Abstract
Purpose
To study pulmonary hypoplasia (PH) associated with congenital diaphragmatic hernia (CDH), investigators have been employing a fetal rat model based on nitrofen administration to dams. Herein, we aimed to: (1) investigate the validity of the model, and (2) synthesize the main biological pathways implicated in the development of PH associated with CDH.
Methods
Using a defined strategy, we conducted a systematic review of the literature searching for studies reporting the incidence of CDH or factors involved in PH development. We also searched for PH factor interactions, relevance to lung development and to human PH.
Results
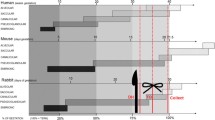
Of 335 full-text articles, 116 reported the incidence of CDH after nitrofen exposure or dysregulated factors in the lungs of nitrofen-exposed rat fetuses. CDH incidence: 54% (27–85%) fetuses developed a diaphragmatic defect, whereas the whole litter had PH in varying degrees. Downregulated signaling pathways included FGF/FGFR, BMP/BMPR, Sonic Hedgehog and retinoid acid signaling pathway, resulting in a delay in early epithelial differentiation, immature distal epithelium and dysfunctional mesenchyme.
Conclusions
The nitrofen model effectively reproduces PH as it disrupts pathways that are critical for lung branching morphogenesis and alveolar differentiation. The low CDH rate confirms that PH is an associated phenomenon rather than the result of mechanical compression alone.




Similar content being viewed by others
References
Burgos CM, Frenckner B (2017) Addressing the hidden mortality in CDH: a population-based study. J Pediatr Surg 52:522–525. https://doi.org/10.1016/j.jpedsurg.2016.09.061
McGivern MR, Best KE, Rankin J, Wellesley D, Greenlees R, Addor M-C, Arriola L, de Walle H, Barisic I, Beres J, Bianchi F, Calzolari E, Doray B, Draper ES, Garne E, Gatt M, Haeusler M, Khoshnood B, Klungsoyr K, Latos-Bielenska A, O’Mahony M, Braz P, McDonnell B, Mullaney C, Nelen V, Queisser-Luft A, Randrianaivo H, Rissmann A, Rounding C, Sipek A, Thompson R, Tucker D, Wertelecki W, Martos C (2015) Epidemiology of congenital diaphragmatic hernia in Europe: a register-based study. Arch Dis Child Fetal Neonatal Ed 100:F137–F144. https://doi.org/10.1136/archdischild-2014-306174
Balayla J, Abenhaim HA (2014) Incidence, predictors and outcomes of congenital diaphragmatic hernia: a population-based study of 32 million births in the United States. J Matern Fetal Neonatal Med Off J Eur Assoc Perinat Med Fed Asia Ocean Perinat Soc Int Soc Perinat Obstet 27:1438–1444. https://doi.org/10.3109/14767058.2013.858691
Cauley RP, Potanos K, Fullington N, Bairdain S, Sheils CA, Finkelstein JA, Graham DA, Wilson JM (2015) Pulmonary support on day of life 30 is a strong predictor of increased 1 and 5-year morbidity in survivors of congenital diaphragmatic hernia. J Pediatr Surg 50:849–855. https://doi.org/10.1016/j.jpedsurg.2014.12.007
Russo FM, De Coppi P, Allegaert K, Toelen J, van der Veeken L, Attilakos G, Eastwood MP, David AL, Deprest J (2017) Current and future antenatal management of isolated congenital diaphragmatic hernia. Semin Fetal Neonatal Med 22:383–390. https://doi.org/10.1016/j.siny.2017.11.002
Chiu PPL (2014) New insights into congenital diaphragmatic hernia—a surgeon’s introduction to CDH animal models. Front Pediatr 2:36. https://doi.org/10.3389/fped.2014.00036
Kardon G, Ackerman KG, McCulley DJ, Shen Y, Wynn J, Shang L, Bogenschutz E, Sun X, Chung WK (2017) Congenital diaphragmatic hernias: from genes to mechanisms to therapies. Dis Model Mech 10:955–970. https://doi.org/10.1242/dmm.028365
Wynn J, Yu L, Chung WK (2014) Genetic causes of congenital diaphragmatic hernia. Semin Fetal Neonatal Med 19:324–330. https://doi.org/10.1016/j.siny.2014.09.003
Costlow RD, Manson JM (1981) The heart and diaphragm: target organs in the neonatal death induced by nitrofen (2,4-dichlorophenyl-p-nitrophenyl ether). Toxicology 20:209–227
Kluth D, Kangah R, Reich P, Tenbrinck R, Tibboel D, Lambrecht W (1990) Nitrofen-induced diaphragmatic hernias in rats: an animal model. J Pediatr Surg 25:850–854
Tenbrinck R, Tibboel D, Gaillard JL, Kluth D, Bos AP, Lachmann B, Molenaar JC (1990) Experimentally induced congenital diaphragmatic hernia in rats. J Pediatr Surg 25:426–429
Herriges M, Morrisey EE (2014) Lung development: orchestrating the generation and regeneration of a complex organ. Dev Camb Engl 141:502–513. https://doi.org/10.1242/dev.098186
Suen HC, Catlin EA, Ryan DP, Wain JC, Donahoe PK (1993) Biochemical immaturity of lungs in congenital diaphragmatic hernia. J Pediatr Surg 28:471–475 (discussion 476–477)
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol 62:1006–1012. https://doi.org/10.1016/j.jclinepi.2009.06.005
Chapin CJ, Ertsey R, Yoshizawa J, Hara A, Sbragia L, Greer JJ, Kitterman JA (2005) Congenital diaphragmatic hernia, tracheal occlusion, thyroid transcription factor-1, and fetal pulmonary epithelial maturation. Am J Physiol Lung Cell Mol Physiol 289:L44–L52. https://doi.org/10.1152/ajplung.00342.2004
Alfonso LF, Vilanova J, Aldazabal P, Lopez de Torre B, Tovar JA (1993) Lung growth and maturation in the rat model of experimentally induced congenital diaphragmatic hernia. Eur J Pediatr Surg Off J Austrian Assoc Pediatr Surg Al Z Kinderchir 3:6–11. https://doi.org/10.1055/s-2008-1063498
Lin H, Wang Y, Xiong Z, Tang Y, Liu W (2007) Effect of antenatal tetrandrine administration on endothelin-1 and epidermal growth factor levels in the lungs of rats with experimental diaphragmatic hernia. J Pediatr Surg 42:1644–1651. https://doi.org/10.1016/j.jpedsurg.2007.05.017
