Abstract
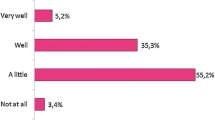
Patient perceptions influence biosimilar uptake in non-mandatory transitions. Companions (support people) are often actively involved in the patient’s medical journey and are likely to have unique perceptions of biosimilars, which may shape patient attitudes. This study explores the congruence between patient and companion perceptions towards biosimilars and their information needs. Patients taking bio-originators for rheumatic diseases (59% for rheumatoid arthritis) and their companions received an explanation about biosimilars. Participants (N = 78) completed questionnaires assessing their familiarity with biosimilars, perceptions, concerns, and benefits of being accompanied. Contingency tables and paired sample t-tests were used to explore differences in familiarity, confidence in knowledge, and perceptions. Intra-class correlation coefficients were calculated to assess the degree of congruence for perceptions towards biosimilars. Companions were significantly less familiar with biosimilars (p = 0.014, Cramer’s V = 0.28) and reported lower confidence in their knowledge (p = 0.006, Cohen’s d = 0.47) than patients. Companions and patients had moderate to good congruency for perceptions toward confidence in biosimilar use and safety, efficacy, and side-effect expectations (intra-class correlation coefficients ranging from 0.75 to 0.81). Companions and patients were most concerned about safety and effectiveness. Companions also reported concerns about cost savings driving the transition, while patients had concerns about uncertainty and testing. Patients reported the ability for discussion, improved understanding, and validation as benefits of being accompanied. Companions and patients have similar levels of perceptions and expectations towards biosimilars but report some unique information needs. Future educational interventions should involve companions and address their concerns to help improve biosimilar acceptance.
Similar content being viewed by others
References
Odinet JS, Day CE, Cruz JL, Heindel GA (2018) The biosimilar nocebo effect? A systematic review of double-blinded versus open-label studies. J Manag Care Spec Pharm 24:952–959. https://doi.org/10.18553/jmcp.2018.24.10.952
Tweehuysen L, van den Bemt BJF, van Ingen IL, de Jong AJL, van der Laan WH, van den Hoogen FHJ, den Broeder AA (2018) Subjective complaints as the main reason for biosimilar discontinuation after open-label transition from reference infliximab to biosimilar infliximab. Arthritis Rheumatol 70:60–68. https://doi.org/10.1002/art.40324
van Overbeeke E, De Beleyr B, de Hoon J, Westhovens R, Huys I (2017) Perception of originator biologics and biosimilars: a survey among Belgian rheumatoid arthritis patients and rheumatologists. BioDrugs 31:447–459. https://doi.org/10.1007/s40259-017-0244-3
Kovitwanichkanont T, Raghunath S, Wang D, Kyi L, Pignataro S, Morton S, Morand E, Leech M (2020) Who is afraid of biosimilars? Openness to biosimilars in an Australian cohort of patients with rheumatoid arthritis. Intern Med J 50:374–377. https://doi.org/10.1111/imj.14753
Jacobs I, Singh E, Sewell KL, Al-Sabbagh A, Shane LG (2016) Patient attitudes and understanding about biosimilars: an international cross-sectional survey. Patient Prefer Adher 10:937–948. https://doi.org/10.2147/PPA.S104891
Oskouei ST, Kusmierczyk AR (2021) Biosimilar uptake: the importance of healthcare provider education. Pharmaceut Med. https://doi.org/10.1007/s40290-021-00396-7
Rezk MF, Pieper B (2017) Treatment outcomes with biosimilars: be aware of the nocebo effect. Rheumatol Ther 4:209–218. https://doi.org/10.1007/s40744-017-0085-z
Peyrin-Biroulet L, Lonnfors S, Roblin X, Danese S, Avedano L (2017) Patient perspectives on biosimilars: a survey by the European Federation of Crohn’s and Ulcerative Colitis Associations. J Crohns Colitis 11:128–133. https://doi.org/10.1093/ecco-jcc/jjw138
Rezk MF, Pieper B (2018) To see or NOsee: the debate on the nocebo effect and optimizing the use of biosimilars. Adv Ther 35:749–753. https://doi.org/10.1007/s12325-018-0719-8
Germain V, Scherlinger M, Barnetche T, Schaeverbeke T (2020) Long-term follow-up after switching from originator infliximab to its biosimilar CT-P13: the weight of nocebo effect. Ann Rheum Dis 79:e11. https://doi.org/10.1136/annrheumdis-2018-214374
