Abstract
Key message
Transgenic Populus alba over-expressing a TIP aquaporin ( aqua1) showed a higher growth rate under Zn excess, suggesting that aqua1 could be involved in water homeostasis, rather than in Zn homeostasis.
Abstract
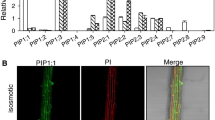
Populus is the internationally accepted model for physiological and developmental studies of tree traits under stress. In plants, aquaporins facilitate and regulate the diffusion of water, however, few poplar aquaporins have been characterized to date. In this study, we reported for the first time an in vivo characterization of Populus alba clone Villafranca transgenic plants over-expressing a TIP aquaporin (aqua1) of P. x euramericana clone I-214. An AQUA1:GFP chimeric construct, over-expressed in P. alba Villafranca clones, shows a cytoplasmic localization in roots, and it localizes in guard cells in leaves. When over-expressed in transgenic plants, aqua1 confers a higher growth rate compared to wild-type (wt) plants, without affecting chlorophyll accumulation, relative water content (RWC), and fluorescence performances, but increasing the intrinsic Transpiration Efficiency. In response to Zn (1 mM), transgenic lines did not show a significant increase in Zn accumulation as compared to wt plants, even though the over-expression of this gene confers higher tolerance in root tissues. These results suggest that, in poplar plants, this gene could be principally involved in regulation of water homeostasis and biomass production, rather than in Zn homeostasis.









Similar content being viewed by others
References
Adams JP, Adeli A, Hsu CY, Harkess RL, Page GP, dePamphilis CW, Schultz EB, Yuceer C (2011) Poplar maintains zinc homeostasis with a heavy metal genes HMA4 and PCS1. J Exp Bot 62:3737–3752
Agree P, Saboori AM, Asimos A, Smith BL (1987) Purification and partial characterization of the Mt 30,000 integral membrane protein associated with the erythrocyte Rh(D) antigen. J Biol Chem 262:17497–17503
Aharon R, Shahak S, Wininger S, Bendov R, Kapulnik Y, Galili G (2003) Overexpression of a plasma membrane aquaporin in transgenic tobacco improves plant vigour under favourable growth conditions but not under drought or salt stress. Plant Cell 15:439–447
Andreucci A, Ariani A, Sebastiani L (2015) In vitro functional characterization of AQUA1: a new poplar (Populus x euramericana clone I-214) aquaporin involved in Zinc stress. International Plant Science Conferences (IPSC) from nature to Technological Exploitations. Florence 2–5 September 2014. Abstract p xviii
Ariani A, Gepts P (2015) Genome-wide identification and characterization of aquaporin gene family in common bean (Phaseolus vulgaris L.). Mol Genet Genomics. doi:10.1007/s00438-015-1038-2
Arnon DI, Hoagland DR (1940) Crop production in artificial culture solutions and in soils with special reference to factors influencing yields and absorption of inorganic nutrients. Soil Sci 50:463–485
Bae EK, Lee H, Lee JS, Noh EW (2011) Drought, salt and wounding stress induce the expression of the plasma membrane intrinsic protein 1 gene in poplar (Populus alba × P. tremula var. glandulosa). Gene 483:43–48
Barceló J, Poschenrieder C, Andreu I, Gunse B (1986) Cadmium-induced decrease of water stress resistance in bush bean plants (Phaseolus vulgaris L. cv. Contender) I. Effects of Cd on water potential, relative water content and cell wall elasticity. J Plant Physiol 125:17–25
Besse M, Knipfer T, Miller AJ, Verdeil J-L, Jahn TP et al (2011) Developmental pattern of aquaporin expression in barley (Hordeum vulgare L.) leaves. J Exp Bot 62:4127–4142
Brunner AM, Yakovlev IA, Strauss SH (2004) Validating internal controls for quantitative plant gene expression studies. BMC Plant Biol 4:14. doi:10.1186/1471-2229-4-14
