Abstract
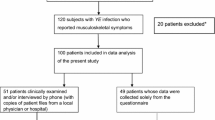
We investigated the association with Yersinia infection in patients with arthropathies in our region. To assess the reactivity to articular antigens, the correlation of anti-Yersinia with anti-type I and type II collagen antibodies was studied. Sera from 124 patients with musculoskeletal symptoms, and 47 synovial fluids (SF) from patients with rheumatoid arthritis (RA), spondyloarthopathies (SpA) or osteoarthritis (OA) were examined. Immunoglobulins against Yersinia enterocolitica, type I and type II collagens were determined by enzyme-linked immunosorbent assay. Immunoglobulin (Ig) A to Yersinia lipopolysaccharide (LPS) was present in 13/124 sera (10%) and 3/47 SF (6%). By Western blot, IgA to Yersinia outer proteins (Yops) was found in 14/124 sera (11%) and 2/47 SF (4%). Yersinia DNA from SF was not amplified by polymerase chain reaction. We found a significant correlation with anti-collagen type I but not type II antibodies. These results suggest different reactivity to articular collagen in patients with Yersinia antibodies.


Similar content being viewed by others
References
Cover TL, Alber RC (1989) Yersinia enterocolitica. N Engl J Med 321:16–24
Ahvonen P, Sievers K, Aho K (1969) Arthritis associated with Yersinia enterocolitica infection. Acta Rheumatol Scand 15:232–253
Tertti R, Granfors K, Lehtonen OP, Mertsola J, Makela AL, Valimaki I, Hanninen P, Toivanen A (1984) An outbreak of Yersinia pseudotuberculosis infection. J Infect Dis 149:245–250
Toivanen A, Lahesmaa-Rantala R, Vuento R, Granfors K (1987) Association of persisting IgA response with Yersinia triggered reactive arthritis: a study on 104 patients. Ann Rheum Dis 46:898–901
Heesemann J, Egger C, Shroeder J (1987) Serological diagnosis of yersiniosis by immunoblot technique using virulence-associated antigen of enteropathogenic Yersiniae. Contrib Microbiol Immunol 9:285–289
Gerard HC, Wang Z, Wang GF, El-Gabalawy H, Goldbach-Mansky R, Li Y, Majeed W, Zhang H, Ngai N, Hudson AP, Schumacher HR (2001) Chromosomal DNA from a variety of bacterial species is present in synovial tissue from patients with various forms of arthritis. Arthritis Rheum 44:1689–1697
Wilkinson NZ, Ward ME, Kingsley GH (1997) Detection of bacteria in rheumatoid and reactive arthritis synovial fluid using a kingdom specific polymerase chain reaction: evidence in support of a role for bacteria in the pathogenesis of inflammatory arthritis. Arthritis Rheum 40(Suppl):270
Sibilia J, Limbach FX (2002) Reactive arthritis or chronic infectious arthritis? Ann Rheum Dis 61:580–587
Di Genaro MS, Muñoz E, Aguilera C, de Guzmán AMS (2000) Yersinia enterocolitica O:8 and O:5 lipopolysaccharide arthritogenicity in hamsters. Rheumatology 39:73–78
Michiels T, Wattiau P, Brasseur R, Ruysschaert JM, Cornelis G (1990) Secretion of Yop proteins by Yersiniae. Infect Immun 58:2840–2849
Schulze-Koops H, Burkhardt H, Heesemann J, von der Mark K, Emmrich F (1995) Characterization of the binding region for the Yersinia enterocolitica adhesin Yad A on types I and II collagen. Arthritis Rheum 38:1283–1289
Viitanen AM, Arstila PT, Lahesmaa R, Granfors S, Skurnik M, Toivanen P (1991) Application of the polymerase chain reaction and immunofluorescence techniques to the detection of bacteria in Yersinia-triggered reactive arthritis. Arthritis Rheum 34:89–96