Vandenplas Y, Simoens S, Van Wilder P, Vulto AG, Huys I (2021) Informing patients about biosimilar medicines: the role of European patient associations. Pharmaceuticals (Basel). https://doi.org/10.3390/ph14020117
Gasteiger C, Jones ASK, Kleinstäuber M, Lobo M, Horne R, Dalbeth N, Petrie KJ (2020) Effects of message framing on patients’ perceptions and willingness to change to a biosimilar in a hypothetical drug switch. Arthritis Care Res 72:1323–1330. https://doi.org/10.1002/acr.24012
Wolff JL, Roter DL (2011) Family presence in routine medical visits: a meta-analytical review. Soc Sci Med 72:823–831. https://doi.org/10.1016/j.socscimed.2011.01.015
Laidsaar-Powell RC, Butow PN, Bu S, Charles C, Gafni A, Lam WWT, Jansen J, McCaffery KJ, Shepherd HL, Tattersall MHN, Juraskova I (2013) Physician–patient–companion communication and decision-making: a systematic review of triadic medical consultations. Patient Educ Couns 91:3–13. https://doi.org/10.1016/j.pec.2012.11.007
Ho A (2008) Relational autonomy or undue pressure? Family’s role in medical decision-making. Scand J Caring Sci 22:128–135. https://doi.org/10.1111/j.1471-6712.2007.00561.x
Epstein RM, Street RL Jr (2011) The values and value of patient-centered care. Ann Fam Med 9:100–103. https://doi.org/10.1370/afm.1239
Hale JL, Householder BJ, Greene KL (2002) The theory of reasoned action. In: Dillard JP, Pfau M (eds) The persuasion handbook: developments in theory and practice, vol 14. SAGE Publications, California, pp 259–286. https://doi.org/10.4135/9781412976046
Ajzen I (1985) From intentions to actions: A theory of planned behavior. In: Kuhl J, Beckmann J (eds) Action control: from cognition to behavior. Springer, Berlin, pp 11–39
Holt-Lunstad J (2018) Why social relationships are important for physical health: a systems approach to understanding and modifying risk and protection. Annu Rev Psychol 69:437–458. https://doi.org/10.1146/annurev-psych-
Cené CW, Haymore LB, Lin FC, Laux J, Jones CD, Wu JR, DeWalt D, Pignone M, Corbie-Smith G (2015) Family member accompaniment to routine medical visits is associated with better self-care in heart failure patients. Chronic Illn 11:21–32. https://doi.org/10.1177/1742395314532142
Betegnie A, Gauchet A, Lehmann A, Grange L, Roustit M, Baudrant M, Bedouch P, Allenet B (2016) An assessment of patients’ adherence using a self-report questionnaire. J Rheumatol 43:724–730. https://doi.org/10.3899/jrheum.150414
Vriezekolk JE, Peters AJF, van den Ende CHM, Geenen R (2019) Solicitous and invalidating responses are associated with health-care visits in fibromyalgia. Rheumatol Adv Pract 3:rkz008. https://doi.org/10.1093/rap/rkz008
Renton WD, Leveret H, Guly C, Smee H, Leveret J, Ramanan AV (2019) Same but different? A thematic analysis on adalimumab biosimilar switching among patients with juvenile idiopathic arthritis. Pediatr Rheumatol. https://doi.org/10.1186/s12969-019-0366-x
Laidsaar-Powell R, Butow P, Bu S, Fisher A, Juraskova I (2016) Attitudes and experiences of family involvement in cancer consultations: a qualitative exploration of patient and family member perspectives. Support Care Cancer 24:4131–4140. https://doi.org/10.1007/s00520-016-3237-8
Hemmington A, Dalbeth N, Jarrett P, Fraser AG, Broom R, Browett P, Petrie KJ (2017) Medical specialists’ attitudes to prescribing biosimilars. Pharmacoepidemiol Drug Saf 26:570–577. https://doi.org/10.1002/pds.4186
Gasteiger C, Groom KM, Lobo M, Scholz U, Dalbeth N, Petrie KJ (2021) Is three a crowd? The influence of companions on a patient’s decision to transition to a biosimilar. Ann Behav Med. https://doi.org/10.1093/abm/kaab082
Lamore K, Montalescot L, Untas A (2017) Treatment decision-making in chronic diseases: What are the family members’ roles, needs and attitudes? A systematic review. Patient Educ Couns 100:2172–2181. https://doi.org/10.1016/j.pec.2017.08.003
Bracher M, Stewart S, Reidy C, Allen C, Townsend K, Brindle L (2020) Partner involvement in treatment-related decision making in triadic clinical consultations: a systematic review of qualitative and quantitative studies. Patient Educ Couns 103:245–253. https://doi.org/10.1016/j.pec.2019.08.031
Danese S, Fiorino G, Raine T, Ferrante M, Kemp K, Kierkus J, Lakatos PL, Mantzaris G, van der Woude J, Panes J, Peyrin-Biroulet L (2017) ECCO position statement on the use of biosimilars for inflammatory bowel disease-an update. J Crohns Colitis 11:26–34. https://doi.org/10.1093/ecco-jcc/jjw198
