108 (IQR, 84–144) months.
Liver histopathological findings
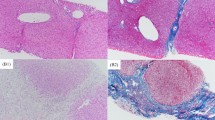
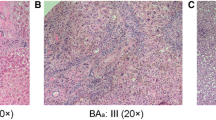
Liver histopathological findings of patients with BA were determined using the Laennec system [12, 16].
Quantitative real-time polymerase chain reaction (qPCR)
qPCR was conducted according to our recent study [12] using the following primer sets: APTR 5′-TTGCCAAACGGTTCTTTC-3′ (forward) and 5′-CGGAGGAGAGTAAATACAACAG-3′ (reverse); Fn14 5′-CCACTCATCATTCATTCATCC-3′ (forward) and 5′-AGTTCCTTAGTCAGTGTCAG-3′ (reverse); and CD133 5′-AGTCGGAAACTGGCAGATAGC-3′ (forward) and 5′-GGTAGTGTTGTACTGGGCCAAT-3′ (reverse). We utilized GAPDH as reference gene [11,12,13]. We determined APTR, Fn14, and CD133 expressions using Livak (2−ΔΔCT) method [12, 17].
Statistical analysis
The baseline characteristics were presented as frequency (percentage) or median [interquartile range (IQR)], while the APTR, Fn14, and CD133 expressions were presented as mean values ± SD. We analyzed the statistical difference of gene expressions using t test and considered p value < 0.05 as a significant level.
Results
Patients’ features
There were 4, 5 and 10 patients with BA type 2A, 2B and 3, whereas the age of patients underwent Kasai surgery was 98 (IQR, 66.5–129) days. The survival rate of BA patients after Kasai surgery was 52.6% (Fig. 1). The liver biopsy was performed simultaneously during the Kasai procedure and showed a liver cirrhosis in 26.3% patients (Table 1). Among 19 BA patients after Kasai procedure in our institution, 1 patient underwent a living donor liver transplantation at age of 14 months due to failed Kasai portoenterostomy.
Expressions of APTR, Fn14 and CD133 in liver BA patients and controls
The APTR expression was strongly increased (1.5-fold) in liver BA specimens compared to liver controls (ΔCT 3.2 ± 0.6 vs 3.8 ± 0.51; p = 0.028) (Fig. 2). Moreover, the Fn14 and CD133 expressions were not different between the BA and control groups (ΔCT 2.7 ± 1.3 vs. 1.4 ± 1.6, p = 0.07; and 12.0 ± 3.7 vs. 11.78 ± 2.30, p = 0.88, respectively) (Table 2).
The APTR, Fn14, and CD133 expressions in the BA patients and control liver. The APTR expression in the BA patients’ liver is significantly up-regulated (1.5-fold) compared with the controls; liver (p = 0.037). Data represent the means ± SD of APTR expressions normalized to GAPDH and relative to control tissues. *p < 0.05
Expressions of APTR, Fn14, CD133 and patients’ characteristics
Subsequently, we associated the expressions of APTR, Fn14 and CD133 and the age at Kasai surgery performed, liver cirrhosis, and survival. Interestingly, the CD133 expression was strongly linked with the BA patients’ survival (p = 0.0061), but neither with the presence of cirrhosis (p = 0.77) nor with age at Kasai procedure (p = 0.36) (Table 3). Moreover, the APTR and Fn14 expressions did not associate with the presence of cirrhosis (p = 0.53 and 0.33), the age at Kasai procedure (p = 0.42 and 0.87), and the survival of BA patients (p = 0.78 and 0.95), respectively. (Table 3).
Discussion
We show new evidence on the APTR, Fn14, and CD133 expressions in BA patients in Indonesia. We are able to show the impact of lncRNA APTR expression in the development of liver fibrosis in BA patients in Indonesia by presenting its 1.5-fold up-regulated expressions in the BA group compared to the control. This finding provides new proof of the APTR role in the liver fibrosis in BA.
It has been shown that APTR was up-regulated in liver cirrhosis both in mice and human subjects and activated HSCs [13]. They also revealed that APTR knockdown diminishes the α-SMA expression [13]. α-SMA was strongly expressed during liver fibrogenesis [12, 18]. Intriguingly, APTR is increased in the liver cirrhotic patients’ serum as well [13]. Therefore, it is interesting to determine whether the APTR expression is also increased in the serum of BA patients with liver cirrhosis compared to those patients without liver cirrhosis. In addition, our previous study has shown that miRNA-21 expression was increased in BA infants compared to controls [12]. Both miRNAs and lnRNAs are non-coding RNA that are stable and easily measurable in human serum [12, 19, 20], thus, have great potential as a novel biomarker for liver cirrhosis in BA patients in the future. Furthermore, the APTR was described as a good predictor biomarker for the portal hypertension patients following transjugular intrahepatic portosystemic shunt procedure [21].