Chaumont F, Barrieu F, Wojcik E, Chrispeels MJ, Jung R (2001) Aquaporins constitute a large and highly divergent protein family in maize. Plant Physiol 125:1206–1215
Cohen D, Bogeat-Triboulot MB, Vialet-Chabrand S, Merret R, Courty PE, Moretti S, Bizet F, Guilliot A, Hummel I (2013) Developmental and environmental regulation of aquaporin gene expression across Populus species: divergence or redundancy? PLoS One 8:e55506
Confalonieri M, Belenghi B, Balestrazzi A, Negri S, Facciotto G, Schenone G, Delledonne M (2000) Transformation of elite white poplar (Populus alba L.) cv. ‘Villafranca’ and evaluation of herbicide resistance. Plant Cell Rep 19:978–982
Cui XH, Hao FS, Chen H, Chen J, Wang XC (2008) Expression of the Vicia faba VfPIP1 gene in Arabidopsis thaliana plants improves their drought resistance. J Plant Res 121:207–214
Di Baccio D, Tognetti R, Minnoci A, Sebastiani L (2009) Responses of the Populus × euramericana clone I-214 to excess zinc:carbon assimilation, structural modifications, metal distribution and cellular localization. Environ Exp Bot 67:153–163
Di Baccio D, Galla G, Bracci T, Andreucci A, Barcaccia G, Tognetti R, Sebastiani L (2011) Transcriptome analyses of Populus × euramericana clone I-214 leaves exposed to excess zinc. Tree Physiol 31:1293–1308
Dickmann DI (1972) Photosynthesis and respiration by developing leaves of cottonwood (Populus deltoids Bartr.). Bot Gaz 132:253–259
Du J, Miura E, Robischon M, Martinez C, Groover A (2011) The Populus class II HD ZIP transcription factor POPCORONA affects cell differentiation during secondary growth on woody stems. PLoS One 6:e17458
Flexas J, Ribas-Carbó M, Hanson DT, Bota J, Otto B, Cifre J, McDowell N, Medrano H, Kaldenh R (2006) Tobacco aquaporin NtAQP1 is involved in mesophyll conductance to CO2 in vivo. Plant J 48:427–439
Francini A, Nali C, Picchi V, Lorenzini G (2007) Metabolic changes in white clover clones exposed to ozone. Env Exp Bot 60:11–19
Gupt AB, Sankararamakrishnan R (2009) Genome wide analysis of major intrinsic proteins in the tree plant Populus trichocarpa: characterization of XIP subfamily of aquaporins from evolutionary perspective. BMC Plant Biol 9:134
Hacke UG, Plavcova L, Almeida-Rodriguez A, King-Jones S, Zhou WC et al (2010) Influence of nitrogen fertilization on xylem traits and aquaporin expression in stems of hybrid poplar. Tree Physiol 30:1016–1025
Hanba YT, Shibasaka M, Hayashi Y, Hayakawa T, Kasamo K, Terashima I, Katsuhara M (2004) Overexpression of the barley aquaporin HvPIP2;1 increase internal CO2 conductance and CO2 assimilation in the leaves of transgenic rice plants. Plant Cell Physiol 45:521–529
Heckwolf M, Pate DT, Keldenhoff R (2011) The Arabidopsis thaliana aquaporin AtPIP1;2 is a physiological relevant CO2 transport facilitator. Plant J 67:795–804
Heinein RB, Ye Q, Chaumont F (2009) Role of aquaporins in leaf physiology. J Exp Bot 60:2971–2985
Hove RM, Bhave M (2011) Plant aquaporins with non-aqua functions: deciphering the signature sequences. Plant Mol Biol 75:413–430
Hunt R (1978) Plant growth analysis. Camelot Press Ltd, Southampton
Kjeldahl J (1883) Neue methode zur bestimmung des stickstoffs in organischen körpern (New method for the determination of nitrogen in organic substances). Z Anal Chem 22(1):366–383
Kohler A, Delaruelle C, Martin D, Encelot N, Martin F (2003) The poplar root transcriptome: analysis of 7000 expressed sequence tags. FEBS Lett 542:37–41
Lee SH, Chung GC, Jang JY, Ahn SJ, Zwiazek JJ (2012) Overexpression of PIP2;5 aquaporin alleviates effects of low root temperature on cell hydraulic conductivity and growth in Arabidopsis. Plant Physiol 159:479–488