Pacheco-Tena C, Alvarado De La Barrera C, López-Vidal Y, Velázquez-Mellado J, Richaud-Patin Y, Amieva RI, Llorente L, Martínez A, Zúñiga J, Cifuentes-Alvarado M, Burgos-Vargas R (2001) Bacterial DNA in synovial fluid cells of patients with juvenile onset spondyloarthropathies. Rheumatology 40:920–927
Ririe KM, Rasmussen RP, Wittewer CT (1997) Product differentiation by analysis of DNA melting curves during the polymerase chain reaction. Anal Biochem 245:154–160
Marx A, Sandulache R, Pop A, Cerbu A (1974) Serological cross-reaction between Brucella abortus and Yersinia enterocolitica IX. J Hyg Epidemiol Microbiol Immunol 18:429–431
Arnett F, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, Healey LA, Kaplan SR, Liang MH, Luthra HS (1988) The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31:315–324
Dougados M, van der Linden S, Juhlin R, Huitfeldt B, Amor B, Calin A, Cats A, Dijkmans B, Olivieri I, Pasero G, Veyo E, Zeidler H (1991) The European Spondyloarthropathy Study Group preliminary criteria for the classification of spondyloarthropathy. Arthritis Rheum 34:1218–1227
Balsa A, Carmona L, Gonzalez-Alvaro I, Belmonte MA, Tena X, Sanmarti R, EMECAR Study Group (2004) Value of Disease Activity Score 28 (DAS28) and DAS28–3 compared to American College of Rheumatology-defined remission in rheumatoid arthritis. J Rheumatol 31:40–46
Heesemann J, Gross U, Schmidt N, Laufs R (1986) Immunochemical analysis of plasmid-encoded proteins released by enteropathogenic Yersinis sp grown in calcium deficient media. Infect Immun 54:561–567
Trček J, Wilharm G, Jacovi CA, Heesemann J (2002) Yersinia enterocolitica Yop Q: strain-dependent cytosolic accumulation and post-translational secretion. Microbiology 148:1457–1465
Chatzipanagiotou S, Legakis JN, Boufidou F, Petroyianni V, Nicolaou C (2001) Prevalence of Yersinia plasmid-encoded outer protein (Yop) class-specific antibodies in patients with Hashimoto’s thyroiditis. Clin Microbiol Infect 7:138–143
Kuipers JG, Nietfeld L, Dreses-Werringloer U, Koehler L, Wollenhaupt J, Zeidler H, Hammer M (1999) Optimised sample preparation of synovial fluid for detection of Chlamydia trachomatis DNA by polymerase chain reaction. Ann Rheum Dis 58:103–108
Altschul SF, Madden TL, Schalfer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Bhaduri S, Pickard AR (1995) A method for isolation of chromosomal and plasmid DNA from Yersinia enterocolitica for simultaneous amplification by polymerase chain reaction: a possible model for other bacteria. J Rapid Methods Autom Microbiol 4:107–113
Sen KA (2000) Rapid identification of Yersinia enterocolitica in blood by the 5′ nuclease PCR assay. J Clin Microbiol 38:1953–1958
Wang X, Seed B (2003) A PCR primer bank for quantitative gene expression analysis. Nucleic Acids Res 31:154
Fendler C, Laitko S, Soerensen H, Gripenberg-Lerche C, Groh A, Uksila J, Granfors K, Braun J, Sieper J (2001) Frequency of triggering bacteria in patients with reactive arthritis and undifferentiated oligoarthritis and the relative importance of the tests used for diagnosis. Ann Rheum Dis 60:337–343
Bas S, Grifais R, Kvien TK, Glennas A, Melby K, Vischer TL (1995) Amplification of plasmid and chromosomal Chlamydia DNA synovial fluid of patients with reactive arthritis and undifferentiated seronegative oligoarthritis. Arthritis Rheum 38:1005–1013
Careless DJ, Inman RD (1995) Etiopathogenesis of reactive arthritis and ankylosing spondylitis. Curr Opin Rheum 7:290–298
Aggarwal A, Misra R, Chandrasekhar S, Prasad KN, Dayal R, Ayyagari A (1997) Is undifferentiated seronegative spondyloarthropathy a forme fruste of reactive arthritis? Br J Rheum 36:1001–1004