In this study, we classified the histopathology of liver using the Laennec system [12, 16]. There are several other liver fibrosis scoring systems, such as Ishak and Okamoto scoring system [22], however, we do not have data on those scoring systems due to our research was a retrospective design, becoming a weakness of our study.
lnRNA is a non-coding RNA with > 200 nucleotides and is unable to be translated into protein [23, 24]. Its deregulation is involved in various biological process and many human diseases, including liver fibrogenesis and cirrhotic [13, 21, 23]. However, we provide three novelties in our reports: (1) the study was performed in Indonesian patients (versus Chinese cases [13]), (2) we determined the impact of APTR expression in liver BA patients (versus the APTR expression in liver cirrhotic patients [13, 21], and (3) we analyzed the effect of APTR, Fn14 and CD133 expressions in BA patients following Kasai procedure (versus the impact of Fn14 and CD133 expression in BA patients [11]).
Our findings imply that the liver fibrogenesis in BA patients is a complex process involving epigenetics, such as lncRNAs. In addition, our study should be interpreted cautiously because of a small sample size and might be considered as a preliminary data. Further study with a larger sample size and the changes of APTR, Fn14, and CD133 expressions level according to the liver fibrosis progression is important to confirm our findings. Moreover, the median age of our BA patients was 98 days, that might be far too older than the recommended age for the Kasai procedure and affect the liver fibrosis progression. Therefore, these facts should be taken into consideration during interpretation of our findings.
Contrary to the APTR expression, Fn14 and CD133 expressions did not show any differences between BA patients and controls. Our findings are different from a recent report [11]. These differences might be affected by genetic background of population [14, 15, 18, 25]. Interestingly, CD133 expression was significantly associated with our BA patients’ survival. CD133 was highly expressed in the hepatic progenitor cells in the liver fibrogenesis [11, 26]. CD133 is a transmembrane cell-surface glycoprotein and a marker of progenitor cells and cancer stem cells, including intrahepatic cholangiocarcinoma (ICC) and hepatocellular carcinoma (HCC) [27]. Intriguingly, CD133 expressions were also associated with the survival (i.e., poor prognosis) of both ICC and HCC patients [27, 28]. Again, further research with a larger sample size is necessary to clarify the association between the CD133 expressions and the BA patients’ survival.
In conclusion, we present the first study of aberrant APTR expressions in the liver of BA infants which might contribute to liver fibrogenesis in BA infants.
References
Sokol RJ, Shepherd RW, Superina R, Bezerra JA, Robuck P, Hoofnagle JH (2007) Screening and outcomes in biliary atresia: summary of a National Institutes of Health workshop. Hepatology 46:566–581
Lakshminarayanan B, Davenport M (2016) Biliary atresia: a comprehensive review. J Autoimmun 73:1–9
Sundaram SS, Mack CL, Feldman AG, Sokol RJ (2017) Biliary atresia: indications and timing of liver transplantation and optimization of pretransplant care. Liver Transpl 23:96–109
Wada H, Muraji T, Yokoi A et al (2007) Insignificant seasonal and geographical variation in incidence of biliary atresia in Japan: a regional survey of over 20 years. J Pediatr Surg 42:2090–2092
Chiu CY, Chen PH, Chan CF, Chang MH, Wu TC, Taiwan Infant Stool Color Card Study Group et al (2013) Biliary atresia in preterm infants in Taiwan: a nationwide survey. J. Pediatr 163:100–103
Gunadi, Gunawan TA, Widiyanto G, Yuanita A, Mulyani NS, Makhmudi A (2018) Liver transplant score for prediction of biliary atresia patients' survival following Kasai procedure. BMC Res Notes 11:381
Chardot C, Buet C, Serinet MO, Golmard JL, Lachaux A, Roquelaure B et al (2013) Improving outcomes of biliary atresia: French national series 1986–2009. J Hepatol 58:1209–1217
Livesey E, Cortina Borja M, Sharif K et al (2009) Epidemiology of biliary atresia in England and Wales (1999–2006). Arch Dis Child Fetal Neonatal Ed 94:F451–F455
Schreiber RA, Barker CC, Roberts EA et al (2007) Biliary atresia: the Canadian experience. J Pediatr 151:659–665
Dong R, Shen Z, Zheng C, Chen G, Zheng S (2016) Serum microRNA microarray analysis identifies miR-4429 and miR-4689 are potential diagnostic biomarkers for biliary atresia. Sci Rep 6:21084