Frantzen L, Cohen JD, Trope S, Beck M, Munos A, Sittler MA, Diebolt R, Metzler I, Sordet C (2019) Patients’ information and perspectives on biosimilars in rheumatology: a French nation-wide survey. Jt Bone Spine 86:491–496. https://doi.org/10.1016/j.jbspin.2019.01.001
Wilkins AR, Venkat MV, Brown AS, Dong JP, Ran NA, Hirsch JS, Close KL (2014) Patient perspectives on biosimilar insulin. J Diabetes Sci Technol 8:23–25. https://doi.org/10.1177/1932296813515132
Scherlinger M, Langlois E, Germain V, Schaeverbeke T (2019) Acceptance rate and sociological factors involved in the switch from originator to biosimilar etanercept (SB4). Semin Arthritis Rheum 48:927–932. https://doi.org/10.1016/j.semarthrit.2018.07.005
Petit J, Antignac M, Poilverd RM, Baratto R, Darthout S, Desouches S, Louati K, Deparis N, Berenbaum F, Beauvais C (2021) Multidisciplinary team intervention to reduce the nocebo effect when switching from the originator infliximab to a biosimilar. RMD Open. https://doi.org/10.1136/rmdopen-2020-001396
Gasteiger C, Lobo M, Dalbeth N, Petrie KJ (2021) Patients’ beliefs and behaviours are associated with perceptions of safety and concerns in a hypothetical biosimilar switch. Rheumatol Int 41:163–171. https://doi.org/10.1007/s00296-020-04576-7
Laidsaar-Powell R, Butow P, Charles C, Gafni A, Entwistle V, Epstein R, Juraskova I (2017) The TRIO framework: conceptual insights into family caregiver involvement and influence throughout cancer treatment decision-making. Patient Educ Couns 100:2035–2046. https://doi.org/10.1016/j.pec.2017.05.014
Weir K, Nickel B, Naganathan V, Bonner C, McCaffery K, Carter SM, McLachlan A, Jansen J (2018) Decision-making preferences and deprescribing: perspectives of older adults and companions about their medicines. J Gerontol B Psychol Sci Soc Sci 73:e98–e107. https://doi.org/10.1093/geronb/gbx138
Clayman ML, Roter D, Wissow LS, Bandeen-Roche K (2005) Autonomy-related behaviors of patient companions and their effect on decision-making activity in geriatric primary care visits. Soc Sci Med 60:1583–1591. https://doi.org/10.1016/j.socscimed.2004.08.004
Tweehuysen L, Huiskes VJB, van den Bemt BJF, Vriezekolk JE, Teerenstra S, van den Hoogen FHJ, van den Ende CH, den Broeder AA (2018) Open-label, non-mandatory transitioning from originator etanercept to biosimilar SB4: Six-month results from a controlled cohort study. Arthritis Rheumatol 70:1408–1418. https://doi.org/10.1002/art.40516
Beisecker AE, Brecheisen MA, Ashworth J, Hayes J (1997) Perceptions of the role of cancer patients’ companions during medical appointments. J Psychosoc Oncol 14:29–45. https://doi.org/10.1300/J077v14n04_03
Acknowledgements
The authors would like to thank Associate Professor Katie Groom for her help with the explanation, the participants who generously gave their time to take part in this study, and the clinical and administrative staff at the Auckland and Waitematā District Health Boards Rheumatology and Endocrinology Departments for their help with recruiting and data collection.
Funding
This study was not financially supported by any funding agency or grant.
Author information
Authors and Affiliations
Contributions
CG, KP, US, and ND contributed to the study conception and design, and material preparation. CG and ND contributed to data collection. CG analyzed the data. The first draft of the manuscript was written by CG and ND. All authors commented on previous versions of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Nicola Dalbeth has received speaker fees, consulting fees, or grants from Janssen and Abbvie, AstraZeneca, and Amgen. The other authors declare no conflicts of interest.
Ethical approval
Ethical approval was obtained from the New Zealand Health and Disability Ethics Committee (19/CEN/163) and institutional approval was obtained from Auckland District Health Board (A+8700) and Waitematā District Health Board (RM14629). Research was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gasteiger, C., Scholz, U., Petrie, K.J. et al. A bio-what? Medical companions’ perceptions towards biosimilars and information needs in rheumatology. Rheumatol Int 42, 1993–2002 (2022). https://doi.org/10.1007/s00296-021-05037-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-021-05037-5