Granfors K, Toivanen A (1986) IgA anti-Yersinia antibodies in Yersinia triggered reactive arthritis. Ann Rheum Dis 45:561–565
Heesemann J, Keller C, Mosawa R, Schmid N, Siemens H J, Laufs R (1983) Plasmids of human strains of Yersinia enterocolitica molecular relatedness and possible importance for pathogenesis. J Infect Dis 147:107–115
Chatzipanagiotou S, Ladis V, Berdousi H, Palamidou F, Kourti E, Kattamis C (1999) Prevalence of Yersinia plasmid-encoded outer protein (Yop) class-specific antibodies in multitransfused Greek patients with thalassemic syndromes. Clin Microbiol Infect 5:67–72
Hoogkamp-Korstanje JAA, de Koning J, Heesemann J (1988) Persistence of Yersinia enterocolitica in man. Infection 6:81–85
Hoogkamp-Korstaje JAA, de Koning J, Samson JP (1986) Incidence of human infection with Yersinia enterocolitica serotypes O3, O8, O9 and the use of indirect immunofluorescence in diagnosis. J Infect Dis 153:138–141
Kotaniemi A, Risteli J, Aho K, Hakala M (2003) Increased type I collagen degradation correlates with disease activity in reactive arthritis. Clin Exp Rheumatol 21:95–98
Noyori K, Okamoto R, Takagi T, Hyodo A, Suzuki K, Koshino T (1994) Experimental induction of arthritis in rats immunized with Escherichia coli O:14 lipopolysaccharide. J Rheumatol 21:484–488
Terato K, Ye XJ, Miyahara H, Cremer MA, Griffiths MM (1996) Induction of chronic autoimmune arthritis in DBA/I mice by oral administration of type II collagen and Echerichia coli lipopolysaccharide. Br J Rheumatol 35:828–838
Idogawa H, Imamura A, Matsuo K, Yoshitake K, Umemura T, Ohashi M (1998) A monoarthritis model in rabbits induced by repeated intra-articular injections of lipoplysaccharide. Int J Exp Pathol 79:93–104
Otterness IG, Bliven ML, Milici AJ, Poole AR (1994) Comparison of mobility changes with histological and biochemical changes during lipopolysaccharide-induced arthritis in the hamster. Am J Pathol 144:1098–1108
Wuorela M, Jalkanen S, Toivanen P, Granfors K (1993) Yersinia lipopolyssacharide is modified by human monocytes. Infect Immun 61:5261–5270
Singh SK, Girschick HJ (2004) Lyme borreliosis: from infection to autoimmunity. Clin Microbiol Infect 10:598–614
Tiwana H, Natt RS, Benitez-Brito R, Shah S, Wilson C, Bridger S, Harbord M, Sarner M, Ebringer A (2001) Correlation between the immune responses to collagens type I, III, IV and V and Klebsiella pneumoniae in patients with Crohn disease and ankylosing spondylitis. Rheumatology 40:15–23
Rastawicki W, Jagielski M, Giercynski R, Garwolinska H, Kwiatkowska B, Filipowicz-Sosnowska A (2005) The role of Salmonella and Yersinia in the pathogenesis of spondyloarthropathies and rheumatoid arthritis. I Detection of bacteria, bacterial DNA, lipopolysaccharide and ECA antigen in synovial fluid or blood. Med Dosw Mikrobiol 57:131–142
Acknowledgment
This study was supported by the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET, PIP 6224) and Universidad Nacional de San Luis (Proj. 0401 and 8803). Silvia Di Genaro and Silvia G. Correa are member career, and Gabriela Lacoste fellow of CONICET.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lacoste, M.G., Tamashiro, H., Correa, S.G. et al. Correlation between Yersinia enterocolitica and type I collagen reactivity in patients with arthropathies. Rheumatol Int 27, 613–620 (2007). https://doi.org/10.1007/s00296-006-0274-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-006-0274-5