Utsuki T, Hashizume K, Iwamori M (2001) Impaired spreading of surfactant phospholipids in the lungs of newborn rats with pulmonary hypoplasia as a model of congenital diaphragmatic hernia induced by nitrofen. Biochim Biophys Acta 1531:90–98
Tovar JA, Alfonso LF, Aldazabal P, Lopez de Torre B, Uriarte S, Vilanova J (1992) The kidney in the fetal rat model of congenital diaphragmatic hernia induced by nitrofen. J Pediatr Surg 27:1356–1360
Alles AJ, Losty PD, Donahoe PK, Manganaro TF, Schnitzer JJ (1995) Embryonic cell death patterns associated with nitrofen-induced congenital diaphragmatic hernia. J Pediatr Surg 30:353–358 (discussion 359–360)
North AJ, Moya FR, Mysore MR, Thomas VL, Wells LB, Wu LC, Shaul PW (1995) Pulmonary endothelial nitric oxide synthase gene expression is decreased in a rat model of congenital diaphragmatic hernia. Am J Respir Cell Mol Biol 13:676–682. https://doi.org/10.1165/ajrcmb.13.6.7576705
Alfonso LF, Arnaiz A, Alvarez FJ, Qi B, Diez-Pardo JA, Vallis-i-Soler A, Tovar JA (1996) Lung hypoplasia and surfactant system immaturity induced in the fetal rat by prenatal exposure to nitrofen. Neonatology 69:94–100. https://doi.org/10.1159/000244283
Allan DW, Greer JJ (1997) Pathogenesis of nitrofen-induced congenital diaphragmatic hernia in fetal rats. J Appl Physiol Bethesda Md 83:338–347. https://doi.org/10.1152/jappl.1997.83.2.338
Xia H, Migliazza L, Diez-Pardo JA, Tovar JA (1999) The tracheobronchial tree is abnormal in experimental congenital diaphragmatic hernia. Pediatr Surg Int 15:184–187
Migliazza L, Xia H, Alvarez JI, Arnaiz A, Diez-Pardo JA, Alfonso LF, Tovar JA (1999) Heart hypoplasia in experimental congenital diaphragmatic hernia. J Pediatr Surg 34:706–710 (discussion 710–711)
Migliazza L, Xia H, Diez-Pardo JA, Tovar JA (1999) Skeletal malformations associated with congenital diaphragmatic hernia: experimental and human studies. J Pediatr Surg 34:1624–1629
Hoydu AK, Kitano Y, Kriss A, Hensley H, Bergey P, Flake A, Hubbard A, Leigh JS (2000) In vivo, in utero microscopic magnetic resonance imaging: application in a rat model of diaphragmatic hernia. Magn Reson Med 44:331–335
Migliazza L, Xia HM, Arnaiz A, Alvarez JI, Alfonso LF, Diez-Pardo JA, Valls i Soler A, Tovar JA (2000) Prenatal dexamethasone rescues heart hypoplasia in fetal rats with congenital diaphragmatic hernia. J Pediatr Surg 35:1757–1761
Yu J, Gonzalez S, Rodriguez JI, Diez-Pardo JA, Tovar JA (2001) Neural crest-derived defects in experimental congenital diaphragmatic hernia. Pediatr Surg Int 17:294–298. https://doi.org/10.1007/s003830100597
Yu J, Gonzalez S, Diez-Pardo JA, Tovar JA (2002) Effects of vitamin A on malformations of neural-crest-controlled organs induced by nitrofen in rats. Pediatr Surg Int 18:600–605. https://doi.org/10.1007/s00383-002-0865-5
Correia-Pinto J, Baptista MJ, Pedrosa C, Estevão-Costa J, Flake AW, Leite-Moreira AF (2003) Fetal heart development in the nitrofen-induced CDH rat model: the role of mechanical and nonmechanical factors. J Pediatr Surg 38:1444–1451. https://doi.org/10.1016/S0022-3468(03)00494-9
Rodriguez-Matas MJ, Gonzalez-Reyes S, Martínez L, Martínez I, Rodriguez JI, Diez-Pardo JA, Tovar JA (2003) The adrenal cortex in experimental congenital diaphragmatic hernia. J Pediatr Surg 38:682–684. https://doi.org/10.1016/jpsu.2003.50182
Gonzalez-Reyes S, Alvarez L, Diez-Pardo JA, Tovar JA (2003) Prenatal vitamin E improves lung and heart hypoplasia in experimental diaphargamatic hernia. Pediatr Surg Int 19:331–334. https://doi.org/10.1007/s00383-003-1005-6
Martínez L, González-Reyes S, Burgos E, Tovar JA (2004) The vagus and recurrent laryngeal nerves in experimental congenital diaphragmatic hernia. Pediatr Surg Int 20:253–257. https://doi.org/10.1007/s00383-003-1121-3
Baptista MJ, Melo-Rocha G, Pedrosa C, Gonzaga S, Teles A, Estevão-Costa J, Areias JC, Flake AW, Leite-Moreira AF, Correia-Pinto J (2005) Antenatal vitamin A administration attenuates lung hypoplasia by interfering with early instead of late determinants of lung underdevelopment in congenital diaphragmatic hernia. J Pediatr Surg 40:658–665. https://doi.org/10.1016/j.jpedsurg.2005.01.034
Oshiro T, Asato Y, Sakanashi M, Ohta T, Sugahara K (2005) Differential effects of vitamin A on fetal lung growth and diaphragmatic formation in nitrofen-induced rat model. Pulm Pharmacol Ther 18:155–164. https://doi.org/10.1016/j.pupt.2004.11.004
Folkesson HG, Chapin CJ, Beard LL, Ertsey R, Matthay MA, Kitterman JA (2006) Congenital diaphragmatic hernia prevents absorption of distal air space fluid in late-gestation rat fetuses. Am J Physiol Lung Cell Mol Physiol 290:L478–L484. https://doi.org/10.1152/ajplung.00124.2005
Montedonico S, Nakazawa N, Shinkai T, Bannigan J, Puri P (2007) Kidney development in the nitrofen-induced pulmonary hypoplasia and congenital diaphragmatic hernia in rats. J Pediatr Surg 42:239–243. https://doi.org/10.1016/j.jpedsurg.2006.09.062
Baird R, Khan N, Flageole H, Anselmo M, Puligandla P, Laberge J-M (2008) The effect of tracheal occlusion on lung branching in the rat nitrofen CDH model. J Surg Res 148:224–229. https://doi.org/10.1016/j.jss.2007.07.019
Sakai K, Kimura O, Furukawa T, Fumino S, Higuchi K, Wakao J, Kimura K, Aoi S, Masumoto K, Tajiri T (2014) Prenatal administration of neuropeptide bombesin promotes lung development in a rat model of nitrofen-induced congenital diaphragmatic hernia. J Pediatr Surg 49:1749–1752. https://doi.org/10.1016/j.jpedsurg.2014.09.015
Tsuda H, Kotani T, Nakano T, Imai K, Hirako S, Li H, Kikkawa F (2016) Lipocalin 2 as a new biomarker for fetal lung hypoplasia in congenital diaphragmatic hernia. Clin Chim Acta Int J Clin Chem 462:71–76. https://doi.org/10.1016/j.cca.2016.08.023
