Abstract
Objectives
To evaluate a deep learning model for predicting gestational age from fetal brain MRI acquired after the first trimester in comparison to biparietal diameter (BPD).
Materials and methods
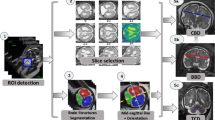
Our Institutional Review Board approved this retrospective study, and a total of 184 T2-weighted MRI acquisitions from 184 fetuses (mean gestational age: 29.4 weeks) who underwent MRI between January 2014 and June 2019 were included. The reference standard gestational age was based on the last menstruation and ultrasonography measurements in the first trimester. The deep learning model was trained with T2-weighted images from 126 training cases and 29 validation cases. The remaining 29 cases were used as test data, with fetal age estimated by both the model and BPD measurement. The relationship between the estimated gestational age and the reference standard was evaluated with Lin’s concordance correlation coefficient (ρc) and a Bland-Altman plot. The ρc was assessed with McBride’s definition.
Results
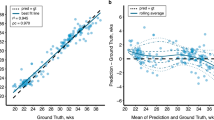
The ρc of the model prediction was substantial (ρc = 0.964), but the ρc of the BPD prediction was moderate (ρc = 0.920). Both the model and BPD predictions had greater differences from the reference standard at increasing gestational age. However, the upper limit of the model’s prediction (2.45 weeks) was significantly shorter than that of BPD (5.62 weeks).
Conclusions
Deep learning can accurately predict gestational age from fetal brain MR acquired after the first trimester.
Key Points
• The prediction of gestational age using ultrasound is accurate in the first trimester but becomes inaccurate as gestational age increases.
• Deep learning can accurately predict gestational age from fetal brain MRI acquired in the second and third trimester.
• Prediction of gestational age by deep learning may have benefits for prenatal care in pregnancies that are underserved during the first trimester.






Similar content being viewed by others
Abbreviations
- BPD:
-
Biparietal diameter
- CI:
-
Confidence interval
- CNN:
-
Convolutional neural network
- DICOM:
-
Digital Imaging and Communications in Medicine
- ρc:
-
Lin’s concordance correlation coefficient
References
Committee on Obstetric Practice, American Institute of Ultrasound in Medicine, Society for Maternal-Fetal Medicine. Committee Opinion No 700 (2017) Methods for estimating the due date. Obstet Gynecol. Available via https://journals.lww.com/greenjournal/Fulltext/2017/05000/Committee_Opinion_No_700__Methods_for_Estimating.50.aspx. Accessed 16 March 2020
Reddy UM, Abuhamad AZ, Levine D et al (2014) Fetal imaging: executive summary of a joint Eunice Kennedy Shriver National Institute of Child Health and Human Development, Society for Maternal-Fetal Medicine, American Institute of Ultrasound in Medicine, American College of Obstetricians and Gynecologists, American College of Radiology, Society for Pediatric Radiology, and Society of Radiologists in Ultrasound Fetal Imaging workshop. Obstet Gynecol 123:1070–1082
Mongelli M, Wilcox M, Gardosi J (1996) Estimating the date of confinement: ultrasonographic biometry versus certain menstrual dates. Am J Obstet Gynecol 174:278–281
Bennett KA, Crane JM, O’shea P et al (2004) First trimester ultrasound screening is effective in reducing postterm labor induction rates: a randomized controlled trial. Am J Obstet Gynecol 190:1077–1081
LeCun Y, Bengio Y, Hinton G (2015) Deep learning. Nature 521:436–444
Wang S, Summers RM (2012) Machine learning and radiology. Med Image Anal 16:933–951
Krizhevsky A, Sutskever I, Hinton GE (2012) ImageNet classification with deep convolutional neural networks. Advances in Neural Information Processing System 25:1097–1105
Akkus Z, Galimzianova A, Hoogi A et al (2017) Deep learning for brain MRI segmentation: state of the art and future directions. J Digit Imaging 449–459
Gong E, Pauly JM, Wintermark M et al (2018) Deep learning enables reduced gadolinium dose for contrast-enhanced brain MRI. J Magn Reson Imaging 48:330–340
