Abstract
Purpose
To explore pre-treatment risk factors for overall survival (OS) in advanced urothelial carcinoma (UC) patients treated with first-line (1L) chemotherapy in sequential therapy (ST) era. Additionally, to evaluate the proportion of patients who were not able to undergo subsequent immune checkpoint inhibitor (ICI) therapy according to the subgroups stratified by the risk factors.
Methods
A multicenter retrospective study was conducted. Metastatic or locally advanced UC patients treated between 2017 and 2022 were included. The Kaplan–Meier method with the log-rank test and multivariate Cox regression models were used to address OS.
Results
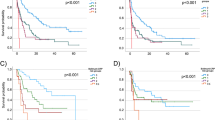
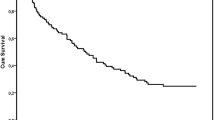
Three hundred and fourteen patients treated with 1L chemotherapy were included in the study and 57 (18.2%) patients were not able to proceed to subsequent ICI therapy. Pre-chemotherapy risk factors for OS in 314 patients were ECOG-PS 1 or more, having no primary site resection, C-reactive protein (CRP) level of 3 mg/dL or more, and non-cisplatin-based regimen. Patients having 3 or 4 risk factors had higher risk for not being able to receive ST (Mann–Whitney U test, P < 0.001). As risk factors for OS in 230 patients who were able to receive ST, having no primary site resection, a neutrophil to lymphocyte ratio of 3 or more, and the presence of liver metastasis were identified.
Conclusion
We reported the risk factors for OS in advanced UC patients treated with 1L chemotherapy in ST era. Patients with high risk for OS may not be able to proceed to subsequent ICI therapy even in the ST era.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, [KH], upon reasonable request.
References
Sternberg CN, Yagoda A, Scher HI, Watson RC, Ahmed T, Weiselberg LR, Geller N, Hollander PS, Herr HW, Sogani PC et al (1985) Preliminary results of M-VAC (methotrexate, vinblastine, doxorubicin and cisplatin) for transitional cell carcinoma of the urothelium. J Urol 133(3):403–407
von der Maase H, Hansen SW, Roberts JT, Dogliotti L, Oliver T, Moore MJ, Bodrogi I, Albers P, Knuth A, Lippert CM et al (2000) Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol 18(17):3068–3077
Bajorin DF, Dodd PM, Mazumdar M, Fazzari M, McCaffrey JA, Scher HI, Herr H, Higgins G, Boyle MG (1999) Long-term survival in metastatic transitional-cell carcinoma and prognostic factors predicting outcome of therapy. J Clin Oncol 17(10):3173–3181
Bellmunt J, Albanell J, Paz-Ares L, Climent MA, González-Larriba JL, Carles J, de la Cruz JJ, Guillem V, Díaz-Rubio E, Cortés-Funes H et al (2002) Pretreatment prognostic factors for survival in patients with advanced urothelial tumors treated in a phase I/II trial with paclitaxel, cisplatin, and gemcitabine. Cancer 95(4):751–757
von der Maase H, Sengelov L, Roberts JT, Ricci S, Dogliotti L, Oliver T, Moore MJ, Zimmermann A, Arning M (2005) Long-term survival results of a randomized trial comparing gemcitabine plus cisplatin, with methotrexate, vinblastine, doxorubicin, plus cisplatin in patients with bladder cancer. J Clin Oncol 23(21):4602–4608
Shinohara N, Harabayashi T, Suzuki S, Nagao K, Seki H, Murakumo M, Mitsuhashi K, Demura T, Nagamori S, Matsuyama H et al (2006) Salvage chemotherapy with paclitaxel, ifosfamide, and nedaplatin in patients with urothelial cancer who had received prior cisplatin-based therapy. Cancer Chemother Pharmacol 58(3):402–407
Kouno T, Ando M, Yonemori K, Matsumoto K, Shimizu C, Katsumata N, Komiyama M, Okajima E, Matsuoka N, Fujimoto H et al (2007) Weekly paclitaxel and carboplatin against advanced transitional cell cancer after failure of a platinum-based regimen. Eur Urol 52(4):1115–1122
