Abstract
Purpose
Tumors escape from the immune system by decreasing CD8+ and increasing CD4+ T cells’ activity, druggable targets. Thermal ablation might activate tumor-specific T cells by raising the presentation of tumor-specific antigens and hindering tumor negative immune regulation. Our aim was to assess T cell infiltrate pre- and post-cryoablation in a prospective observational study.
Methods
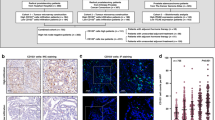
A total of 240 sextant prostate biopsies cores (12 cores/patient) were collected from 10 unilateral prostate cancer patients (T1c, PSA density < 0.15 ng/dL, Gleason grade group 1, ≤ 2 cancer biopsy cores, and < 50% cancer core involvement) at diagnosis and 12 months after hemi-cryoablation. Cancer-positive (Diag+) and cancer-negative (Diag−) lobes at diagnosis and the same areas 12 months after hemi-cryoablation (Cryo+ and Cryo−, respectively) were explored by immunohistochemistry for infiltrating CD4+ and CD8+ T cells (in 45 random fields per prostate lobe, 400× magnification). The quantitative analysis of cells/mm2 and CD4+/CD8+ ratio were performed and compared among Diag+, Diag−, Cryo+, and Cryo− using ImageJ software.
Results
There was a significant increase in tumor-infiltrating CD8+ T cells/mm2 in the Cryo+ tissue (mean, SD 0.31, 0.30) compared to Diag+ (0.18, 0.15), p = 0.015; confirmed in prostate acini (hot spots), p = 0.029, in which infiltrating CD4+/CD8+ T cells’ ratio decreased after hemi-cryoablation, p = 0.006. Infiltrating CD4+ T cells/mm2 presented a trend to decrease in Cryo+ (0.26, 0.27) compared to Diag+ (0.38, 0.32).
Conclusions
This is the first study to show local immune modulation after prostate cancer cryoablation, characterized by decreasing CD4+/CD8+ T cells’ ratio, potential for clinical impact by unleashing the T-cell response to cancer. Future studies are necessary to explore different energies and longer follow-up clinical endpoints.



Similar content being viewed by others
References
Reis LO, Billis A, Zequi SC, Tobias-Machado M, Viana P, Cerqueira M, Ward JF (2014) Supporting prostate cancer focal therapy: a multidisciplinary International Consensus of Experts (“ICE”). Aging Male 17(2):66–71
Sha TT, Peters M, Eldred-Evans D, Miah S, Yap T, Faure-Walker NA, Hosking-Jervis F, Thomas B, Dudderidge T, Hindley RG, McCracken S, Greene D, Nigam R, Valerio M, Minhas S, Winkler M, Arya M, Ahmed HU (2019) Early-medium-term outcomes of primary focal cryotherapy to treat nonmetastatic clinically significant prostate cancer from a prospective multicentre registry. Eur Urol. https://doi.org/10.1016/j.eururo.2018.12.030
Cerqueira MA, Laranja WW, Sanches BC et al (2015) Burden of focal cryoablation versus brachytherapy versus active surveillance in the treatment of very low-risk prostate cancer: a preliminary head-to-head comprehensive assessment. Eur J Cancer Care (Engl) 24:929–937
Reis LO, Carter HB (2015) The mind: focal cryotherapy in low-risk prostate cancer: are we treating the cancer or the mind? Int Braz J Urol 41(1):10–14
Gannon PO, Poisson AO, Delvoye N et al (2009) Characterization of the intra-prostatic immune cell infiltration in androgen-deprived prostate cancer patients. J Immunol Methods 348:9–17
Sanches BCF, Lalli AL, Azal Neto W, Billis A, Reis LO (2018) Are 10-, 10–12-, or > 12-mm prostate biopsy core quality control cutoffs reasonable? World J Urol 36(7):1055–1058
The nobel prize in physiology or medicine 2018. NobelPrize.org. Nobel Media AB 2018. https://www.nobelprize.org/prizes/medicine/2018/summary/. Accessed 2 Oct 2018
Rueden CT, Eliceiri KW (2007) Visualization approaches for multidimensional biological image data. BioTechniques 43(1 Suppl):31–36
Shah TT, Kasivisvanathan V, Jameson C et al (2015) Histological outcomes after focal high-intensity focused ultrasound and cryotherapy. World J Urol 33:955–964
Soanes WA, Ablin RJ, Gonder MJ (1970) Remission of metastatic lesions following cryosurgery in prostatic cancer: immunologic considerations. J Urol 104:154–159
Di Carlo E, Magnasco S, D’Antuono T et al (2007) The prostate-associated lymphoid tissue (PALT) is linked to the expression of homing chemokines CXCL13 and CCL21. Prostate 67:1070–1080
Sabel MS (2009) Cryo-immunology: a review of the literature and proposed mechanisms for stimulatory versus suppressive immune responses. Cryobiology 58(1):1–11