Zhu S, He Q, Zhang R, Wang Y, Zhong W, Xia H, Yu J (2016) Decreased expression of miR-33 in fetal lungs of nitrofen-induced congenital diaphragmatic hernia rat model. J Pediatr Surg 51:1096–1100. https://doi.org/10.1016/j.jpedsurg.2016.02.083
Teramoto H, Yoneda A, Puri P (2003) Gene expression of fibroblast growth factors 10 and 7 is downregulated in the lung of nitrofen-induced diaphragmatic hernia in rats. J Pediatr Surg 38:1021–1024
Candilera V, Bouchè C, Schleef J, Pederiva F (2016) Lung growth factors in the amniotic fluid of normal pregnancies and with congenital diaphragmatic hernia. J Matern Fetal Neonatal Med 29(13):2104–2108. https://doi.org/10.3109/14767058.2015.1076387
Oue T, Shima H, Taira Y, Puri P (2000) Administration of antenatal glucocorticoids upregulates peptide growth factor gene expression in nitrofen-induced congenital diaphragmatic hernia in rats. J Pediatr Surg 35:109–112
Boucherat O, Benachi A, Chailley-Heu B, Franco-Montoya M-L, Elie C, Martinovic J, Bourbon JR (2007) Surfactant maturation is not delayed in human fetuses with diaphragmatic hernia. PLoS Med 4:e237. https://doi.org/10.1371/journal.pmed.0040237
Takahashi H, Friedmacher F, Fujiwara N, Hofmann A, Puri P (2014) Pulmonary FGF9 gene expression is downregulated during the pseudoglandular stage in nitrofen-induced hypoplastic lungs. Eur J Pediatr Surg Off J Austrian Assoc Pediatr Surg Al Z Kinderchir 24:75–78. https://doi.org/10.1055/s-0033-1351392
Takahashi H, Friedmacher F, Fujiwara N, Hofmann A, Kutasy B, Gosemann J-H, Puri P (2013) Pulmonary FGF-18 gene expression is downregulated during the canalicular-saccular stages in nitrofen-induced hypoplastic lungs. Pediatr Surg Int 29:1199–1203. https://doi.org/10.1007/s00383-013-3387-4
Boucherat O, Benachi A, Barlier-Mur A-M, Franco-Montoya M-L, Martinovic J, Thébaud B, Chailley-Heu B, Bourbon JR (2007) Decreased lung fibroblast growth factor 18 and elastin in human congenital diaphragmatic hernia and animal models. Am J Respir Crit Care Med 175:1066–1077. https://doi.org/10.1164/rccm.200601-050OC
Burgos CM, Uggla AR, Fagerström-Billai F, Eklöf A-C, Frenckner B, Nord M (2010) Gene expression analysis in hypoplastic lungs in the nitrofen model of congenital diaphragmatic hernia. J Pediatr Surg 45:1445–1454. https://doi.org/10.1016/j.jpedsurg.2009.09.023
Friedmacher F, Doi T, Gosemann J-H, Fujiwara N, Kutasy B, Puri P (2012) Upregulation of fibroblast growth factor receptor 2 and 3 in the late stages of fetal lung development in the nitrofen rat model. Pediatr Surg Int 28:195–199. https://doi.org/10.1007/s00383-011-2985-2
Friedmacher F, Gosemann J-H, Takahashi H, Corcionivoschi N, Puri P (2013) Decreased pulmonary c-Cbl expression and tyrosine phosphorylation in the nitrofen-induced rat model of congenital diaphragmatic hernia. Pediatr Surg Int 29:19–24. https://doi.org/10.1007/s00383-012-3191-6
Friedmacher F, Gosemann J-H, Fujiwara N, Alvarez LAJ, Corcionivoschi N, Puri P (2013) Spatiotemporal alterations in Sprouty-2 expression and tyrosine phosphorylation in nitrofen-induced pulmonary hypoplasia. J Pediatr Surg 48:2219–2225. https://doi.org/10.1016/j.jpedsurg.2013.07.003
Friedmacher F, Gosemann J-H, Fujiwara N, Takahashi H, Hofmann A, Puri P (2013) Expression of Sproutys and SPREDs is decreased during lung branching morphogenesis in nitrofen-induced pulmonary hypoplasia. Pediatr Surg Int 29:1193–1198. https://doi.org/10.1007/s00383-013-3385-6
Thompson SM, Connell MG, van Kuppevelt TH, Xu R, Turnbull JE, Losty PD, Fernig DG, Jesudason EC (2011) Structure and epitope distribution of heparan sulfate is disrupted in experimental lung hypoplasia: a glycobiological epigenetic cause for malformation? BMC Dev Biol 11:38. https://doi.org/10.1186/1471-213X-11-38
Kling DE, Narra V, Islam S, Kinane TB, Alessandrini A, Ercolani L, Donahoe PK, Schnitzer JJ (2001) Decreased mitogen activated protein kinase activities in congenital diaphragmatic hernia-associated pulmonary hypoplasia. J Pediatr Surg 36:1490–1496
Doi T, Sugimoto K, Ruttenstock E, Dingemann J, Puri P (2010) Prenatal retinoic acid upregulates pulmonary gene expression of PI3K and AKT in nitrofen-induced pulmonary hypoplasia. Pediatr Surg Int 26:1011–1015. https://doi.org/10.1007/s00383-010-2654-x
Takahashi T, Friedmacher F, Takahashi H, Daniel Hofmann A, Puri P (2015) Lysyl oxidase expression is decreased in the developing diaphragm and lungs of nitrofen-induced congenital diaphragmatic hernia. Eur J Pediatr Surg Off J Austrian Assoc Pediatr Surg Al Z Kinderchir 25:15–19. https://doi.org/10.1055/s-0034-1386644
Takayasu H, Nakazawa N, Montedonico S, Puri P (2007) Down-regulation of Wnt signal pathway in nitrofen-induced hypoplastic lung. J Pediatr Surg 42:426–430. https://doi.org/10.1016/j.jpedsurg.2006.10.018
Makanga M, Dewachter C, Maruyama H, Vuckovic A, Rondelet B, Naeije R, Dewachter L (2013) Downregulated bone morphogenetic protein signaling in nitrofen-induced congenital diaphragmatic hernia. Pediatr Surg Int 29:823–834. https://doi.org/10.1007/s00383-013-3340-6
Gosemann J-H, Friedmacher F, Fujiwara N, Alvarez LAJ, Corcionivoschi N, Puri P (2013) Disruption of the bone morphogenetic protein receptor 2 pathway in nitrofen-induced congenital diaphragmatic hernia. Birth Defects Res B Dev Reprod Toxicol 98:304–309. https://doi.org/10.1002/bdrb.21065
Takahashi T, Zimmer J, Friedmacher F, Puri P (2017) Follistatin-like 1 expression is decreased in the alveolar epithelium of hypoplastic rat lungs with nitrofen-induced congenital diaphragmatic hernia. J Pediatr Surg 52:706–709. https://doi.org/10.1016/j.jpedsurg.2017.01.020
