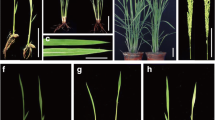
Abstract
Key message
Mutation of the AM1 gene causes an albino midrib phenotype and enhances tolerance to drought in rice
Abstract
K+ efflux antiporter (KEA) genes encode putative potassium efflux antiporters that are mainly located in plastid-containing organisms, ranging from lower green algae to higher flowering plants. However, little genetic evidence has been provided on the functions of KEA in chloroplast development. In this study, we isolated a rice mutant, albino midrib 1 (am1), with green- and white-variegation in the first few leaves, and albino midrib phenotype in older tissues. We found that AM1 encoded a putative KEA in chloroplast. AM1 was highly expressed in leaves, while lowly in roots. Chloroplast gene expression and proteins accumulation were affected during chlorophyll biosynthesis and photosynthesis in am1 mutants. Interestingly, AM1 was induced by salt and PEG, and am1 showed enhanced sensitivity to salinity in seed germination and increased tolerance to drought. Taken together, we concluded that KEAs were involved in chloroplast development and played important roles in drought tolerance.








Similar content being viewed by others
Abbreviations
- Car:
-
Carotenoid
- Chl:
-
Chlorophyll
- KEA:
-
Potassium (K+) efflux antiporter
- ORF:
-
Open reading frame
- DAB:
-
Diaminobenzidine
- GFP:
-
Green fluorescent protein
References
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. doi:10.1146/annurev.arplant.55.031903.141701
Aranda-Sicilia MN, Cagnac O, Chanroj S, Sze H, Rodriguez-Rosales MP, Venema K (2012) Arabidopsis KEA2, a homolog of bacterial KefC, encodes a K(+)/H(+) antiporter with a chloroplast transit peptide. Biochim Biophys Acta 1818(9):2362–2371. doi:10.1016/j.bbamem.2012.04.011
Archer EK, Bonnett HT (1987) Characterization of a virescent chloroplast mutant of tobacco. Plant Physiol 83(4):920–925
Arnon DI (1949) Copper enzymes in isolated chloroplasts polyphenoloxidase in beta vulgaris. Plant Physiol 24(1):1–15
Booth IR (2003) Bacterial ion channels. Genet Eng (N Y) 25:91–111
Brautigam A, Weber AP (2009) Proteomic analysis of the proplastid envelope membrane provides novel insights into small molecule and protein transport across proplastid membranes. Mol Plant 2(6):1247–1261. doi:10.1093/mp/ssp070
Bright J, Desikan R, Hancock JT, Weir IS, Neill SJ (2006) ABA-induced NO generation and stomatal closure in Arabidopsis are dependent on H2O2 synthesis. Plant J 45(1):113–122. doi:10.1111/j.1365-313X.2005.02615.x
Carraretto L, Formentin E, Teardo E, Checchetto V, Tomizioli M, Morosinotto T, Giacometti GM, Finazzi G, Szabo I (2013) A thylakoid-located two-pore K+ channel controls photosynthetic light utilization in plants. Science 342(6154):114–118. doi:10.1126/science.1242113
Chanroj S, Wang G, Venema K, Zhang MW, Delwiche CF, Sze H (2012) Conserved and diversified gene families of monovalent cation/H(+) antiporters from algae to flowering plants. Front Plant Sci 3:25. doi:10.3389/fpls.2012.00025
Chen S, Tao L, Zeng L, Vega-Sanchez ME, Umemura K, Wang GL (2006) A highly efficient transient protoplast system for analyzing defence gene expression and protein–protein interactions in rice. Mol Plant Pathol 7(5):417–427. doi:10.1111/j.1364-3703.2006.00346.x
Choe S (2002) Potassium channel structures. Nat Rev Neurosci 3(2):115–121. doi:10.1038/nrn727
Dong H, Fei GL, Wu CY, Wu FQ, Sun YY, Chen MJ, Ren YL, Zhou KN, Cheng ZJ, Wang JL, Jiang L, Zhang X, Guo XP, Lei CL, Su N, Wang H, Wan JM (2013) A rice virescent-yellow leaf mutant reveals new insights into the role and assembly of plastid caseinolytic protease in higher plants. Plant Physiol 162(4):1867–1880. doi:10.1104/pp.113.217604
Emanuelsson O, Nielsen H, von Heijne G (1999) ChloroP, a neural network-based method for predicting chloroplast transit peptides and their cleavage sites. Protein Sci 8(5):978–984. doi:10.1110/ps.8.5.978
