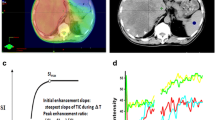
Abstract
Objectives
To correlate early changes in the parameters of dynamic contrast-enhanced magnetic resonance imaging (DCE-MRI) within 1 week of systemic therapy with overall survival (OS) in patients with advanced hepatocellular carcinoma (HCC).
Methods
Eighty-nine patients with advanced HCC underwent DCE-MRI before and within 1 week following systemic therapy. The relative changes of six DCE-MRI parameters (Peak, Slope, AUC, Ktrans, Kep and Ve) of the tumours were correlated with OS using the Kaplan–Meier model and the double-sided log-rank test.
Results
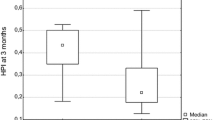
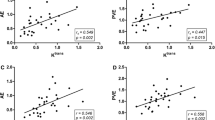
All patients died and the median survival was 174 days. Among the six DCE-MRI parameters, reductions in Peak, AUC, and Ktrans, were significantly correlated with one another. In addition, patients with a high Peak reduction following treatment had longer OS (P = 0.023) compared with those with a low Peak reduction. In multivariate analysis, a high Peak reduction was an independent favourable prognostic factor in all patients [hazard ratio (HR), 0.622; P = 0.038] after controlling for age, sex, treatment methods, tumour size and stage, and Eastern Cooperative Oncology Group performance status.
Conclusions
Early perfusion changes within 1 week following systemic therapy measured by DCE-MRI may aid in the prediction of the clinical outcome in patients with advanced HCC.
Key points
• DCE-MRI is helpful to evaluate perfusion changes of HCC after systemic treatment.
• Early perfusion changes within 1 week after treatment may predict overall survival.
• High Peak reduction was an independent favourable prognostic factor after systemic treatment.




Similar content being viewed by others
References
Altekruse SF, McGlynn KA, Reichman ME (2009) Hepatocellular carcinoma incidence, mortality, and survival trends in the United States from 1975 to 2005. J Clin Oncol 27:1485–1491
Chen KW, Ou TM, Hsu CW et al (2015) Current systemic treatment of hepatocellular carcinoma: a review of the literature. World J Hepatol 7:1412–1420
Cheng AL, Kang YK, Chen Z et al (2009) Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol 10:25–34
Llovet JM, Ricci S, Mazzaferro V et al (2008) Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 359:378–390
Deng GL, Zeng S, Shen H (2015) Chemotherapy and target therapy for hepatocellular carcinoma: new advances and challenges. World J Hepatol 7:787–798
Shao YY, Hsu CH, Cheng AL (2015) Predictive biomarkers of sorafenib efficacy in advanced hepatocellular carcinoma: are we getting there? World J Gastroenterol 21:10336–10347
Corona-Villalobos CP, Halappa VG, Geschwind JF et al (2015) Volumetric assessment of tumour response using functional MR imaging in patients with hepatocellular carcinoma treated with a combination of doxorubicin-eluting beads and sorafenib. Eur Radiol 25:380–390
De Robertis R, Tinazzi Martini P, Demozzi E et al (2015) Prognostication and response assessment in liver and pancreatic tumors: the new imaging. World J Gastroenterol 21:6794–6808
Duffy AG, Ulahannan SV, Cao L et al (2015) A phase II study of TRC105 in patients with hepatocellular carcinoma who have progressed on sorafenib. United European Gastroenterol J 3:453–461
O'Connor JP, Jackson A, Parker GJ, Roberts C, Jayson GC (2012) Dynamic contrast-enhanced MRI in clinical trials of antivascular therapies. Nat Rev Clin Oncol 9:167–177
Fischer MA, Kartalis N, Grigoriadis A et al (2015) Perfusion computed tomography for detection of hepatocellular carcinoma in patients with liver cirrhosis. Eur Radiol 25:3123–3132
Chen BB, Shih TT (2014) DCE-MRI in hepatocellular carcinoma-clinical and therapeutic image biomarker. World J Gastroenterol 20:3125–3134
Jajamovich GH, Huang W, Besa C et al (2016) DCE-MRI of hepatocellular carcinoma: perfusion quantification with Tofts model versus shutter-speed model-initial experience. MAGMA 29:49–58
Pickles MD, Lowry M, Manton DJ, Turnbull LW (2015) Prognostic value of DCE-MRI in breast cancer patients undergoing neoadjuvant chemotherapy: a comparison with traditional survival indicators. Eur Radiol 25:1097–1106
Yuan Z, Li WT, Ye XD, Zhu HY, Peng WJ (2014) Novel functional magnetic resonance imaging biomarkers for assessing response to therapy in hepatocellular carcinoma. Clin Transl Oncol 16:599–605