Matsumoto K, Irie A, Satoh T, Okazaki M, Iwamura M, Baba S (2007) Gemcitabine and paclitaxel chemotherapy as a second-line treatment for advanced or metastatic urothelial carcinoma. Int J Urol 14(11):1000–1004 (discussion 1004)
Kitamura H, Taguchi K, Kunishima Y, Yanase M, Takahashi A, Shigyo M, Tanaka T, Mutoh M, Fukuta F, Masumori N et al (2011) Paclitaxel, ifosfamide, and nedaplatin as second-line treatment for patients with metastatic urothelial carcinoma: a phase II study of the SUOC group. Cancer Sci 102(6):1171–1175
Terakawa T, Miyake H, Yokoyama N, Miyazaki A, Tanaka H, Inoue T, Fujisawa M (2014) Clinical outcome of paclitaxel and carboplatin as second-line chemotherapy for advanced urothelial carcinoma resistant to first-line therapy with gemcitabine and cisplatin. Urol Int 92(2):180–185
Bellmunt J, de Wit R, Vaughn DJ, Fradet Y, Lee JL, Fong L, Vogelzang NJ, Climent MA, Petrylak DP, Choueiri TK et al (2017) Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N Engl J Med 376(11):1015–1026
Powles T, Park SH, Voog E, Caserta C, Valderrama BP, Gurney H, Kalofonos H, Radulovic S, Demey W, Ullen A et al (2020) Avelumab maintenance therapy for advanced or metastatic urothelial carcinoma. N Engl J Med 383(13):1218–1230
Miyake M, Nishimura N, Shimizu T, Ohnishi M, Kuwada M, Itami Y, Inoue T, Ohnishi K, Matsumoto Y, Yoshida T et al (2022) Significant improvement of prognosis after the advent of immune checkpoint inhibitors in patients with advanced, unresectable, or metastatic urothelial carcinoma: a propensity score matching and inverse probability of treatment weighting analysis on real-world data. Cancer Manage Res 14:623–635
Shindo T, Maehana T, Tanaka T, Hashimoto K, Kobayashi K, Takahashi A, Hotta H, Kunishima Y, Taguchi K, Tachiki H et al (2022) Are there differences in the characteristics of patients who respond to gemcitabine plus cisplatin chemotherapy and those who respond to pembrolizumab therapy for metastatic urothelial carcinoma? Multicenter retrospective study. Int J Urol 29(9):1010–1016
Taguchi S, Kawai T, Nakagawa T, Miyakawa J, Kishitani K, Sugimoto K, Nakamura Y, Kamei J, Obinata D, Yamaguchi K et al (2022) Improved survival in real-world patients with advanced urothelial carcinoma: a multicenter propensity score-matched cohort study comparing a period before the introduction of pembrolizumab (2003–2011) and a more recent period (2016–2020). Int J Urol 29(12):1462–1469
Jessen C, Agerbaek M, Von Der Maase H (2009) Predictive factors for response and prognostic factors for long-term survival in consecutive, single institution patients with locally advanced and/or metastatic transitional cell carcinoma following cisplatin-based chemotherapy. Acta Oncol 48(3):411–417
Galsky MD, Moshier E, Krege S, Lin CC, Hahn N, Ecke T, Sonpavde G, Godbold J, Oh WK, Bamias A (2013) Nomogram for predicting survival in patients with unresectable and/or metastatic urothelial cancer who are treated with cisplatin-based chemotherapy. Cancer 119(16):3012–3019
Kobayashi T, Ito K, Kojima T, Kato M, Kanda S, Hatakeyama S, Matsui Y, Matsushita Y, Naito S, Shiga M et al (2020) Risk stratification for the prognosis of patients with chemoresistant urothelial cancer treated with pembrolizumab. Cancer Sci 112(2):760–773
Sonpavde G, Manitz J, Gao C, Tayama D, Kaiser C, Hennessy D, Makari D, Gupta A, Abdullah SE, Niegisch G et al (2020) Five-factor prognostic model for survival of post-platinum patients with metastatic urothelial carcinoma receiving PD-L1 inhibitors. J Urol 204(6):1173–1179
Tamura D, Jinnouchi N, Abe M, Ikarashi D, Matsuura T, Kato R, Maekawa S, Kato Y, Kanehira M, Takata R et al (2020) Prognostic outcomes and safety in patients treated with pembrolizumab for advanced urothelial carcinoma: experience in real-world clinical practice. Int J Clin Oncol 25(5):899–905