Fujiwara N, Doi T, Gosemann J-H, Kutasy B, Friedmacher F, Puri P (2012) Smad1 and WIF1 genes are downregulated during saccular stage of lung development in the nitrofen rat model. Pediatr Surg Int 28:189–193. https://doi.org/10.1007/s00383-011-2987-0
Takahashi T, Friedmacher F, Zimmer J, Puri P (2017) Expression of T-box transcription factors 2, 4 and 5 is decreased in the branching airway mesenchyme of nitrofen-induced hypoplastic lungs. Pediatr Surg Int 33:139–143. https://doi.org/10.1007/s00383-016-4005-z
Unger S, Copland I, Tibboel D, Post M (2003) Down-regulation of sonic hedgehog expression in pulmonary hypoplasia is associated with congenital diaphragmatic hernia. Am J Pathol 162:547–555. https://doi.org/10.1016/S0002-9440(10)63848-5
Takahashi T, Friedmacher F, Takahashi H, Hofmann AD, Puri P (2015) Kif7 expression is decreased in the diaphragmatic and pulmonary mesenchyme of nitrofen-induced congenital diaphragmatic hernia. J Pediatr Surg 50:904–907. https://doi.org/10.1016/j.jpedsurg.2015.03.058
Zimmer J, Takahashi T, Hofmann AD, Puri P (2016) Downregulation of Forkhead box F1 gene expression in the pulmonary vasculature of nitrofen-induced congenital diaphragmatic hernia. Pediatr Surg Int 32:1121–1126. https://doi.org/10.1007/s00383-016-3967-1
Doi T, Puri P (2009) Up-regulation of Wnt5a gene expression in the nitrofen-induced hypoplastic lung. J Pediatr Surg 44:2302–2306. https://doi.org/10.1016/j.jpedsurg.2009.07.069
Takahashi T, Friedmacher F, Zimmer J, Puri P (2018) Gata-6 expression is decreased in diaphragmatic and pulmonary mesenchyme of fetal rats with nitrofen-induced congenital diaphragmatic hernia. Pediatr Surg Int 34:315–321. https://doi.org/10.1007/s00383-017-4219-8
Nakazawa N, Montedonico S, Takayasu H, Paradisi F, Puri P (2007) Disturbance of retinol transportation causes nitrofen-induced hypoplastic lung. J Pediatr Surg 42:345–349. https://doi.org/10.1016/j.jpedsurg.2006.10.028
Major D, Cadenas M, Fournier L, Leclerc S, Lefebvre M, Cloutier R (1998) Retinol status of newborn infants with congenital diaphragmatic hernia. Pediatr Surg Int 13:547–549. https://doi.org/10.1007/s003830050399
Beurskens LWJE, Tibboel D, Lindemans J, Duvekot JJ, Cohen-Overbeek TE, Veenma DCM, de Klein A, Greer JJ, Steegers-Theunissen RPM (2010) Retinol status of newborn infants is associated with congenital diaphragmatic hernia. Pediatrics 126:712–720. https://doi.org/10.1542/peds.2010-0521
Kutasy B, Friedmacher F, Duess JW, Puri P (2014) Prenatal administration of retinoic acid increases the trophoblastic insulin-like growth factor 2 protein expression in the nitrofen model of congenital diaphragmatic hernia. Pediatr Surg Int 30:137–142. https://doi.org/10.1007/s00383-013-3449-7
Coste K, Beurskens LWJE, Blanc P, Gallot D, Delabaere A, Blanchon L, Tibboel D, Labbé A, Rottier RJ, Sapin V (2015) Metabolic disturbances of the vitamin A pathway in human diaphragmatic hernia. Am J Physiol Lung Cell Mol Physiol 308:L147–L157. https://doi.org/10.1152/ajplung.00108.2014
Pereira-Terra P, Moura RS, Nogueira-Silva C, Correia-Pinto J (2015) Neuroendocrine factors regulate retinoic acid receptors in normal and hypoplastic lung development. J Physiol 593:3301–3311. https://doi.org/10.1113/JP270477
Nakazawa N, Takayasu H, Montedonico S, Puri P (2007) Altered regulation of retinoic acid synthesis in nitrofen-induced hypoplastic lung. Pediatr Surg Int 23:391–396. https://doi.org/10.1007/s00383-006-1848-8
Kutasy B, Friedmacher F, Pes L, Paradisi F, Puri P (2014) Increased uptake of dietary retinoids at the maternal-fetal barrier in the nitrofen model of congenital diaphragmatic hernia. J Pediatr Surg 49:866–870. https://doi.org/10.1016/j.jpedsurg.2014.01.014
Doi T, Sugimoto K, Puri P (2009) Up-regulation of COUP-TFII gene expression in the nitrofen-induced hypoplastic lung. J Pediatr Surg 44:321–324. https://doi.org/10.1016/j.jpedsurg.2008.10.079
Doi T, Shintaku M, Dingemann J, Ruttenstock E, Puri P (2011) Downregulation of Midkine gene expression and its response to retinoic acid treatment in the nitrofen-induced hypoplastic lung. Pediatr Surg Int 27:199–204. https://doi.org/10.1007/s00383-010-2773-4
Ruttenstock EM, Doi T, Dingemann J, Puri P (2011) Prenatal retinoic acid treatment upregulates late gestation lung protein 1 in the nitrofen-induced hypoplastic lung in late gestation. Pediatr Surg Int 27:125–129. https://doi.org/10.1007/s00383-010-2783-2
Oue T, Taira Y, Shima H, Miyazaki E, Puri P (1999) Effect of antenatal glucocorticoid administration on insulin-like growth factor I and II levels in hypoplastic lung in nitrofen-induced congenital diaphragmatic hernia in rats. Pediatr Surg Int 15:175–179
Miyazaki E, Ohshiro K, Taira Y, Puri P (1998) Altered insulin-like growth factor I mRNA expression in human hypoplastic lung in congenital diaphragmatic hernia. J Pediatr Surg 33:1476–1479
Ruttenstock E, Doi T, Dingemann J, Puri P (2010) Insulinlike growth factor receptor type 1 and type 2 are downregulated in the nitrofen-induced hypoplastic lung. J Pediatr Surg 45:1349–1353. https://doi.org/10.1016/j.jpedsurg.2010.02.111
Ruttenstock E, Doi T, Dingemann J, Puri P (2010) Insulin receptor is downregulated in the nitrofen-induced hypoplastic lung. J Pediatr Surg 45:948–952. https://doi.org/10.1016/j.jpedsurg.2010.02.018
Ruttenstock E, Doi T, Dingemann J, Puri P (2010) Downregulation of insulin-like growth factor binding protein 3 and 5 in nitrofen-induced pulmonary hypoplasia. Pediatr Surg Int 26:59–63. https://doi.org/10.1007/s00383-009-2509-5