Lee SH, Hayano K, Zhu AX, Sahani DV, Yoshida H (2015) Water-exchange-modified kinetic parameters from dynamic contrast-enhanced MRI as prognostic biomarkers of survival in advanced hepatocellular carcinoma treated with antiangiogenic monotherapy. PLoS One 10, e0136725
Lee SH, Hayano K, Zhu AX, Sahani DV, Yoshida H (2015) Dynamic contrast-enhanced MRI kinetic parameters as prognostic biomarkers for prediction of survival of patient with advanced hepatocellular carcinoma: a pilot comparative study. Acad Radiol 22:1344–1360
Hsu CY, Shen YC, Yu CW et al (2011) Dynamic contrast-enhanced magnetic resonance imaging biomarkers predict survival and response in hepatocellular carcinoma patients treated with sorafenib and metronomic tegafur/uracil. J Hepatol 55:858–865
Mross K, Fasol U, Frost A et al (2009) DCE-MRI assessment of the effect of vandetanib on tumor vasculature in patients with advanced colorectal cancer and liver metastases: a randomized phase I study. J Angiogenes Res 1:5
Hsu C, Yang TS, Huo TI et al (2012) Vandetanib in patients with inoperable hepatocellular carcinoma: a phase II, randomized, double-blind, placebo-controlled study. J Hepatol 56:1097–1103
Kim KA, Park MS, Ji HJ et al (2014) Diffusion and perfusion MRI prediction of progression-free survival in patients with hepatocellular carcinoma treated with concurrent chemoradiotherapy. J Magn Reson Imaging 39:286–292
Hoff BA, Bhojani MS, Rudge J et al (2012) DCE and DW-MRI monitoring of vascular disruption following VEGF-Trap treatment of a rat glioma model. NMR Biomed 25:935–942
Moestue SA, Huuse EM, Lindholm EM et al (2013) Low-molecular contrast agent dynamic contrast-enhanced (DCE)-MRI and diffusion-weighted (DW)-MRI in early assessment of bevacizumab treatment in breast cancer xenografts. J Magn Reson Imaging 38:1043–1053
Gaustad JV, Simonsen TG, Smistad R, Wegner CS, Andersen LM, Rofstad EK (2015) Early effects of low dose bevacizumab treatment assessed by magnetic resonance imaging. BMC Cancer 15:900
Chen B-B, Hsu C-Y, Yu C-W et al (2016) Dynamic contrast-enhanced MR imaging of advanced hepatocellular carcinoma: comparison with the liver parenchyma and correlation with the survival of patients receiving systemic therapy. Radiology 281:454–464
Bokacheva L, Rusinek H, Chen Q et al (2007) Quantitative determination of Gd-DTPA concentration in T1-weighted MR renography studies. Magn Reson Med 57:1012–1018
Tofts PS, Brix G, Buckley DL et al (1999) Estimating kinetic parameters from dynamic contrast-enhanced T(1)-weighted MRI of a diffusable tracer: standardized quantities and symbols. J Magn Reson Imaging 10:223–232
Leach MO, Brindle KM, Evelhoch JL et al (2005) The assessment of antiangiogenic and antivascular therapies in early-stage clinical trials using magnetic resonance imaging: issues and recommendations. Br J Cancer 92:1599–1610
Morgan B, Thomas AL, Drevs J et al (2003) Dynamic contrast-enhanced magnetic resonance imaging as a biomarker for the pharmacological response of PTK787/ZK 222584, an inhibitor of the vascular endothelial growth factor receptor tyrosine kinases, in patients with advanced colorectal cancer and liver metastases: results from two phase I studies. J Clin Oncol 21:3955–3964
van Laarhoven HW, Rijpkema M, Punt CJ et al (2003) Method for quantitation of dynamic MRI contrast agent uptake in colorectal liver metastases. J Magn Reson Imaging 18:315–320
Jiang T, Zhu AX, Sahani DV (2013) Established and novel imaging biomarkers for assessing response to therapy in hepatocellular carcinoma. J Hepatol 58:169–177
Goh V, Padhani AR (2006) Imaging tumor angiogenesis: functional assessment using MDCT or MRI? Abdom Imaging 31:194–199
Sahani DV, Jiang T, Hayano K et al (2013) Magnetic resonance imaging biomarkers in hepatocellular carcinoma: association with response and circulating biomarkers after sunitinib therapy. J Hematol Oncol 6:51
Zhu AX, Holalkere NS, Muzikansky A, Horgan K, Sahani DV (2008) Early antiangiogenic activity of bevacizumab evaluated by computed tomography perfusion scan in patients with advanced hepatocellular carcinoma. Oncologist 13:120–125
Jiang T, Kambadakone A, Kulkarni NM, Zhu AX, Sahani DV (2012) Monitoring response to antiangiogenic treatment and predicting outcomes in advanced hepatocellular carcinoma using image biomarkers, CT perfusion, tumor density, and tumor size (RECIST). Investig Radiol 47:11–17
