Abstract
Objectives
To assess the performance of a simplified MRI protocol consisting of a contrast-enhanced three-dimensional MR angiography (CE-MRA) in association with a post-contrast T1-weighted sequence (T1WIV) for the detection of HNPGLs in SDHx mutation carriers.
Methods
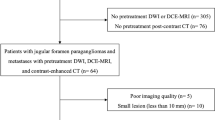
This retrospective sub-study is based on the multicenter PGL.EVA cohort, which prospectively enrolled SDHx mutation carriers from 2005 to 2009; 157 index cases or relatives were included. CE-MRA and the T1WIV images were read solely with knowledge of the clinical data but blind to the diagnosis. Sensitivity, specificity and likelihood ratios for the simplified MRI protocol were compared to the full MRI protocol reading results and to the gold standard status obtained through the consensus of an expert committee.
Results
The sensitivity and specificity of the readings of the simplified MRI protocol were, respectively, 88.7 % (95 % CI = 78.1–95.3) and 93.7 % (95 % CI = 86.8–97.7) versus 80.7 % (95 % CI = 68.6–89.6) and 94.7 % (95 % CI = 88.1–98.3) for the readings of the full MRI protocol.
Conclusions
The simplified post-contrast MRI with shorter duration (5 to 10 minutes) showed no performance difference compared to the lengthy standard full MRI and can be proposed for the detection of head and neck paragangliomas (HNPGLs) in SDHx mutation carriers.
Key Points
• Rapid angio-MRI protocol and the usual lengthy protocol show equal diagnostic performance.
• The CE-MRA is the key sequence for the detection of HNPGLs.
• The T1WIV sequence assists in localizing HNPGLs.




Similar content being viewed by others
Abbreviations
- HNPGL:
-
Head and neck paraganglioma
- CE-MRA:
-
Contrast-enhanced three-dimensional MR angiography
- T1WIV:
-
Post-contrast T1-weighted sequence
References
Mariman EC, van Beersum SE, Cremers CW et al (1993) Analysis of a second family with hereditary non-chromaffin paragangliomas locates the underlying gene at the proximal region of chromosome 11q. Hum Genet 91:357–361
Lack EE, Cubilla AL, Woodruff JM, Farr HW (1977) Paragangliomas of the head and neck region. A clinical study of 69 patients. Cancer 39:397–409
Castelblanco E, Gallel P, Ros S et al (2012) Thyroid paraganglioma. Report of 3 cases and description of an immunohistochemical profile useful in the differential diagnosis with medullary thyroid carcinoma, based on complementary DNA array results. Hum Pathol 43:1103–1112
Alkadhi H, Schuknecht B, Stoeckli SJ, Valavanis A (2002) Evaluation of topography and vascularization of cervical paragangliomas by magnetic resonance imaging and color duplex sonography. Neuroradiology 44:83–90
Olsen WL, Dillon WP, Kelly WM et al (1987) MR imaging of paragangliomas. AJR Am J Roentgenol 148:201–204
van Gils AP, van den Berg R, Falke TH et al (1994) MR diagnosis of paraganglioma of the head and neck: value of contrast enhancement. AJR Am J Roentgenol 162:147–153
Barber B, Ingram M, Khan S et al (2011) Clinicoradiological manifestations of paraganglioma syndromes associated with succinyl dehydrogenase enzyme mutation. Insights Imaging 2:431–438
Gimenez-Roqueplo A-P, Caumont-Prim A, Houzard C et al (2013) Imaging work-up for screening of paraganglioma and pheochromocytoma in SDHx mutation carriers: a multicenter prospective study from the PGL.EVA Investigators. J Clin Endocrinol Metab 98:E162–E173
Baysal BE, Willett-Brozick JE, Lawrence EC et al (2002) Prevalence of SDHB, SDHC, and SDHD germline mutations in clinic patients with head and neck paragangliomas. J Med Genet 39:178–183
Boedeker CC, Neumann HPH, Maier W et al (2007) Malignant head and neck paragangliomas in SDHB mutation carriers. Off J Am Acad Otolaryngol-Head Neck Surg 137:126–129
Drovdlic CM, Myers EN, Peters JA et al (2001) Proportion of heritable paraganglioma cases and associated clinical characteristics. Laryngoscope 111:1822–1827
Badenhop RF, Jansen JC, Fagan PA et al (2004) The prevalence of SDHB, SDHC, and SDHD mutations in patients with head and neck paraganglioma and association of mutations with clinical features. J Med Genet 41:e99
Favier J, Amar L, Gimenez-Roqueplo A-P (2014) Paraganglioma and phaeochromocytoma: from genetics to personalized medicine. Nat Rev Endocrinol. doi:10.1038/nrendo.2014.188
Favier J, Gimenez-Roqueplo A-P (2010) Pheochromocytomas: the (pseudo)-hypoxia hypothesis. Best Pract Res Clin Endocrinol Metab 24:957–968
Burnichon N, Rohmer V, Amar L et al (2009) The succinate dehydrogenase genetic testing in a large prospective series of patients with paragangliomas. J Clin Endocrinol Metab 94:2817–2827
Neumann HPH, Pawlu C, Peczkowska M et al (2004) Distinct clinical features of paraganglioma syndromes associated with SDHB and SDHD gene mutations. JAMA J Am Med Assoc 292:943–951