Li GW, Zhang MH, Cai WM, Sun WN, Su WA (2008) Characterization of OsPIP2;7, a water channel protein in rice. Plant Cell Physiol 49(12):1851–1858. doi:10.1093/pcp/pcn166
Li G, Santoni V, Maurel C (2013) Plant aquaporins: roles in plant physiology. Biochim Biophys Acta. doi:10.1016/j.bbagen.2013.11.004
Lloyd G, McCown B (1981) Commercially-feasible micropropagation of Mountain laurel, Kolmia latifolia, by use of shoot tip culture. Inter Plant Propagat Soc 30:421–427
Lopez D, Bronner G, Brunel N, Auguin D, Bourgerie S et al (2012) Insights into Populus XIP aquaporins: evolutionary expansion, protein functionality, and environmental regulation. J Exp Bot 63:2217–2230
Ludevid D, Höfte H, Himelblau E, Chrispeels MJ (1992) The expression pattern of the tonoplastic intrinsic protein γ-TIP in Arabidopsis thaliana is correlated with cell enlargement. Plant Physiol 100:1633–1639
Marjanovic Z, Uehlein N, Kaldenhoff R, Zwiazek JJ, Weiss M et al (2005) Aquaporins in poplar: what a difference a symbiont makes! Planta 222:258–268
Matsumoto T, Hian HL, Su WA, Tanaka D, Liu C, Iwasaki I, Kitagawa Y (2009) Role of the aquaporin PIP1 subfamily in the chilling tolerance of rice. Plant Cell Physiol 50:216–229
Maurel C (1997) Aquaporins and water permeability of plant membranes. Annu Rev Plant Physiol Plant Mol Biol 48:399–429
Maurel C, Verdoucq L, Luu DT, Santoni V (2008) Plant aquaporins: membrane channels with multiple integrated functions. Ann Rev Plant Biol 59:595–624
Nakagawa T, Nakamura S, Tanaka K, Kawamukai M, Suzuki T, Nakamura K, Kimura T, Ishiguro S (2008) Development of R4 gateway binary vectors (R4pGWB) enabling high-throughput promoter swapping for plant research. Biosci Biotechnol Biochem 72:624–629
Negishi T, Oshima K, Hattori M, Kanai M, Mano S, Nishimura M, Yoshida K (2012) Tonoplast- and plasma membrane-localized aquaporin-family transporters in blue hydrangea sepals of aluminum hyperaccumulating plant. PLoS One 7:e43189
Pandey N, Sharma CP (2002) Effect of heavy metals Co2+, Ni2+ and Cd2+ on growth and metabolism of cabbage. Plant Sci 163:753–758
Peng Y, Lin W, Cai W, Arora R (2007) Over-expression of a Panax ginseng tonoplast aquaporin alters salt tolerance, drought tolerance and cold acclimation ability in transgenic Arabidopsis plants. Planta 226:729–740
Perrone I, Gambino G, Chitarra W, Vitali M, Pagliarini C et al (2012) The grapevine root-specific aquaporin VvPIP2;4N control root hydraulic conductance and leaf gas exchange under well-watered conditions but not under water stress. Plant Physiol 160:956–977
Philips AL, Huttly AK (1994) Cloning of two gibberellin-regulated cDNAs from Arabidopsis thaliana by subtractive hybridization: expression of the tonoplast water channel, γ-TIP, is increased by GA3. Plant Mol Biol 24:603–615
Postaire O, Tornaire-Roux C, Grondin A, Boursiac Y, Morillon R, Schäffner AR, Maurel C (2010) A PIP1 aquaporin contributes to hydrostatic pressure-induced water transport in both the root and rosette of Arabidopsis. Plant Physionl 152:1418–1430
Prasad MNV (1997) Trace metals. In: Prasad MNV (ed) Plant ecophysiology. New York, USA, pp 207–249
Quigley F, Rosenberg JM, Shachar-Hill Y, Bohnert HJ (2001) From genome to function: the Arabidopsis aquaporins. Genome Biol 3:1–17
Robinschon M, Du J, Miura E, Groover A (2011) The Populus class II HD ZIP, popREVOLUTA, influence cambium initiation and patterning of woody stems. Plant Physiol 155:1214–1225
Rodgers-Melnick E, Mane SP, Dharmawardhana P, Slavov GT, Crasta OR et al (2012) Contrasting patterns of evolution following whole genome versus tandem duplication events in Populus. Genom Res 22:95–105
Roelfsema MRG, Hedrich R (2005) In the light of stomatal opening: new insight into ‘the Watergate’. New Phytol 167:665–691