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45(2):228–247
Bamias A, Merseburger A, Loriot Y, James N, Choy E, Castellano D, Lopez-Rios F, Calabro F, Kramer M, de Velasco G et al (2023) New prognostic model in patients with advanced urothelial carcinoma treated with second-line immune checkpoint inhibitors. J Immunother Cancer 11(1):e005977
Kanda Y (2013) Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transpl 48(3):452–458
Grivas P, Agarwal N, Pal S, Kalebasty AR, Sridhar SS, Smith J, Devgan G, Sternberg CN, Bellmunt J (2021) Avelumab first-line maintenance in locally advanced or metastatic urothelial carcinoma: applying clinical trial findings to clinical practice. Cancer Treat Rev 97:102187
Okita K, Hatakeyama S, Hagiwara K, Suzuki Y, Tanaka T, Noro D, Tokui N, Fujita N, Konishi S, Okamoto T et al (2021) The effect of number of treatment cycles of platinum-based first-line chemotherapy on maximum radiological response in patients with advanced urothelial carcinoma. Urol Oncol 39(12):832.e17-832.e23
Hoimes CJ, Flaig TW, Milowsky MI, Friedlander TW, Bilen MA, Gupta S, Srinivas S, Merchan JR, McKay RR, Petrylak DP et al (2023) Enfortumab vedotin plus pembrolizumab in previously untreated advanced urothelial cancer. J Clin Oncol 41(1):22–31
Galsky MD, Chen GJ, Oh WK, Bellmunt J, Roth BJ, Petrioli R, Dogliotti L, Dreicer R, Sonpavde G (2012) Comparative effectiveness of cisplatin-based and carboplatin-based chemotherapy for treatment of advanced urothelial carcinoma. Ann Oncol 23(2):406–410
Miron B, Handorf E, Zarrabi K, Zibelman MR, Anari F, Ghatalia P, Plimack ER, Geynisman DM (2022) Influence of first-line chemotherapy regimen on survival outcomes of patients with advanced urothelial carcinoma who received second-line immune checkpoint inhibitors. Urol Oncol 40(10):454.e9-454.e16
Kobayashi T, Ito K, Kojima T, Maruyama S, Mukai S, Tsutsumi M, Miki J, Okuno T, Yoshio Y, Matsumoto H et al (2021) Pre-pembrolizumab neutrophil-to-lymphocyte ratio (NLR) predicts the efficacy of second-line pembrolizumab treatment in urothelial cancer regardless of the pre-chemo NLR. Cancer Immunol Immunother 71(2):461–471
Yamamoto Y, Yatsuda J, Shimokawa M, Fuji N, Aoki A, Sakano S, Yamamoto M, Suga A, Tei Y, Yoshihiro S et al (2021) Prognostic value of pre-treatment risk stratification and post-treatment neutrophil/lymphocyte ratio change for pembrolizumab in patients with advanced urothelial carcinoma. Int J Clin Oncol 26(1):169–177
Abuhelwa AY, Bellmunt J, Kichenadasse G, McKinnon RA, Rowland A, Sorich MJ, Hopkins AM (2022) Enhanced Bellmunt risk score for survival prediction in urothelial carcinoma treated with immunotherapy. Clin Genitourin Cancer 20(2):132–138
Acknowledgements
This project was made possible with help of the data collected in the Sapporo Medical University Urologic Oncology Consortium (SUOC).
Funding
None.
Author information
Authors and Affiliations
Contributions
Protocol/project development: TS, KH, KK, TT, NM. Data collection or management: TS, AT, HH, TM, YK, FF, NI, AW, RK, MO, MM, HH, AT. Data analysis: TS, NM, KF. Manuscript writing/editing: TS, KH, NM.
Corresponding author
Ethics declarations
Conflict of interest
All other authors have nothing to declare.
Ethical approval
The study was performed in accordance with the Declaration of Helsinki and was approved by the institutional review board (Institutional Review Board number 332-300).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shindo, T., Hashimoto, K., Fujino, K. et al. Pre-first-line chemotherapy risk stratification for overall survival in advanced urothelial carcinoma in sequential therapy era. World J Urol 42, 307 (2024). https://doi.org/10.1007/s00345-024-04952-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00345-024-04952-z