Fambrini M, Pugliesi C, Vernieri P, Giuliano G, Baroncelli S (1993) Characterization of a sunflower (Helianthus annuus L.) mutant, deficient in carotenoid synthesis and abscisic-acid content, induced by in vitro tissue culture. Theor Appl Genet 87(1–2):65–69. doi:10.1007/BF00223746
Ferro M, Brugiere S, Salvi D, Seigneurin-Berny D, Court M, Moyet L, Ramus C, Miras S, Mellal M, Le Gall S, Kieffer-Jaquinod S, Bruley C, Garin J, Joyard J, Masselon C, Rolland N (2010) AT_CHLORO, a comprehensive chloroplast proteome database with subplastidial localization and curated information on envelope proteins. Mol Cell Proteomics 9(6):1063–1084. doi:10.1074/mcp.M900325-MCP200
Finn RD, Mistry J, Tate J, Coggill P, Heger A, Pollington JE, Gavin OL, Gunasekaran P, Ceric G, Forslund K, Holm L, Sonnhammer EL, Eddy SR, Bateman A (2010) The Pfam protein families database. Nucleic Acids Res 38:D211–D222. doi:10.1093/nar/gkp985
Hamilton DW, Hills A, Kohler B, Blatt MR (2000) Ca2+ channels at the plasma membrane of stomatal guard cells are activated by hyperpolarization and abscisic acid. Proc Natl Acad Sci USA 97(9):4967–4972. doi:10.1073/pnas.080068897
Hiei Y, Ohta S, Komari T, Kumashiro T (1994) Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J 6(2):271–282
Holtorf H, Apel K (1996) Transcripts of the two NADPH protochlorophyllide oxidereductase genes PorA and PorB are differentially degraded in etiolated barley seedlings. Plant Mol Biol 31(2):387–392
Huang XY, Chao DY, Gao JP, Zhu MZ, Shi M, Lin HX (2009) A previously unknown zinc finger protein, DST, regulates drought and salt tolerance in rice via stomatal aperture control. Genes Dev 23(15):1805–1817. doi:10.1101/gad.1812409
Hwang SR, Tabita FR (1989) Cloning and expression of the chloroplast-encoded rbcL and rbcS genes from the marine diatom Cylindrotheca sp. strain N1. Plant Mol Biol 13(1):69–79
Jiang Y, Pico A, Cadene M, Chait BT, MacKinnon R (2001) Structure of the RCK domain from the E. coli K+ channel and demonstration of its presence in the human BK channel. Neuron 29(3):593–601
Jung KH, Hur J, Ryu CH, Choi Y, Chung YY, Miyao A, Hirochika H, An G (2003) Characterization of a rice chlorophyll-deficient mutant using the T-DNA gene-trap system. Plant Cell Physiol 44(5):463–472
Kohler B, Hills A, Blatt MR (2003) Control of guard cell ion channels by hydrogen peroxide and abscisic acid indicates their action through alternate signaling pathways. Plant Physiol 131(2):385–388. doi:10.1104/pp.016014
Kumar AM, Soll D (2000) Antisense HEMA1 RNA expression inhibits heme and chlorophyll biosynthesis in Arabidopsis. Plant Physiol 122(1):49–56
Kusumi K, Sakata C, Nakamura T, Kawasaki S, Yoshimura A, Iba K (2011) A plastid protein NUS1 is essential for build-up of the genetic system for early chloroplast development under cold stress conditions. Plant J 68(6):1039–1050. doi:10.1111/j.1365-313X.2011.04755.x
Maser P, Thomine S, Schroeder JI, Ward JM, Hirschi K, Sze H, Talke IN, Amtmann A, Maathuis FJ, Sanders D, Harper JF, Tchieu J, Gribskov M, Persans MW, Salt DE, Kim SA, Guerinot ML (2001) Phylogenetic relationships within cation transporter families of Arabidopsis. Plant Physiol 126(4):1646–1667
Matsuoka M (1990) Classification and characterization of cDNA that encodes the light-harvesting chlorophyll a/b binding protein of photosystem II from rice. Plant Cell Physiol 31(4):519–526
McAinsh MR, Clayton H, Mansfield TA, Hetherington AM (1996) Changes in stomatal behavior and guard cell cytosolic free calcium in response to oxidative stress. Plant Physiol 111(4):1031–1042
Rodermel S (2001) Pathways of plastid-to-nucleus signaling. Trends Plant Sci 6(10):471–478
Roosild TP, Miller S, Booth IR, Choe S (2002) A mechanism of regulating transmembrane potassium flux through a ligand-mediated conformational switch. Cell 109(6):781–791
Seo M, Koshiba T (2002) Complex regulation of ABA biosynthesis in plants. Trends Plant Sci 7(1):41–48
