Abstract
Objectives
To establish a nomogram for predicting central lymph node metastasis (CLNM) based on the preoperative clinical and multimodal ultrasound (US) features of papillary thyroid carcinoma (PTC) and cervical LNs.
Methods
Overall, 822 patients with PTC were included in this retrospective study. A thyroid tumor ultrasound model (TTUM) and thyroid tumor and cervical LN ultrasound model (TTCLNUM) were constructed as nomograms to predict the CLNM risk. Areas under the curve (AUCs) evaluated model performance. Calibration and decision curves were applied to assess the accuracy and clinical utility.
Results
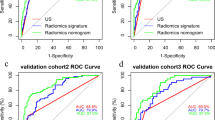
For the TTUM training and test sets, the AUCs were 0.786 and 0.789 and bias-corrected AUCs were 0.786 and 0.831, respectively. For the TTCLNUM training and test sets, the AUCs were 0.806 and 0.804 and bias-corrected AUCs were 0.807 and 0.827, respectively. Calibration and decision curves for the TTCLNUM nomogram exhibited higher accuracy and clinical practicability. The AUCs were 0.746 and 0.719 and specificities were 0.942 and 0.905 for the training and test sets, respectively, when the US tumor size was ≤ 8.45 mm, while the AUCs were 0.737 and 0.824 and sensitivity were 0.905 and 0.880, respectively, when the US tumor size was > 8.45 mm.
Conclusion
The TTCLNUM nomogram exhibited better predictive performance, especially for the CLNM risk of different PTC tumor sizes. Thus, it serves as a useful clinical tool to supply valuable information for active surveillance and treatment decisions.
Key Points
• Our preoperative noninvasive and intuitive prediction method can improve the accuracy of central lymph node metastasis (CLNM) risk assessment and guide clinical treatment in line with current trends toward personalized treatments.
• Preoperative clinical and multimodal ultrasound features of primary papillary thyroid carcinoma (PTC) tumors and cervical LNs were directly used to build an accurate and easy-to-use nomogram for predicting CLNM.
• The thyroid tumor and cervical lymph node ultrasound model exhibited better performance for predicting the CLNM of different PTC tumor sizes. It may serve as a useful clinical tool to provide valuable information for active surveillance and treatment decisions.






Similar content being viewed by others
Abbreviations
- CI:
-
Confidence interval
- CLN:
-
Central lymph node
- CLND:
-
Central lymph node dissection
- CLNM:
-
Central lymph node metastasis
- LLN:
-
Lateral lymph node
- LLNM:
-
Lateral lymph node metastasis
- OR:
-
Odds ratio
- PTC:
-
Papillary thyroid carcinoma
References
Cabanillas ME, McFadden DG, Durante C (2016) Thyroid cancer. Lancet 388:2783–2795
Papaleontiou M, Evron JM, Esfandiari NH et al (2020) Patient report of recurrent and persistent thyroid cancer. Thyroid 30:1297–1305
Al Afif A, Williams BA, Rigby MH et al (2015) Multifocal papillary thyroid cancer increases the risk of central lymph node metastasis. Thyroid 25:1008–1012
Machens A, Hinze R, Thomusch O, Dralle H (2002) Pattern of nodal metastasis for primary and reoperative thyroid cancer. World J Surg 26:22–28
Lan X, Sun W, Zhang H, Dong W, Wang Z, Zhang T (2015) A meta-analysis of central lymph node metastasis for predicting lateral involvement in papillary thyroid carcinoma. Otolaryngol Head Neck Surg 153:731–738
Podnos YD, Smith D, Wagman LD, Ellenhorn JD (2005) The implication of lymph node metastasis on survival in patients with well-differentiated thyroid cancer. Am Surg 71:731–734
Zaydfudim V, Feurer ID, Griffin MR, Phay JE (2008) The impact of lymph node involvement on survival in patients with papillary and follicular thyroid carcinoma. Surgery 144:1070–1078
Haddad RI, Nasr C, Bischoff L et al (2018) NCCN guidelines insights: thyroid carcinoma, version 2.2018. J Natl Compr Cancer Netw 16:1429–1440
Conzo G, Mauriello C, Docimo G et al (2014) Clinicopathological pattern of lymph node recurrence of papillary thyroid cancer. Implications for surgery. Int J Surg 12(Suppl 1):S194–S197
Randolph GW, Duh QY, Heller KS et al (2012) The prognostic significance of nodal metastases from papillary thyroid carcinoma can be stratified based on the size and number of metastatic lymph nodes, as well as the presence of extranodal extension. Thyroid 22:1144–1152