Ruttenstock EM, Doi T, Dingemann J, Puri P (2011) IGFBP-4 gene overexpression in the nitrofen-induced hypoplastic lung. Eur J Pediatr Surg Off J Austrian Assoc Pediatr Surg Al Z Kinderchir 21:42–45. https://doi.org/10.1055/s-0030-1262851
Mižíková I, Palumbo F, Tábi T, Herold S, Vadász I, Mayer K, Seeger W, Morty RE (2017) Perturbations to lysyl oxidase expression broadly influence the transcriptome of lung fibroblasts. Physiol Genom 49:416–429. https://doi.org/10.1152/physiolgenomics.00026.2017
Chailley-Heu B, Boucherat O, Barlier-Mur A-M, Bourbon JR (2005) FGF-18 is upregulated in the postnatal rat lung and enhances elastogenesis in myofibroblasts. Am J Physiol Lung Cell Mol Physiol 288:L43–L51. https://doi.org/10.1152/ajplung.00096.2004
Kugler MC, Joyner AL, Loomis CA, Munger JS (2015) Sonic hedgehog signaling in the lung. From development to disease. Am J Respir Cell Mol Biol 52:1–13. https://doi.org/10.1165/rcmb.2014-0132TR
Ho UY, Wainwright BJ (2017) Patched1 patterns fibroblast growth factor 10 and Forkhead box F1 expression during pulmonary branch formation. Mech Dev 147:37–48. https://doi.org/10.1016/j.mod.2017.09.001
Weidenfeld J, Shu W, Zhang L, Millar SE, Morrisey EE (2002) The WNT7b promoter is regulated by TTF-1, GATA6, and Foxa2 in lung epithelium. J Biol Chem 277:21061–21070. https://doi.org/10.1074/jbc.M111702200
Keijzer R, van Tuyl M, Meijers C, Post M, Tibboel D, Grosveld F, Koutsourakis M (2001) The transcription factor GATA6 is essential for branching morphogenesis and epithelial cell differentiation during fetal pulmonary development. Dev Camb Engl 128:503–511
Wilson JG, Roth CB, Warkany J (1953) An analysis of the syndrome of malformations induced by maternal vitamin A deficiency. Effects of restoration of vitamin A at various times during gestation. Am J Anat 92:189–217. https://doi.org/10.1002/aja.1000920202
Thébaud B, Tibboel D, Rambaud C, Mercier JC, Bourbon JR, Dinh-Xuan AT, Archer SL (1999) Vitamin A decreases the incidence and severity of nitrofen-induced congenital diaphragmatic hernia in rats. Am J Physiol 277:L423–L429
Babiuk RP, Thébaud B, Greer JJ (2004) Reductions in the incidence of nitrofen-induced diaphragmatic hernia by vitamin A and retinoic acid. Am J Physiol Lung Cell Mol Physiol 286:L970–L973. https://doi.org/10.1152/ajplung.00403.2003
Takahashi YI, Smith JE, Goodman DS (1977) Vitamin A and retinol-binding protein metabolism during fetal development in the rat. Am J Physiol Endocrinol Metab 233:E263. https://doi.org/10.1152/ajpendo.1977.233.4.E263
Greer JJ, Babiuk RP, Thebaud B (2003) Etiology of congenital diaphragmatic hernia: the retinoid hypothesis. Pediatr Res 53:726–730. https://doi.org/10.1203/01.PDR.0000062660.12769.E6
Mey J, Babiuk RP, Clugston R, Zhang W, Greer JJ (2003) Retinal dehydrogenase-2 is inhibited by compounds that induce congenital diaphragmatic hernias in rodents. Am J Pathol 162:673–679. https://doi.org/10.1016/S0002-9440(10)63861-8
Manson JM (1986) Mechanism of nitrofen teratogenesis. Environ Health Perspect 70:137–147
Toriyama K, Muramatsu H, Hoshino T, Torii S, Muramatsu T (1997) Evaluation of heparin-binding growth factors in rescuing morphogenesis of heparitinase-treated mouse embryonic lung explants. Differ Res Biol Divers 61:161–167. https://doi.org/10.1046/j.1432-0436.1997.6130161.x
Zhang H, Garber SJ, Cui Z, Foley JP, Mohan GS, Jobanputra M, Kaplan F, Sweezey NB, Gonzales LW, Savani RC (2009) The angiogenic factor midkine is regulated by dexamethasone and retinoic acid during alveolarization and in alveolar epithelial cells. Respir Res 10:77. https://doi.org/10.1186/1465-9921-10-77
Masumoto K, Teshiba R, Esumi G, Nagata K, Takahata Y, Hikino S, Hara T, Hojo S, Tsukimori K, Wake N, Kinukawa N, Taguchi T (2009) Improvement in the outcome of patients with antenatally diagnosed congenital diaphragmatic hernia using gentle ventilation and circulatory stabilization. Pediatr Surg Int 25:487–492. https://doi.org/10.1007/s00383-009-2370-6
Ruttenstock E, Doi T, Dingemann J, Puri P (2011) Prenatal administration of retinoic acid upregulates insulin-like growth factor receptors in the nitrofen-induced hypoplastic lung. Birth Defects Res B Dev Reprod Toxicol 92:148–151. https://doi.org/10.1002/bdrb.20293
Price WA (1999) Peptide growth factors regulate insulin-like growth factor binding protein production by fetal rat lung fibroblasts. Am J Respir Cell Mol Biol 20:332–341. https://doi.org/10.1165/ajrcmb.20.2.3304
Santos M, Nogueira-Silva C, Baptista MJ, Soares-Fernandes J, Moura RS, Correia-Pinto J (2007) Pulmonary epithelial cell differentiation in the nitrofen-induced congenital diaphragmatic hernia. J Pediatr Surg 42:1231–1237. https://doi.org/10.1016/j.jpedsurg.2007.02.014
IJsselstijn H, Perrin DG, de Jongste JC, Cutz E, Tibboel D (1995) Pulmonary neuroendocrine cells in neonatal rats with congenital diaphragmatic hernia. J Pediatr Surg 30:413–415
Yamataka T, Puri P (1996) Increased intracellular levels of calcitonin gene-related peptide-like immunoreactivity in pulmonary endocrine cells in an experimental model of congenital diaphragmatic hernia. Pediatr Surg Int 11:448–452. https://doi.org/10.1007/BF00180080
IJsselstijn H, Hung N, de Jongste JC, Tibboel D, Cutz E (1998) Calcitonin gene-related peptide expression is altered in pulmonary neuroendocrine cells in developing lungs of rats with congenital diaphragmatic hernia. Am J Respir Cell Mol Biol 19:278–285. https://doi.org/10.1165/ajrcmb.19.2.2853
Gosney JR, Okoye BO, Lloyd DA, Losty PD (1999) Pulmonary neuroendocrine cells in nitrofen-induced diaphragmatic hernia and the effect of prenatal glucocorticoids. Pediatr Surg Int 15:180–183