Romeo S, Francini A, Ariani A, Sebastiani L (2014a) Phytoremediation of Zn: identify the diverging resistance, uptake and biomass production behaviours of poplar clones under high zinc stress. Water Air Soil Pollut. doi:10.1007/s11270-013-1813-9
Romeo S, Trupiano D, Ariani A, Renzone G, Scippa GS, Scaloni A, Sebastiani L (2014b) Proteomic analysis of Populus × euramericana (clone I-214) roots to identify key factors involved in zinc stress response. J Plant Physiol 171:1054–1063
Romeo S, Lombardi L, Andreucci A, Lux A, Stephen Horner D, Sebastiani L (2015) RNA sequencing of Populus x canadensis roots identifies key molecular mechanisms underlying physiological adaption to excess zinc. PloS One. doi:10.1371/journal.pone.0117571
Sade N, Vinocur BJ, Diber A, Shatil A, Ronen G, Rissan H, Wallach R, Karchi H, Moshelion M (2009) Improving plant stress tolerance and yield production: is the tonoplast aquaporin SlTIP2;2 a key to isohydric to anisohydric conversion? New Phytol 181:651–661
Sakurai J, Ishikawa F, Yamaguchi T, Uemure M, Maeshima M (2005) Identification of 33 rice aquaporin genes and analysis of their expression and function. Plant Cell Physiol 26:1568–1577
Sarda X, Tousch D, Ferrare K, Legrand E, Dupuis JM, Casse-Delbart F, Lamaze T (1997) Two TIP-like genes encoding aquaporins are expressed in sunflower guard cells. The Plant J 12:1103–1111
Secchi F, Zwieniecki MA (2010) Patterns of PIP gene expression in Populus trichocarpa during recovery from xylem embolism suggest a major role for the PIP1 aquaporin subfamily as moderators of refilling process. Plant Cell Environ 33:1285–1297
Shope JC, Mott KA (2006) Membrane trafficking and osmotically induced volume changes in guard cells. J Exp Bot 57:4123–4413
Sinclair SA, Krämer U (2012) The zinc homeostasis network of land plantRoes. Biochim Biophys Acta 1823:1553–1567
Song J, Lu S, Chen ZZ, Lourenco R, Chiang VL (2006) Genetic transformation of Populus trichocarpa genotype Nisqually-1: a functional genomic tool for woody plants. Plant Cell Physiol 47:1582–1589
Su X, Chu Y, Li H, Hou Y, Zhang B et al (2011) Expression of multiple resistance genes enhances tolerance to environmental stressors in transgenic poplar (Populus x euramericana ‘Guariento’). PLoS One 6:e24614
Tuskan GA, Difazio S, Jansson S, Bohlmann J, Grigoriev I et al (2006) The genome of black cottonwood, Populus trichocarpa (Torr. & Gray). Science 313:1596–1604
Uehlein N, Lovisolo C, Siefritz F, Kaldenhoff R (2003) The tobacco aquaporin ntAQP1 is a membrane CO2 pore with physiological functions. Nature 425:734–737
Wilkins DA (1957) A technique for the measurement of lead tolerance in plants. Nature 180:37–38
Wullschleger SD, Weston DJ, DiFazio SP, Tuskan GA (2012) Revisiting the sequencing of the first tree genome: populus trichocarpa. Tree Physiol 33:357–364
Zhang Y, Ma K, Sadana P, Chowdhury F, Gaillard S, Wang F, McDonnell DP, Unterman TG, Elam MB, Park EA (2006) Estrogen related receptors stimulate pyruvate dehydrogenase kinase isoform 4 (PDK4) gene expression. J Biol Chem 281(52):39897–39906
Zhang Y, Wang Z, Chai T, Wen Z, Zhang H (2008) Indian mustard aquaporin improves drought and heavy-metal resistance in tobacco. Mol Biotechnol 40:280–292
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by P. Lakshmanan.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ariani, A., Francini, A., Andreucci, A. et al. Over-expression of AQUA1 in Populus alba Villafranca clone increases relative growth rate and water use efficiency, under Zn excess condition. Plant Cell Rep 35, 289–301 (2016). https://doi.org/10.1007/s00299-015-1883-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-015-1883-9