Zheng L, Lv Z, Gong Z, Sheng Q, Gao Z, Zhang Y, Yu S, Zhou J, Xi Z, Wang X (2017) Fn14 hepatic progenitor cells are associated with liver fibrosis in biliary atresia. Pediatr Surg Int 33:593–599
Makhmudi A, Kalim AS, Gunadi (2019) microRNA-21 expressions impact on liver fibrosis in biliary atresia patients. BMC Res Notes 12:189
Yu F, Zheng J, Mao Y, Dong P, Li G, Lu Z, Guo C, Liu Z (2015) Fan X (2015) Long non-coding RNA APTR promotes the activation of hepatic stellate cells and the progression of liver fibrosis. Biochem Biophys Res Commun 463:679–685
Tajima A, Pan IH, Fucharoen G, Fucharoen S, Matsuo M, Tokunaga K et al (2002) Three major lineages of Asian Y chromosomes: implications for the peopling of east and southeast Asia. Hum Genet 110:80–88
Gunadi, Makhmudi A, Agustriani N, Rochadi (2016) Effects of SEMA3 polymorphisms in Hirschsprung disease patients. Pediatr Surg Int 32:1025–1028
Kim SU, Oh HJ, Wanless IR, Lee S, Han KH, Park YN (2012) The Laennec staging system for histological sub-classification of cirrhosis is useful for stratification of prognosis in patients with liver cirrhosis. J Hepatol 57:556–563
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25:402–408
Huang GC, Zhang JS, Tang QQ (2004) Involvement of C/EBP-alpha gene in in vitro activation of rat hepatic stellate cells. Biochem Biophys Res Commun 324:1309–1318
Yuan SX, Yang F, Yang Y, Tao QF, Zhang J, Huang G et al (2012) Long noncoding RNA associated with microvascular invasion in hepatocellular carcinoma promotes angiogenesis and serves as a predictor for hepatocellular carcinoma patients' poor recurrence-free survival after hepatectomy. Hepatology 56:2231–2241
Lorenzen JM, Schauerte C, Kielstein JT, Hübner A, Martino F, Fiedler J et al (2015) Circulating long noncoding RNATapSaki is a predictor of mortality in critically ill patients with acute kidney injury. Clin Chem 61:191–201
Yu S, Qi Y, Jiang J, Wang H, Zhou Q (2018) APTR is a prognostic marker in cirrhotic patients with portal hypertension during TIPS procedure. Gene 645:30–33
Mannan R, Misra V, Misra SP, Singh PA, Dwivedi M (2014) A comparative evaluation of scoring systems for assessing necro-inflammatory activity and fibrosis in liver biopsies of patients with chronic viral hepatitis. J Clin Diagn Res 8:FC08-12
Yang Z, Jiang S, Shang J, Jiang Y, Dai Y, Xu B et al (2019) LncRNA: shedding light on mechanisms and opportunities in fibrosis and aging. Ageing Res Rev 52:17–31
Teng KY, Ghoshal K (2015) Role of noncoding RNAs as biomarker and therapeutic targets for liver fibrosis. Gene Expr 16:155–162
Gunadi, Budi NYP, Kalim AS, Santiko W, Musthofa FD, Iskandar K, Makhmudi A (2019) Aberrant expressions of miRNA-206 target, FN1, in multifactorial Hirschsprung disease. Orphanet J Rare Dis 14:5
Kuramitsu K, Sverdlov DY, Liu SB, Csizmadia E, Burkly L, Schuppan D, Hanto DW, Otterbein LE, Popov Y (2013) Failure of fibrotic liver regeneration in mice is linked to a severe fibrogenic response driven by hepatic progenitor cell activation. Am J Pathol 183:182–194
Glumac PM, LeBeau AM (2018) The role of CD133 in cancer: a concise review. Clin Transl Med 7:18
Cai X, Li J, Yuan X, Xiao J, Dooley S, Wan X, Weng H, Lu L (2018) CD133 expression in cancer cells predicts poor prognosis of non-mucin producing intrahepatic cholangiocarcinoma. J Transl Med 16:50
Acknowledgements
We want to give thanks to everybody who assisted the study. We are also thankful to an English editing service staff at the Faculty of Medicine, Public Health and Nursing, Universitas Gadjah Mada (FK-KMK UGM) for checking the manuscript grammar.
Funding
This study was funded by the FK-KMK UGM (Grant nos. UPPM/220/M/05/04/05.17 and UPPM/363/M/05/04/05.18 to G and AM).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no potential conflicts of interest.
Ethical approval
This study has an approval from the Ethical Committee of the FK-KMK UGM/Dr. Sardjito Hospital (KE/FK/1252/EC/2018 and KE/FK/1088/EC/2017).
Informed consent
Written informed consent was obtained from the parents’ BA patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Makhmudi, A., Supanji, R., Putra, B.P. et al. The effect of APTR, Fn14 and CD133 expressions on liver fibrosis in biliary atresia patients. Pediatr Surg Int 36, 75–79 (2020). https://doi.org/10.1007/s00383-019-04582-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-019-04582-2