Santos M, Bastos P, Gonzaga S, Roriz J-M, Baptista MJ, Nogueira-Silva C, Melo-Rocha G, Henriques-Coelho T, Roncon-Albuquerque R, Leite-Moreira AF, De Krijger RR, Tibboel D, Rottier R, Correia-Pinto J (2006) Ghrelin expression in human and rat fetal lungs and the effect of ghrelin administration in nitrofen-induced congenital diaphragmatic hernia. Pediatr Res 59:531–537. https://doi.org/10.1203/01.pdr.0000202748.66359.a9
Asabe K, Tsuji K, Handa N, Kajiwara M, Suita S (1999) Immunohistochemical distribution of bombesin-positive pulmonary neuroendocrine cells in a congenital diaphragmatic hernia. Surg Today 29:407–412. https://doi.org/10.1007/BF02483031
Ijsselstijn H, Gaillard JL, de Jongste JC, Tibboel D, Cutz E (1997) Abnormal expression of pulmonary bombesin-like peptide immunostaining cells in infants with congenital diaphragmatic hernia. Pediatr Res 42:715–720. https://doi.org/10.1203/00006450-199711000-00026
Takayasu H, Nakazawa N, Montedonico S, Puri P (2007) Reduced expression of aquaporin 5 water channel in nitrofen-induced hypoplastic lung with congenital diaphragmatic hernia rat model. J Pediatr Surg 42:415–419. https://doi.org/10.1016/j.jpedsurg.2006.10.029
Takayasu H, Nakazawa N, Montedonico S, Sugimoto K, Sato H, Puri P (2007) Impaired alveolar epithelial cell differentiation in the hypoplastic lung in nitrofen-induced congenital diaphragmatic hernia. Pediatr Surg Int 23:405–410. https://doi.org/10.1007/s00383-006-1853-y
Sugimoto K, Takayasu H, Nakazawa N, Montedonico S, Puri P (2008) Prenatal treatment with retinoic acid accelerates type 1 alveolar cell proliferation of the hypoplastic lung in the nitrofen model of congenital diaphragmatic hernia. J Pediatr Surg 43:367–372. https://doi.org/10.1016/j.jpedsurg.2007.10.050
Tovar JA, Qi B, Diez-Pardo JA, Alfonso LF, Arnaiz A, Alvarez FJ, Valls-i-Soler A, Morreale de Escobar G (1997) Thyroid hormones in the pathogenesis of lung hypoplasia and immaturity induced in fetal rats by prenatal exposure to nitrofen. J Pediatr Surg 32:1295–1297
Teramoto H, Guarino N, Puri P (2001) Altered gene level expression of thyroid hormone receptors alpha-1 and beta-1 in the lung of nitrofen-induced diaphragmatic hernia. J Pediatr Surg 36:1675–1678. https://doi.org/10.1053/jpsu.2001.27958
Lukošiūtė A, Doi T, Dingemann J, Ruttenstock EM, Puri P (2011) Down-regulation of lung Kruppel-like factor in the nitrofen-induced hypoplastic lung. Eur J Pediatr Surg Off J Austrian Assoc Pediatr Surg Al Z Kinderchir 21:38–41. https://doi.org/10.1055/s-0030-1262800
Ruttenstock EM, Doi T, Dingemann J, Puri P (2012) Prenatal retinoic acid upregulates connexin 43 (Cx43) gene expression in pulmonary hypoplasia in the nitrofen-induced congenital diaphragmatic hernia rat model. J Pediatr Surg 47:336–340. https://doi.org/10.1016/j.jpedsurg.2011.11.026
Ringman A, Zelenina M, Eklöf A-C, Aperia A, Frenckner B (2008) NKCC-1 and ENaC are down-regulated in nitrofen-induced hypoplastic lungs with congenital diaphragmatic hernia. Pediatr Surg Int 24:993–1000. https://doi.org/10.1007/s00383-008-2209-6
Losada A, Tovar JA, Xia HM, Diez-Pardo JA, Santisteban P (2000) Down-regulation of thyroid transcription factor-1 gene expression in fetal lung hypoplasia is restored by glucocorticoids. Endocrinology 141:2166–2173. https://doi.org/10.1210/endo.141.6.7522
Losada A, Xia H, Migliazza L, Diez-Pardo JA, Santisteban P, Tovar JA (1999) Lung hypoplasia caused by nitrofen is mediated by down-regulation of thyroid transcription factor TTF-1. Pediatr Surg Int 15:188–191
Gonzalez-Reyes S, Martinez L, Martinez-Calonge W, Fernandez-Dumont V, Tovar JA (2006) Effects of antioxidant vitamins on molecular regulators involved in lung hypoplasia induced by nitrofen. J Pediatr Surg 41:1446–1452. https://doi.org/10.1016/j.jpedsurg.2006.04.022
Hösgör M, Ijzendoorn Y, Mooi WJ, Tibboel D, De Krijger RR (2002) Thyroid transcription factor-1 expression during normal human lung development and in patients with congenital diaphragmatic hernia. J Pediatr Surg 37:1258–1262
Mysore MR, Margraf LR, Jaramillo MA, Breed DR, Chau VL, Arévalo M, Moya FR (1998) Surfactant protein A is decreased in a rat model of congenital diaphragmatic hernia. Am J Respir Crit Care Med 157:654–657. https://doi.org/10.1164/ajrccm.157.2.9612072
Shima H, Guarino N, Puri P (2000) Effect of hyperoxia on surfactant protein gene expression in hypoplastic lung in nitrofen-induced diaphragmatic hernia in rats. Pediatr Surg Int 16:473–477. https://doi.org/10.1007/s003830000427
Van Tuyl M, Blommaart P, Keijzer E, Wert R, Ruijter SE, Lamers JM, Tibboel WH D (2003) Pulmonary surfactant protein A, B, and C mRNA and protein expression in the nitrofen-induced congenital diaphragmatic hernia rat model. Pediatr Res 54:641–652. https://doi.org/10.1203/01.PDR.0000086906.19683.42
Guilbert TW, Gebb SA, Shannon JM (2000) Lung hypoplasia in the nitrofen model of congenital diaphragmatic hernia occurs early in development. Am J Physiol Lung Cell Mol Physiol 279:L1159–L1171. https://doi.org/10.1152/ajplung.2000.279.6.L1159
Fox ZD, Jiang G, Ho KKY, Walker KA, Liu AP, Kunisaki SM (2018) Fetal lung transcriptome patterns in an ex vivo compression model of diaphragmatic hernia. J Surg Res 231:411–420. https://doi.org/10.1016/j.jss.2018.06.064
Guarino N, Oue T, Shima H, Puri P (2000) Antenatal dexamethasone enhances surfactant protein synthesis in the hypoplastic lung of nitrofen-induced diaphragmatic hernia in rats. J Pediatr Surg 35:1468–1473. https://doi.org/10.1053/jpsu.2000.16416
Cogo PE, Simonato M, Danhaive O, Verlato G, Cobellis G, Savignoni F, Peca D, Baritussio A, Carnielli VP (2013) Impaired surfactant protein B synthesis in infants with congenital diaphragmatic hernia. Eur Respir J 41:677–682. https://doi.org/10.1183/09031936.00032212