Garcia-Hernnández ML, Uribe-Uribe NO, Espinosa-González R, Kast WM, Khader SA, Rangel-Moreno J (2017) A unique cellular and molecular microenvironment is present in tertiary lymphoid organs of patients with spontaneous prostate cancer regression. Front Immunol 17(8):563
Davidsson S, Ohlson A, Andersson S et al (2013) CD4 helper T cells, CD8 cytotoxic T cells, and FOXP3þ regulatory T cells with respect to lethal prostate cancer. Mod Pathol 26:448–455
Becht E, Giraldo NA, Germain C et al (2016) Immune contexture, immunoscore, and malignant cell molecular subgroups for prognostic and theranostic classifications of cancers. Adv Immunol 130:96–190
Tucker JA, Jochems C, Gulley JL, Schlom J, Tsang KY (2012) Immunotherapy: shifting the balance of cell-mediated immunity and suppression in human prostate cancer. Cancers (Basel) 4(4):1333–1348
Waitz R, Solomon SB, Petre EN et al (2012) Potent induction of tumor immunity by combining tumor cryoablation with anti-CTLA-4 therapy. Cancer Res 72(2):430–439
Davidsson S, Andren O, Ohlson AL, Carlsson J, Andersson SO, Giunchi F, Rider JR, Fiorentino M (2018) FOXP3+ regulatory T cells in normal prostate tissue, post-atrophic hyperplasia, prostatic intraepithelial neoplasia, and tumor histological lesions in men with and without prostate cancer. Prostate 78(1):40–47
McArdle PA, Canna K, Mcmillan DC, Underwood MA et al (2004) The relationship between T-lymphocyte subset infiltration and survival in patients with prostate cancer. Br J Cancer 91:541–543
Sharabi AB, Nirschl CJ, Kochel CM (2015) Stereotactic radiation therapy augments antigen-specific PD-1-mediated anti-tumor immune responses via cross-presentation of tumor antigen. Cancer Immunol Res 3:345–355
Waitz R, Solomon SB, Petre EN, Trumble AE, Fasso M, Norton L et al (2012) Potent induction of tumor immunity by combining tumor cryoablation with anti-CTLA-4 therapy. Cancer Res 72:430–439
Benzon B, Glavaris SA, Simons BW, Hughes RM, Ghabili K, Mullane P, Miller R, Nugent K, Shinder B, Tosoian J, Fuchs EJ, Tran PT, Hurley PJ, Vuica-Ross M, Schaeffer EM, Drake CG, Ross AE (2018) Combining immune check-point blockade and cryoablation in an immunocompetent hormone sensitive murine model of prostate cancer. Prostate Cancer Prostatic Dis 21(1):126–136
ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US) (2015) Identifier NCT02423928, Phase I Clinical Trial of Cryoimmunotherapy Against Prostate Cancer (CryoIT). https://clinicaltrials.gov/ct2/show/study/NCT02423928. Accessed 26 May 2019
Yang ZR, Zhao N, Meng J, Shi ZL, Li BX, Wu XW, Li P, Zhang Q, Wei XB, Fu S (2016) Peripheral lymphocyte subset variation predicts prostate cancer carbon ion radiotherapy outcomes. Oncotarget 7(18):26422–26435
Haen SP, Pereira PL, Salih HR, Rammensee HG, Gouttefangeas C (2011) More than just tumor destruction: immunomodulation by thermal ablation of cancer. Clin Dev Immunol 2011:160250
Acknowledgements
To the involved institution(s), the patients and those that provided and cared for study patients.
Funding
Reis LO: CAPES, Brazil—Grant: BEX 14679/13-2 and CNPq Research Productivity, Brazil—Grant: 302622/2015-2
Author information
Authors and Affiliations
Contributions
MAC: clinical data collection, data analysis, and manuscript editing; KLF and ACM: experimental data collection and analysis, and manuscript writing; CRM: funding acquisition, manuscript editing and supervision. LOR: project development, funding acquisition, manuscript writing, and supervision.
Corresponding author
Ethics declarations
Conflict of interest
None declared.
Research involving human participants
Research ethics committee approval protocol nº 2.013.568.
Informed consent
Obtained from all patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Leonardo Oliveira Reis: Preliminary results awarded Best Moderated Poster at 2018 American Urological Association Annual Meeting, May 18–21, San Francisco.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cerqueira, M.A., Ferrari, K.L., de Mattos, A.C. et al. T cells CD4+/CD8+ local immune modulation by prostate cancer hemi-cryoablation. World J Urol 38, 673–680 (2020). https://doi.org/10.1007/s00345-019-02861-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-019-02861-0