Yopp AC, Schwartz LH, Kemeny N et al (2011) Antiangiogenic therapy for primary liver cancer: correlation of changes in dynamic contrast-enhanced magnetic resonance imaging with tissue hypoxia markers and clinical response. Ann Surg Oncol 18:2192–2199
Jarnagin WR, Schwartz LH, Gultekin DH et al (2009) Regional chemotherapy for unresectable primary liver cancer: results of a phase II clinical trial and assessment of DCE-MRI as a biomarker of survival. Ann Oncol 20:1589–1595
Zhu AX, Sahani DV, Duda DG et al (2009) Efficacy, safety, and potential biomarkers of sunitinib monotherapy in advanced hepatocellular carcinoma: a phase II study. J Clin Oncol 27:3027–3035
Hayano K, Fuentes-Orrego JM, Sahani DV (2014) New approaches for precise response evaluation in hepatocellular carcinoma. World J Gastroenterol 20:3059–3068
Galbraith SM, Lodge MA, Taylor NJ et al (2002) Reproducibility of dynamic contrast-enhanced MRI in human muscle and tumours: comparison of quantitative and semi-quantitative analysis. NMR Biomed 15:132–142
Sahani DV, Holalkere NS, Mueller PR, Zhu AX (2007) Advanced hepatocellular carcinoma: CT perfusion of liver and tumor tissue—initial experience. Radiology 243:736–743
Donati OF, Do RK, Hötker AM et al (2015) Interreader and inter-test agreement in assessing treatment response following transarterial embolization for hepatocellular Carcinoma. Eur Radiol 25:2779–2788
Chapiro J, Duran R, Lin M et al (2015) Early survival prediction after intra-arterial therapies: a 3D quantitative MRI assessment of tumour response after TACE or radioembolization of colorectal cancer metastases to the liver. Eur Radiol 25:1993–2003
Acknowledgements
The scientific guarantor of this publication is Tiffany Ting-Fang Shih. The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article. This study has received funding by National Taiwan University Hospital (grant number NTUH. 101-001863); and the Ministry of Science and Technology, Executive Yuan, ROC, Taiwan (grant number NSC 100-2314-B-002-053). One of the authors has significant statistical expertise. No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was waived by the Institutional Review Board. Some study subjects or cohorts have been previously reported. All patients included in this study were associated with two other trials (Study 1— 54 patients in a phase II clinical trial using vandetanib, registered with ClinicalTrials.gov NCT00508001; Study 2—38 patients in a phase II clinical trial using sorafenib and tegafur/uracil, registered with ClinicalTrials.gov NCT00464919) conducted by our group.
In Study 1 with vandetanib, the previously published article [20] found that Ktrans did not differ significantly between the three treatment groups (group 1, vandetanib 300 mg/day; group 2, vandetanib 100 mg/day; group 3, placebo). Vascular response, defined as a greater than 30% reduction of Ktrans values from baseline after 7 days of study drug treatment, was not significantly different between the groups either. The follow-up duration was 12 months.
In Study 2 with sorafenib and tegafur/uracil, the previously published article [18] found that a vascular response, defined as a greater than 40% reduction of Ktrans values from baseline after 14 days of study drug treatment, correlated with longer progression-free survival and overall survival. Percentage of Ktrans change after treatment was an independent predictor of tumour response, progression-free survival, and overall survival. The follow-up duration was 140 months.
The association of baseline DCE-MRI parameters before treatment with overall survival outcome in these patients has been reported in our previous study [25]. The current study investigated the association of changes in six DCE-MRI parameters within 1 week following treatment with overall survival in these patients. The results of the current study have not been published previously.
Methodology: retrospective, diagnostic or prognostic study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, BB., Hsu, CY., Yu, CW. et al. Early perfusion changes within 1 week of systemic treatment measured by dynamic contrast-enhanced MRI may predict survival in patients with advanced hepatocellular carcinoma. Eur Radiol 27, 3069–3079 (2017). https://doi.org/10.1007/s00330-016-4670-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-016-4670-2