Thébaud B, Barlier-Mur AM, Chailley-Heu B, Henrion-Caude A, Tibboel D, Dinh-Xuan AT, Bourbon JR (2001) Restoring effects of vitamin A on surfactant synthesis in nitrofen-induced congenital diaphragmatic hernia in rats. Am J Respir Crit Care Med 164:1083–1089. https://doi.org/10.1164/ajrccm.164.6.2010115
Janssen DJ, Zimmermann LJ, Cogo P, Hamvas A, Bohlin K, Luijendijk IH, Wattimena D, Carnielli VP, Tibboel D (2009) Decreased surfactant phosphatidylcholine synthesis in neonates with congenital diaphragmatic hernia during extracorporeal membrane oxygenation. Intensive Care Med 35:1754–1760. https://doi.org/10.1007/s00134-009-1564-7
IJsselstijn H, Zimmermann LJ, Bunt JE, de Jongste JC, Tibboel D (1998) Prospective evaluation of surfactant composition in bronchoalveolar lavage fluid of infants with congenital diaphragmatic hernia and of age-matched controls. Crit Care Med 26:573–580
Shima H, Ohshiro K, Taira Y, Miyazaki E, Oue T, Puri P (1999) Antenatal dexamethasone suppresses tumor necrosis factor-alpha expression in hypoplastic lung in nitrofen-induced diaphragmatic hernia in rats. Pediatr Res 46:633–637. https://doi.org/10.1203/00006450-199911000-00023
Ohshiro K, Miyazaki E, Taira Y, Puri P (1998) Upregulated tumor necrosis factor-alpha gene expression in the hypoplastic lung in patients with congenital diaphragmatic hernia. Pediatr Surg Int 14:21–24
Schaible T, Veit M, Tautz J, Kehl S, Büsing K, Monz D, Gortner L, Tutdibi E (2011) Serum cytokine levels in neonates with congenital diaphragmatic hernia. Klin Padiatr 223:414–418. https://doi.org/10.1055/s-0031-1295436
Guarino N, Solari V, Shima H, Puri P (2004) Upregulated expression of EGF and TGF-alpha in the proximal respiratory epithelium in the human hypoplastic lung in congenital diaphragmatic hernia. Pediatr Surg Int 19:755–759. https://doi.org/10.1007/s00383-003-1052-z
Kakiashvili E, Dan Q, Vandermeer M, Zhang Y, Waheed F, Pham M, Szászi K (2011) The epidermal growth factor receptor mediates tumor necrosis factor-alpha-induced activation of the ERK/GEF-H1/RhoA pathway in tubular epithelium. J Biol Chem 286:9268–9279. https://doi.org/10.1074/jbc.M110.179903
Wang Y, Huang Z, Nayak PS, Matthews BD, Warburton D, Shi W, Sanchez-Esteban J (2013) Strain-induced differentiation of fetal type II epithelial cells is mediated via the integrin α6β1-ADAM17/tumor necrosis factor-α-converting enzyme (TACE) signaling pathway. J Biol Chem 288:25646–25657. https://doi.org/10.1074/jbc.M113.473777
Towne JE, Krane CM, Bachurski CJ, Menon AG (2001) Tumor necrosis factor-alpha inhibits aquaporin 5 expression in mouse lung epithelial cells. J Biol Chem 276:18657–18664. https://doi.org/10.1074/jbc.M100322200
Candilera V, Bouchè C, Schleef J, Pederiva F (2016) Lung growth factors in the amniotic fluid of normal pregnancies and with congenital diaphragmatic hernia. J Matern Fetal Neonatal Med Off J Eur Assoc Perinat Med Fed Asia Ocean Perinat Soc Int Soc Perinat Obstet 29:2104–2108. https://doi.org/10.3109/14767058.2015.1076387
Fleck S, Bautista G, Keating SM, Lee T-H, Keller RL, Moon-Grady AJ, Gonzales K, Norris PJ, Busch MP, Kim CJ, Romero R, Lee H, Miniati D, MacKenzie TC (2013) Fetal production of growth factors and inflammatory mediators predicts pulmonary hypertension in congenital diaphragmatic hernia. Pediatr Res 74:290–298. https://doi.org/10.1038/pr.2013.98
Friedmacher F, Hofmann AD, Takahashi H, Takahashi T, Gosemann J-H, Puri P (2014) Disruption of THY-1 signaling in alveolar lipofibroblasts in experimentally induced congenital diaphragmatic hernia. Pediatr Surg Int 30:129–135. https://doi.org/10.1007/s00383-013-3444-z
Friedmacher F, Fujiwara N, Hofmann AD, Takahashi H, Alvarez LAJ, Gosemann J-H, Puri P (2014) Prenatal retinoic acid increases lipofibroblast expression in hypoplastic rat lungs with experimental congenital diaphragmatic hernia. J Pediatr Surg 49:876–881. https://doi.org/10.1016/j.jpedsurg.2014.01.017 (discussion 881)
Gosemann J-H, Doi T, Kutasy B, Friedmacher F, Dingemann J, Puri P (2012) Alterations of peroxisome proliferator-activated receptor γ and monocyte chemoattractant protein 1 gene expression in the nitrofen-induced hypoplastic lung. J Pediatr Surg 47:847–851. https://doi.org/10.1016/j.jpedsurg.2012.01.038
Doi T, Lukosiūte A, Ruttenstock E, Dingemann J, Puri P (2010) Disturbance of parathyroid hormone-related protein signaling in the nitrofen-induced hypoplastic lung. Pediatr Surg Int 26:45–50. https://doi.org/10.1007/s00383-009-2506-8
Carroll JL, McCoy DM, McGowan SE, Salome RG, Ryan AJ, Mallampalli RK (2002) Pulmonary-specific expression of tumor necrosis factor-alpha alters surfactant lipid metabolism. Am J Physiol Lung Cell Mol Physiol 282:L735–L742. https://doi.org/10.1152/ajplung.00120.2001
Okawada M, Kobayashi H, Tei E, Okazaki T, Lane GJ, Yamataka A (2007) Serum monocyte chemotactic protein-1 levels in congenital diaphragmatic hernia. Pediatr Surg Int 23:487–491. https://doi.org/10.1007/s00383-006-1858-6
Khoshgoo N, Kholdebarin R, Pereira-Terra P, Mahood TH, Falk L, Day CA, Iwasiow BM, Zhu F, Mulhall D, Fraser C, Correia-Pinto J, Keijzer R (2017) Prenatal microRNA miR-200b therapy improves nitrofen-induced pulmonary hypoplasia associated with congenital diaphragmatic hernia. Ann Surg. https://doi.org/10.1097/SLA.0000000000002595
Santos M, Moura RS, Gonzaga S, Nogueira-Silva C, Ohlmeier S, Correia-Pinto J (2007) Embryonic essential myosin light chain regulates fetal lung development in rats. Am J Respir Cell Mol Biol 37:330–338. https://doi.org/10.1165/rcmb.2006-0349OC