Larson DB, Chen MC, Lungren MP et al (2018) Performance of a deep-learning neural network model in assessing skeletal maturity on pediatric hand radiographs. Radiology 287:313–322
Narayana PA, Coronado I, Sujit SJ et al (2020) Deep learning for predicting enhancing lesions in multiple sclerosis from noncontrast MRI. Radiology 294:398–404
Nishio M, Sugiyama O, Yakami M et al (2018) Computer-aided diagnosis of lung nodule classification between benign nodule, primary lung cancer, and metastatic lung cancer at different image size using deep convolutional neural network with transfer learning. PLoS One. Available via https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0200721. Accessed 16 March 2020
Cole JH, Poudel RPK, Tsagkrasoulis D et al (2017) Predicting brain age with deep learning from raw imaging data results in a reliable and heritable biomarker. Neuroimage. 163:115–124
Cole JH, Franke K (2017) Predicting age using neuroimaging: innovative brain ageing biomarkers. Trends Neurosci 40:681–690
Prayer D, Kasprian G, Krampl E et al (2006) MRI of normal fetal brain development. Eur J Radiol 57:199–216
The Global Library of Women’s Medicine. Assessment of gestational age by ultrasound. The International Federation of Gynecology and Obstetrics Web site. Available via https://www.glowm.com/section_view/heading/AssessmentofGestationalAgebyUltrasound/item/206. Accessed 16 March 2020
Simonyan K, Zisserman A (2014) Very deep convolutional networks for large-scale image recognition. Cornell University. Available via https://arxiv.org/abs/1409.1556. Accessed 16 March 2020
Torrey L, Shavlik J (2010) Transfer learning. Handbook of Research on Machine Learning Applications. Hershey, Pa: IGI Global. http://ftp.cs.wisc.edu/machine-learning/shavlik-group/torrey.handbook09.pdf. Accessed 26 Mar 2021
McBride GB (2005) A proposal for strength-of-agreement criteria for Lin’s concordance correlation coefficient. NIWA Client Report: HAM2005-062
Nishio M, Koyama H, Ohno Y et al (2016) Emphysema quantification using ultralow-dose CT with iterative reconstruction and filtered back projection. AJR Am J Roentgenol 206:1184–1192
Mets OM, Willemink MJ, de Kort FP et al (2012) The effect of iterative reconstruction on computed tomography assessment of emphysema, air trapping and airway dimensions. Eur Radiol 22:2103–2109
Koyama H, Ohno Y, Nishio M et al (2014) Iterative reconstruction technique vs filter back projection: utility for quantitative bronchial assessment on low-dose thin-section MDCT in patients with/ without chronic obstructive pulmonary disease. Eur Radiol 24:1860–1867
Wu J, Awate SP, Licht DJ et al (2015) Assessment of MRI-based automated fetal cerebral cortical folding measures in prediction of gestational age in the third trimester. AJNR Am J Neuroradiol 36:1369–1374
Maria KM, Gerardine Q, Mary AR et al (2012) Reconstruction of fetal brain MRI with intensity matching and complete outlier removal. Med Image Anal 16:1550–1564
Matthew J, Malamateniou C, Knight CL et al (2018) A comparison of ultrasound with magnetic resonance imaging in the assessment of fetal biometry and weight in the second trimester of pregnancy: an observer agreement and variability study. Ultrasound 26:229–244
Prayer D, Malinger G, Brugger PC et al (2017) ISUOG practice guidelines: performance of fetal magnetic resonance imaging. Ultrasound Obstet Gynecol 49:671–680
Ray GJ, Vermeulen MJ, Bharatha A et al (2016) Association between MRI exposure during pregnancy and fetal and childhood outcomes. JAMA 316:952–961
Kyriakopoulou V, Vatansever D, Davidson A et al (2017) Normative biometry of the fetal brain using magnetic resonance imaging. Brain Struct Funct 222:2295–2307
Acknowledgements
Edanz Group (https://en-author-services.edanz.com/ac) for editing a draft of this manuscript.
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Tomonori Kanda.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kojita, Y., Matsuo, H., Kanda, T. et al. Deep learning model for predicting gestational age after the first trimester using fetal MRI. Eur Radiol 31, 3775–3782 (2021). https://doi.org/10.1007/s00330-021-07915-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-021-07915-9