Song CP, Guo Y, Qiu Q, Lambert G, Galbraith DW, Jagendorf A, Zhu JK (2004) A probable Na+ (K+)/H+ exchanger on the chloroplast envelope functions in pH homeostasis and chloroplast development in Arabidopsis thaliana. Proc Natl Acad Sci USA 101(27):10211–10216. doi:10.1073/pnas.0403709101
Su N, Hu ML, Wu DX, Wu FQ, Fei GL, Lan Y, Chen XL, Shu XL, Zhang X, Guo XP, Cheng ZJ, Lei CL, Qi CK, Jiang L, Wang H, Wan JM (2012) Disruption of a rice pentatricopeptide repeat protein causes a seedling-specific albino phenotype and its utilization to enhance seed purity in hybrid rice production. Plant Physiol 159(1):227–238. doi:10.1104/pp.112.195081
Sugimoto H, Kusumi K, Tozawa Y, Yazaki J, Kishimoto N, Kikuchi S, Iba K (2004) The virescent-2 mutation inhibits translation of plastid transcripts for the plastid genetic system at an early stage of chloroplast differentiation. Plant Cell Physiol 45(8):985–996. doi:10.1093/pcp/pch111
Sugimoto H, Kusumi K, Noguchi K, Yano M, Yoshimura A, Iba K (2007) The rice nuclear gene, VIRESCENT 2, is essential for chloroplast development and encodes a novel type of guanylate kinase targeted to plastids and mitochondria. Plant J 52(3):512–527. doi:10.1111/j.1365-313X.2007.03251.x
ThordalChristensen H, Zhang Z, Wei Y, Collinge D (1997) Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley–powdery mildew interaction. Plant J 11(6):1187–1194
Wu Z, Zhang X, He B, Diao L, Sheng S, Wang J, Guo X, Su N, Wang L, Jiang L, Wang C, Zhai H, Wan J (2007) A chlorophyll-deficient rice mutant with impaired chlorophyllide esterification in chlorophyll biosynthesis. Plant Physiol 145(1):29–40. doi:10.1104/pp.107.100321
Yoo SC, Cho SH, Sugimoto H, Li J, Kusumi K, Koh HJ, Iba K, Paek NC (2009) Rice virescent3 and stripe1 encoding the large and small subunits of ribonucleotide reductase are required for chloroplast biogenesis during early leaf development. Plant Physiol 150(1):388–401. doi:10.1104/pp.109.136648
Zhang X, Zhang L, Dong F, Gao J, Galbraith DW, Song CP (2001) Hydrogen peroxide is involved in abscisic acid-induced stomatal closure in Vicia faba. Plant Physiol 126(4):1438–1448
Zhang H, Li J, Yoo JH, Yoo SC, Cho SH, Koh HJ, Seo HS, Paek NC (2006) Rice Chlorina-1 and Chlorina-9 encode ChlD and ChlI subunits of Mg-chelatase, a key enzyme for chlorophyll synthesis and chloroplast development. Plant Mol Biol 62(3):325–337. doi:10.1007/s11103-006-9024-z
Zhou K, Ren Y, Lv J, Wang Y, Liu F, Zhou F, Zhao S, Chen S, Peng C, Zhang X, Guo X, Cheng Z, Wang J, Wu F, Jiang L, Wan J (2013) Young leaf chlorosis 1, a chloroplast-localized gene required for chlorophyll and lutein accumulation during early leaf development in rice. Planta 237(1):279–292. doi:10.1007/s00425-012-1756-1
Zimmermann P, Hirsch-Hoffmann M, Hennig L, Gruissem W (2004) Genevestigator. Arabidopsis microarray database and analysis toolbox. Plant Physiol 136(1):2621–2632. doi:10.1104/pp.104.046367
Zybailov B, Rutschow H, Friso G, Rudella A, Emanuelsson O, Sun Q, van Wijk KJ (2008) Sorting signals, N-terminal modifications and abundance of the chloroplast proteome. PLoS One 3(4):e1994. doi:10.1371/journal.pone.0001994
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31000667), National Transgenic Science and Technology Program (2011ZX08009 003), and the Major State Basic Research Development Program of China (973 Program, 2009CB8500).
Conflict of interest
The authors have declared that no conflict of interests exist.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jeong Sheop Shin.
P. Sheng and J. Tan contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sheng, P., Tan, J., Jin, M. et al. Albino midrib 1, encoding a putative potassium efflux antiporter, affects chloroplast development and drought tolerance in rice. Plant Cell Rep 33, 1581–1594 (2014). https://doi.org/10.1007/s00299-014-1639-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-014-1639-y