Chen G, Qiao Y, Xiao X, Zheng S, Chen L (2010) Effects of estrogen on lung development in a rat model of diaphragmatic hernia. J Pediatr Surg 45:2340–2345. https://doi.org/10.1016/j.jpedsurg.2010.08.028
Xu C, Liu W, Chen Z, Wang Y, Xiong Z, Ji Y (2009) Effect of prenatal tetrandrine administration on transforming growth factor-beta1 level in the lung of nitrofen-induced congenital diaphragmatic hernia rat model. J Pediatr Surg 44:1611–1620. https://doi.org/10.1016/j.jpedsurg.2008.09.021
Schaible T, Reineke J, Gortner L, Monz D, Schaffelder R, Tutdibi E (2016) Are cytokines useful biomarkers to determine disease severity in neonates with congenital diaphragmatic hernia? Am J Perinatol 34:648–654. https://doi.org/10.1055/s-0036-1597133
Dingemann J, Doi T, Ruttenstock E, Puri P (2010) Abnormal platelet-derived growth factor signaling accounting for lung hypoplasia in experimental congenital diaphragmatic hernia. J Pediatr Surg 45:1989–1994. https://doi.org/10.1016/j.jpedsurg.2010.06.014
Gosemann J-H, Friedmacher F, Hunziker M, Alvarez L, Corcionivoschi N, Puri P (2013) Increased activation of NADPH oxidase 4 in the pulmonary vasculature in experimental diaphragmatic hernia. Pediatr Surg Int 29:3–8. https://doi.org/10.1007/s00383-012-3209-0
Aras-López R, Tovar JA, Martínez L (2016) Possible role of increased oxidative stress in pulmonary hypertension in experimental diaphragmatic hernia. Pediatr Surg Int 32:141–145. https://doi.org/10.1007/s00383-015-3826-5
Cigdem MK, Kizil G, Onen A, Kizil M, Nergiz Y, Celik Y (2010) Is there a role for antioxidants in prevention of pulmonary hypoplasia in nitrofen-induced rat model of congenital diaphragmatic hernia? Pediatr Surg Int 26:401–406. https://doi.org/10.1007/s00383-010-2552-2
Hirako S, Tsuda H, Ito F, Okazaki Y, Hirayama T, Nagasawa H, Nakano T, Imai K, Kotani T, Kikkawa F, Toyokuni S (2017) Role of catalytic iron and oxidative stress in nitrofen-induced congenital diaphragmatic hernia and its amelioration by Saireito (TJ-114). J Clin Biochem Nutr 61:176–182. https://doi.org/10.3164/jcbn.17-17
Sluiter W, Bos AP, Silveri F, Tenbrinck R, Kraakslee R, Tibboel D, Koster JF, Molenaar JC (1992) Nitrofen-induced diaphragmatic hernias in rats: pulmonary antioxidant enzyme activities. Pediatr Res 32:394–398. https://doi.org/10.1203/00006450-199210000-00005
Taira Y, Oue T, Shima H, Miyazaki E, Puri P (1999) Increased tropoelastin and procollagen expression in the lung of nitrofen-induced diaphragmatic hernia in rats. J Pediatr Surg 34:715–719
Mychaliska GB, Officer SM, Heintz CK, Starcher BC, Pierce RA (2004) Pulmonary elastin expression is decreased in the nitrofen-induced rat model of congenital diaphragmatic hernia. J Pediatr Surg 39:666–671
Takahashi T, Friedmacher F, Zimmer J, Puri P (2018) Decreased expression of integrin subunits α3, α6, and α8 in the branching airway mesenchyme of nitrofen-induced hypoplastic lungs. Eur J Pediatr Surg Off J Austrian Assoc Pediatr Surg Al Z Kinderchir 28:109–114. https://doi.org/10.1055/s-0037-1604022
Burgos CM, Nord M, Roos A, Didon L, Eklöf A-C, Frenckner B (2010) Connective tissue growth factor expression pattern in lung development. Exp Lung Res 36:441–450. https://doi.org/10.3109/01902141003714056
Ruttenstock EM, Doi T, Dingemann J, Puri P (2011) Prenatal administration of retinoic acid upregulates connective tissue growth factor in the nitrofen CDH model. Pediatr Surg Int 27:573–577. https://doi.org/10.1007/s00383-010-2833-9
Pereira-Terra P, Kholdebarin R, Higgins M, Iwasiow BM, Correia-Pinto J, Keijzer R (2015) Lower NPAS3 expression during the later stages of abnormal lung development in rat congenital diaphragmatic hernia. Pediatr Surg Int 31:659–663. https://doi.org/10.1007/s00383-015-3703-2
Doi T, Hajduk P, Puri P (2009) Upregulation of Slit-2 and Slit-3 gene expressions in the nitrofen-induced hypoplastic lung. J Pediatr Surg 44:2092–2095. https://doi.org/10.1016/j.jpedsurg.2009.02.068
Akpinar İ, Korgun D, Çetin A, Yesilkaya A, Karaguzel G, Boneval C, Melikoglu M (2014) Epimorphin expression in a rat model of pulmonary hypoplasia associated with congenital diaphragmatic hernia. Pediatr Surg Int 30:1037–1043. https://doi.org/10.1007/s00383-014-3579-6
Friedmacher F, Fujiwara N, Hofmann AD, Takahashi H, Gosemann J-H, Puri P (2014) Evidence for decreased lipofibroblast expression in hypoplastic rat lungs with congenital diaphragmatic hernia. Pediatr Surg Int 30:1023–1029. https://doi.org/10.1007/s00383-014-3549-z
Chao C-M, Moiseenko A, Zimmer K-P, Bellusci S (2016) Alveologenesis: key cellular players and fibroblast growth factor 10 signaling. Mol Cell Pediatr. https://doi.org/10.1186/s40348-016-0045-7
Gouveia L, Betsholtz C, Andrae J (2018) PDGF-A signaling is required for secondary alveolar septation and controls epithelial proliferation in the developing lung. Dev Camb Engl. https://doi.org/10.1242/dev.161976
Keijzer R, Liu J, Deimling J, Tibboel D, Post M (2000) Dual-hit hypothesis explains pulmonary hypoplasia in the nitrofen model of congenital diaphragmatic hernia. Am J Pathol 156:1299–1306. https://doi.org/10.1016/S0002-9440(10)65000-6
Funding
This work was supported by Sickkids start-up funds and by the Canadian Institute of Health Research (CIHR)—SickKids Foundation New Investigator Research Grant (NI18-1270R).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Not applicable.
Rights and permissions
About this article
Cite this article
Montalva, L., Zani, A. Assessment of the nitrofen model of congenital diaphragmatic hernia and of the dysregulated factors involved in pulmonary hypoplasia. Pediatr Surg Int 35, 41–61 (2019). https://doi.org/10.1007/s00383-018-4375-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-018-4375-5