Abstract
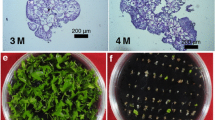
We report here the transformation of two species of orchid, Dendrobium phalaenopsis and D. nobile, by biolistic bombardment. Calli or protocorm-like bodies (PLBs) were used as target explants. Gold particles (1.0 µm) coated with plasmid DNA (pCAMBIA1301) encoding an intron-containing β-glucuronidase gene (gus-int) and a hygromycin phosphotransferase (hpt) gene were introduced into the PLBs or calli using the Bio-Rad PDS-1000/He Biolistic Particle Delivery System. Calli and PLBs were then chopped up and pre-cultured in 1/2-strength MS medium supplemented with 0.4 M mannitol for a 1-h osmoticum treatment before bombardment. Immediately after bombardment, the calli and PLBs were transferred to 1/2-strength MS medium without mannitol for recovery. Putatively transformed plantlets were obtained by selection and regeneration on medium supplemented with 30 mg/l hygromycin. The highest efficiency of transformation was obtained when selection was conducted at 2 days post-bombardment. For D. phalaenopsis and D. nobile, respectively, about 12% and 2% of the bombarded calli or PLBs produced independent transgenic plants. Integration and expression of the transgenes were confirmed by Southern hybridization and Northern hybridization. No nontransformed plants were regenerated, indicating a tight selection scheme. However, separate incorporation of the gus gene and the hpt gene was observed, and in one transgenic line the gus gene was integrated into the genome of the transgenic plant, but not expressed.





Similar content being viewed by others
Abbreviations
- BA :
-
Benzyladenine
- CaMV :
-
Cauliflower mosaic virus
- GUS :
-
β-Glucuronidase
- HPT :
-
Hygromycin phosphotransferase gene
- PLB :
-
Protocorm-like body
References
Anzai H, Ishii Y, Shichinohe M, Katsumata K, Nojiri C, Morikawa H, Tanaka M (1996) Transformation of Phalaenopsis by particle bombardment. Plant Tissue Cult Lett 13:265–271
Belarmino MM, Mii M (2000) Agrobacterium-mediated genetic transformation of a Phalaenopsis orchid. Plant Cell Rep 19:435–442
Chang C, Chang WC (1998) Plant regeneration from callus culture of Cymbidium ensifolium var. misericors. Plant Cell Rep 17:251–255
Chia TF, Chan YS, Chua NH (1994) The firefly luciferase gene as a non-invasive reporter for Dendrobium transformation. Plant J 6:441–446
Chomezynski P, Sachi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162:156–159
Christo P, Ford TL (1995) The impact of selection parameters on the phenotype and genotype of transgenic rice callus and plants. Transgenic Res 4:44–51
Christo P, Ford TL, Kofron M (1991) Production of transgenic rice (Oryza sativa L.) plants from agronomically important Indica and Japonica varieties via electric discharge particle acceleration of exogenous DNA into immature zygotic embryos. Biotechnology 9:957–962
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Kerbauy GB (1984) Plant regeneration of Oncidium varicosum (Orchidaceae) by means of root tip culture. Plant Cell Rep 3:27–29
Kerbauy GB (1991) In vitro conversion of Cattleya root tip cells into protocorm-like bodies. J Plant Physiol 138:248–251
Knapp JE, Kausch AP, Chandlee JM (2000) Transformation of three genera of orchid using the bar gene as a selectable marker. Plant Cell Rep 19:893–898
Kuehnle AR, Sugii N (1992) Transformation of Dendrobium orchid using particle bombardment of protocorms. Plant Cell Rep 11:484–488
McElroy D, Brattel RS (1994) Foreign gene expression in transgenic cereals. Trends Biotechnol 12:62–68
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Murray MG, Thompson WF (1980) Rapid isolation of high-molecular-weight plant DNA. Nucleic Acids Res 8:4321–4325
Sanford JC, Smith FD, Russell JA (1993) Optimizing the biolistic process for different biological applications. Methods Enzymol 217:483–509
Wan Y, Lemaux PG (1994) Generation of large numbers of independently transformed fertile barley plants. Plant Physiol 104:37–48
Yang J, Lee H, Shin DH, Oh SK, Seon JH, Paek KY, Han K (1999) Genetic transformation of Cymbidium orchid by particle bombardment. Plant Cell Rep 18:978–984
Yu H, Goh CH (2001) Molecular genetics of reproductive biology in orchids. Plant Physiol 127:1390–1393
Yu H, Yang SH, Goh CJ (2001) Agrobacterium-mediated transformation of a Dendrobium orchid with the 1 knox gene DOH1. Plant Cell Rep 20:301–305
Yu L, Li Y, Gu G, Wang Y, Zhou B, An C, Chen ZL (1999) A fast, sensitive and specific method for rice dwarf virus detection by northern blot hybridization. Acta Virol 43:361–365
Yu ZH, Chen MY, Nie L, Lu HF, Ming XT, Zheng HH, Qu LJ, Chen ZL (1999) Recovery of transgenic orchid plants with hygromycin selection by particle bombardment to protocorms. Plant Cell Tissue Organ Cult 58:87–92
Zhao ZY, Cai T, Tagliani L, Miller M, Wang N, Pang H, Rudert M, Schroeder S, Hondred D, Seltzer J, Pierce D (2000) Agrobacterium-mediated sorghum transformation. Plant Mol Biol 44:789–798
Zheng HH, Yu L, Wei C H, Hu DW, Chen ZL, Li Y (2000) Assembly of double-shelled, virus-like particles in transgenic rice plants expressing two major structural proteins of rice dwarf virus. J Virol 74:9808–9810
Acknowledgements
We are grateful to Dr. Richard Jefferson for providing the plasmid pCAMBIA1301 and to Dr. Stephen Chisholm for his helpful comments of the manuscript. This investigation was supported by the Bureau of Science and Technology of Shenzhen and Grant no. 985 from Peking University, Beijing, China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Lörz
Rights and permissions
About this article
Cite this article
Men, S., Ming, X., Wang, Y. et al. Genetic transformation of two species of orchid by biolistic bombardment. Plant Cell Rep 21, 592–598 (2003). https://doi.org/10.1007/s00299-002-0559-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-002-0559-4