Mazzaferri EL, Doherty GM, Steward DL (2009) The pros and cons of prophylactic central compartment lymph node dissection for papillary thyroid carcinoma. Thyroid 19:683–689
Haugen BR, Alexander EK, Bible KC et al (2016) 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 26:1–133
Zhou W, Jiang S, Zhan W, Zhou J, Xu S, Zhang L (2017) Ultrasound-guided percutaneous laser ablation of unifocal T1N0M0 papillary thyroid microcarcinoma: preliminary results. Eur Radiol 27:2934–2940
Teng D, Sui G, Liu C, Wang Y, Xia Y, Wang H (2018) Long-term efficacy of ultrasound-guided low power microwave ablation for the treatment of primary papillary thyroid microcarcinoma: a 3-year follow-up study. J Cancer Res Clin Oncol 144:771–779
Jeong SY, Baek JH, Choi YJ et al (2018) Radiofrequency ablation of primary thyroid carcinoma: efficacy according to the types of thyroid carcinoma. Int J Hyperth 34:611–616
Hegedüs L (2004) The thyroid nodule. N Engl J Med 351:1764–1771
Zhou J, Yin L, Wei X et al (2020) Chinese guidelines for ultrasound malignancy risk stratification of thyroid nodules: the C-TIRADS. Endocrine 70:256–279
Moreno MA, Agarwal G, de Luna R et al (2011) Preoperative lateral neck ultrasonography as a long-term outcome predictor in papillary thyroid cancer. Arch Otolaryngol Head Neck Surg 137:157–162
Roh JL, Park JY, Kim JM, Song CJ (2009) Use of preoperative ultrasonography as guidance for neck dissection in patients with papillary thyroid carcinoma. J Surg Oncol 99:28–31
Abboud B, Smayra T, Jabbour H, Ghorra C, Abadjian G (2020) Correlations of neck ultrasound and pathology in cervical lymph node of papillary thyroid carcinoma. Acta Chir Belg 120:238–244
Zhao H, Li H (2019) Meta-analysis of ultrasound for cervical lymph nodes in papillary thyroid cancer: diagnosis of central and lateral compartment nodal metastases. Eur J Radiol 112:14–21
Kim E, Park JS, Son KR, Kim JH, Jeon SJ, Na DG (2008) Preoperative diagnosis of cervical metastatic lymph nodes in papillary thyroid carcinoma: comparison of ultrasound, computed tomography, and combined ultrasound with computed tomography. Thyroid 18:411–418
Ahn JE, Lee JH, Yi JS et al (2008) Diagnostic accuracy of CT and ultrasonography for evaluating metastatic cervical lymph nodes in patients with thyroid cancer. World J Surg 32:1552–1558
Hwang HS, Orloff LA (2011) Efficacy of preoperative neck ultrasound in the detection of cervical lymph node metastasis from thyroid cancer. Laryngoscope 121:487–491
Feng JW, Hong LZ, Wang F et al (2021) A nomogram based on clinical and ultrasound characteristics to predict central lymph node metastasis of papillary thyroid carcinoma. Front Endocrinol (Lausanne) 12:666315
Feng Y, Min Y, Chen H, Xiang K, Wang X, Yin G (2021) Construction and validation of a nomogram for predicting cervical lymph node metastasis in classic papillary thyroid carcinoma. J Endocrinol Invest 44:2203–2211
Kim SK, Chai YJ, Park I et al (2017) Nomogram for predicting central node metastasis in papillary thyroid carcinoma. J Surg Oncol 115:266–272
Chinese Medical Doctors Association Ultrasonic Physicians Branch (2011) Ultrasound guidelines of the vessels and superficial organs, 1st edn. People’s Military Medical Publishing House, Beijing
Adler DD, Carson PL, Rubin JM, Quinn-Reid D (1990) Doppler ultrasound color flow imaging in the study of breast cancer: preliminary findings. Ultrasound Med Biol 16:553–559
Asteria C, Giovanardi A, Pizzocaro A et al (2008) US-elastography in the differential diagnosis of benign and malignant thyroid nodules. Thyroid 18:523–531
Yan S, Zhan YW, Zhou JQ (2009) Ultrasound of the thyroid and parathyroid glands, 1st edn. Scientific and Technical Documents Publishing House, Beijing
Wu LM, Gu HY, Qu XH et al (2012) The accuracy of ultrasonography in the preoperative diagnosis of cervical lymph node metastasis in patients with papillary thyroid carcinoma: a meta-analysis. Eur J Radiol 81:1798–1805
Leboulleux S, Girard E, Rose M et al (2007) Ultrasound criteria of malignancy for cervical lymph nodes in patients followed up for differentiated thyroid cancer. J Clin Endocrinol Metab 92:3590–3594
Svanholm H, Starklint H, Gundersen HJ, Fabricius J, Barlebo H, Olsen S (1989) Reproducibility of histomorphologic diagnoses with special reference to the kappa statistic. APMIS 97:689–698