Timmers HJLM, Kozupa A, Eisenhofer G et al (2007) Clinical presentations, biochemical phenotypes, and genotype-phenotype correlations in patients with succinate dehydrogenase subunit B-associated pheochromocytomas and paragangliomas. J Clin Endocrinol Metab 92:779–786
Brewis C, Bottrill ID, Wharton SB, Moffat DA (2000) Metastases from glomus jugulare tumours. J Laryngol Otol 114:17–23
Lenders JWM, Duh Q-Y, Eisenhofer G et al (2014) Pheochromocytoma and paraganglioma: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 99:1915–1942
Martin TPC, Irving RM, Maher ER (2007) The genetics of paragangliomas: a review. Clin Otolaryngol Off J ENT-UK Off J Neth Soc Oto-Rhino-Laryngol Cervico-Facial Surg 32:7–11
Makeieff M, Raingeard I, Alric P et al (2008) Surgical management of carotid body tumors. Ann Surg Oncol 15:2180–2186
Paris J, Facon F, Thomassin JM, Zanaret M (2006) Cervical paragangliomas: neurovascular surgical risk and therapeutic management. Eur Arch Oto-Rhino-Laryngol Off J Eur Fed Oto-Rhino-Laryngol Soc EUFOS Affil Ger Soc Oto-Rhino-Laryngol - Head Neck Surg 263:860–865
Huy PTB, Kania R, Duet M et al (2009) Evolving concepts in the management of jugular paraganglioma: a comparison of radiotherapy and surgery in 88 cases. Skull Base Off J North Am Skull Base Soc Al 19:83–91
Dupin C, Lang P, Dessard-Diana B et al (2014) Treatment of head and neck paragangliomas with external beam radiation therapy. Int J Radiat Oncol Biol Phys 89:353–359
Jansen JC, van den Berg R, Kuiper A et al (2000) Estimation of growth rate in patients with head and neck paragangliomas influences the treatment proposal. Cancer 88:2811–2816
Vogl T, Brüning R, Schedel H et al (1989) Paragangliomas of the jugular bulb and carotid body: MR imaging with short sequences and Gd-DTPA enhancement. AJR Am J Roentgenol 153:583–587
Timmers HJLM, Chen CC, Carrasquillo JA et al (2012) Staging and functional characterization of pheochromocytoma and paraganglioma by 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography. J Natl Cancer Inst 104:700–708
Neves F, Huwart L, Jourdan G et al (2008) Head and neck paragangliomas: value of contrast-enhanced 3D MR angiography. AJNR Am J Neuroradiol 29:883–889
van den Berg R, Schepers A, de Bruïne FT et al (2004) The value of MR angiography techniques in the detection of head and neck paragangliomas. Eur J Radiol 52:240–245
Jasperson KW, Kohlmann W, Gammon A et al (2014) Role of rapid sequence whole-body MRI screening in SDH-associated hereditary paraganglioma families. Fam Cancer 13:257–265
Hoegerle S, Ghanem N, Altehoefer C et al (2003) 18F-DOPA positron emission tomography for the detection of glomus tumours. Eur J Nucl Med Mol Imaging 30:689–694
King KS, Chen CC, Alexopoulos DK et al (2011) Functional imaging of SDHx-related head and neck paragangliomas: comparison of 18F-fluorodihydroxyphenylalanine, 18F-fluorodopamine, 18F-fluoro-2-deoxy-D-glucose PET, 123I-metaiodobenzylguanidine scintigraphy, and 111In-pentetreotide scintigraphy. J Clin Endocrinol Metab 96:2779–2785
Acknowledgments
Authors would like to thank Steven M. Yevich for editing the English text. The scientific guarantor of this publication is Prof. Philippe Halimi. The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article. This study has received funding by Program Hospitalier National de Recherche Clinique 2004 (PCR05007).
One of the authors has significant statistical expertise: Aurore Caumont-Prim from Assistance Publique-Hôpitaux de Paris, Hôpital Européen Georges Pompidou, Unité d’Épidémiologie et de Recherche Clinique, F-75015 Paris, France. Institutional Review Board approval was obtained. Written informed consent was obtained from all subjects (patients) in this study. Some study subjects or cohorts have been previously reported in: Gimenez-Roqueplo A-P, Caumont-Prim A, Houzard C, et al. (2013) Imaging work-up for screening of paraganglioma and pheochromocytoma in SDHx mutation carriers: a multicenter prospective study from the PGL.EVA Investigators. J Clin Endocrinol Metab 98:E162–173. Methodology: retrospective, diagnostic study, multicenter study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Anne-Paule Gimenez-Roqueplo and Philippe Halimi jointly directed this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 57 kb)
Rights and permissions
About this article
Cite this article
Gravel, G., Niccoli, P., Rohmer, V. et al. The value of a rapid contrast-enhanced angio-MRI protocol in the detection of head and neck paragangliomas in SDHx mutations carriers: a retrospective study on behalf of the PGL.EVA investigators*. Eur Radiol 26, 1696–1704 (2016). https://doi.org/10.1007/s00330-015-4024-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-015-4024-5