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–845
Balachandran VP, Gonen M, Smith JJ, DeMatteo RP (2015) Nomograms in oncology—more than meets the eye. Lancet Oncol 16:173–180
Tian X, Song Q, Xie F et al (2020) Papillary thyroid carcinoma: an ultrasound-based nomogram improves the prediction of lymph node metastases in the central compartment. Eur Radiol 30:5881–5893
Huang XP, Ye TT, Zhang L et al (2018) Sonographic features of papillary thyroid microcarcinoma predicting high-volume central neck lymph node metastasis. Surg Oncol 27:172–176
Wang WH, Xu SY, Zhan WW (2016) Clinicopathologic factors and thyroid nodule sonographic features for predicting central lymph node metastasis in papillary thyroid microcarcinoma a retrospective study of 1204 patients. J Ultrasound Med 35:2475–2481
Ahn BH, Kim JR, Jeong HC, Lee JS, Chang ES, Kim YH (2015) Predictive factors of central lymph node metastasis in papillary thyroid carcinoma. Ann Surg Treat Res 88:63–68
Ferreira LB, Lima RT, Bastos ACSDF et al (2018) OPNa overexpression is associated with matrix calcification in thyroid cancer cell lines. Int J Mol Sci 19:2990
Triggiani V, Guastamacchia E, Licchelli B, Tafaro E (2008) Microcalcifications and psammoma bodies in thyroid tumors. Thyroid 18:1017–1018
Jang JY, Kim DS, Park HY et al (2019) Preoperative serum VEGF-C but not VEGF-A level is correlated with lateral neck metastasis in papillary thyroid carcinoma. Head Neck 41:2602–2609
Tu DG, Chang WW, Jan MS, Tu CW, Lu YC, Tai CK (2016) Promotion of metastasis of thyroid cancer cells via NRP-2-mediated induction. Oncol Lett 12:4224–4230
Wu Q, Li Y, Wang Y, Hu B (2015) Sonographic features of primary tumor as independent predictive factors for lymph node metastasis in papillary thyroid carcinoma. Clin Transl Oncol 17:830–834
Choi YJ, Yun JS, Kook SH, Jung EC, Park YL (2010) Clinical and imaging assessment of cervical lymph node metastasis in papillary thyroid carcinomas. World J Surg 34:1494–1499
Xu JM, Xu XH, Xu HX et al (2016) Prediction of cervical lymph node metastasis in patients with papillary thyroid cancer using combined conventional ultrasound, strain elastography, and acoustic radiation force impulse (ARFI) elastography. Eur Radiol 26:2611–2622
Park AY, Kim JA, Son EJ, Youk JH (2016) Shear-wave elastography for papillary thyroid carcinoma can improve prediction of cervical lymph node metastasis. Ann Surg Oncol 23:722–729
Shin LK, Olcott EW, Jeffrey RB, Desser TS (2013) Sonographic evaluation of cervical lymph nodes in papillary thyroid cancer. Ultrasound Q 29:25–32
Genes I, Mogoantă CA, Lostun G, Lostun A, Mózes H, Műhlfay G (2014) Ultrasonographic and histopathological features of cervical lymph node metastases. Rom J Morphol Embryol 55:369–375
Kim JM (2021) The clinical importance of multifocality on tumor recurrence in papillary thyroid carcinoma. Gland Surg 10:273–278
Wang QC, Cheng W, Wen X, Li JB, Jing H, Nie CL (2014) Shorter distance between the nodule and capsule has greater risk of cervical lymph node metastasis in papillary thyroid carcinoma. Asian Pac J Cancer Prev 15:855–860
Cai J, Fang F, Chen J, Xiang D (2020) Unilateral multifocality and bilaterality could be two different multifocal entities in patients with papillary thyroid microcarcinoma. Biomed Res Int 2020:9854964
Li M, Zhu XY, Lv J et al (2017) Risk factors for predicting central lymph node metastasis in papillary thyroid microcarcinoma (CN0): a study of 273 resections. Eur Rev Med Pharmacol Sci 21:3801–3807
Acknowledgements
We would like to thank Editage (www.editage.cn) for English language editing.
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Xiaoping Leng.
Conflict of Interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and Biometry
One of the authors has significant statistical expertise.
Informed Consent
Written informed consent was waived by the institutional review board.
Ethical Approval
Institutional review board approval was obtained.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dai, Q., Liu, D., Tao, Y. et al. Nomograms based on preoperative multimodal ultrasound of papillary thyroid carcinoma for predicting central lymph node metastasis. Eur Radiol 32, 4596–4608 (2022). https://doi.org/10.1007/s00330-022-08565-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-08565-1