Abstract
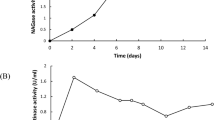
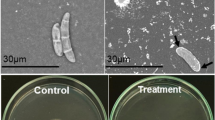
The nag1 gene of the mycoparasitic fungus Trichoderma atroviride encodes a 73-kDa N-acetyl-β-d-glucosaminidase, which is secreted into the medium and partially bound to the cell wall. To elucidate the role of this enzyme in chitinase induction and biocontrol, a nag1-disruption mutant was prepared. It displayed only 4% of the original N-acetyl-β-d-glucosaminidase activity, indicating that the nag1 gene product accounts for the majority of this activity in T. atroviride. The nag1-disruption strain was indistinguishable from the parent strain in growth and morphology, but exhibited delayed autolysis. Northern analysis showed that colloidal chitin disruption does not induce ech42 gene transcription in the nag1-disruption strain. Enzyme activities capable of hydrolysing p-nitrophenyl-N,N′-diacetylchitobioside and p-nitrophenyl-N,N′-diacetylchitotriose were also absent from the nag1-disruption strain under the same conditions. Retransformation of the T. atroviride nag1-disruption strain with the nag1 gene essentially led to the parent-type behaviour in all these experiments. However, addition of N-acetyl-β-d-glucosaminidase to the medium of the nag1-disruption strain did not rescue the mutant phenotype. The disruption-nag1 strain showed 30% reduced ability to protect beans against infection by Rhizoctonia solani and Sclerotinia sclerotiorum. The data indicate that nag1 is essential for triggering chitinase gene expression in T. atroviride and that its functional impairment reduces biocontrol by T. atroviride by a significant extent.


Similar content being viewed by others
References
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (1990) Current protocols in molecular biology. Wiley-Interscience, New York
Baek JM, Howell CR, Kenerley CM (1999) The role of an extracellular chitinase from Trichoderma virens Gv29-8 in the biocontrol of Rhizoctonia solani. Curr Genet 35:41–50
Carsolio C, Gutierrez A, Jiménez B, Van Montagu M, Herrera-Estrella A (1994) Characterization of ech42, a Trichoderma harzianum endochitinase gene expressed during mycoparasitism. Proc Natl Acad Sci USA 91:10903–10907
Carsolio C, Benhamou N, Haran S, Cortes C, Gutierrez A, Chet I, Herrera-Estrella A (1999) Role of the Trichoderma harzianum endochitinase gene, ech42, in mycoparasitism. Appl Environ Microbiol 65:929–935
Chet I, Benhamou N, Haran S (1998) Mycoparasitism and lytic enzymes. In: Harman GE, Kubicek CP (eds) Enzymes, biological control and commercial application. (Trichoderma and Gliocladium, vol 2) Taylor and Francis, London, pp 153–171
Cortes C, Gutierrez A, Olmedo V, Inbar J, Chet I, Herrera-Estrella A (1998) The expression of genes involved in parasitism by Trichoderma harzianum is triggered by a diffusible factor. Mol Gen Genet 260:218–225
Dal Soglio FK, Bertagnolli BL, Sinclair JB, Yu G-Y, Eastburn DM (1998) Production of chitinolytic enzymes and endoglucanase in the soybean rhizosphere in the presence of Trichoderma harzianum and Rhizoctonia solani. Biol Control 12:111–117
Draborg H, Kaupinnen S, Dalboge H, Christgau S (1995) Molecular cloning and expression in Saccharomyces cerevisiae of two exochitinases from Trichoderma harzianum. Biochem Mol Biol Int 36:781–791
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Garcia I, Lora JM, Cruz J de la, Benitez T, Llobell A, Pintor-Toro A (1994) Cloning and characterization of a chitinase (chit42) cDNA from the mycoparasitic fungus Trichoderma harzianum. Curr Genet 27:83–89
Griffiths D (1994) Fungal physiology. Wiley, New York
Haran S, Schickler H, Oppenheim A, Chet I (1995) New components of the chitinolytic system of Trichoderma harzianum. Mycol Res 99:441–446
Harman GE, Björkman T (1998) Potential and existing uses of Trichoderma and Gliocladium for plant disease control and plant growth enhancement. In: Harman GE, Kubicek CP (eds) Enzymes, biological control and commercial application. (Trichoderma and Gliocladium, vol 2) Taylor and Francis, London, pp 229–265
Harman GE, Hayes CK, Lorito M, Broadway RM, Di Pietro A, Tronsmo A (1993) Chitinolytic enzymes of Trichoderma harzianum: purification of chitobiosidase and endochitinase. Phytopathology 83:313–318
Hjeljord L, Tronsmo A (1998) Trichoderma and Gliocladium in biological control: an overview. In: Harman GE, Kubicek CP (eds) Enzymes, biological control and commercial application. (Trichoderma and Gliocladium, vol 2) Taylor and Francis, London, pp 129–151
Inbar J, Chet I (1995) The role of recognition in the induction of specific chitinases during mycoparasitism by Trichoderma harzianum. Microbiology 141:2823–2829
Kelly JM, Hynes MJ (1985) Transformation of Aspergillus niger by the amdS gene of Aspergillus nidulans. EMBO J 4:475–479
Kim DJ, Baek JM, Uribe P, Kenerley CM, Cook DR (2002) Cloning and characterization of multiple glycosyl hydrolase genes from Trichoderma virens. Curr Genet 40:374–384
Koga K, Iwamoto Y, Sakamoto H, Hatano K, Sano M, Kato I (1991) Purification and characterization of β-N-acetylhexosaminidase from Trichoderma harzianum. Agric Biol Chem 55:2817–2833
Kubicek CP, Mach RL, Peterbauer CK, Lorito M (2001) Trichoderma chitinases: from genes to biocontrol. J Plant Pathol 83:11–23
Kullnig CM, Krupica T, Woo SL, Mach RL, Rey M, Benitez T, Lorito M, Kubicek CP (2001) Confusion abounds over identity of Trichoderma biocontrol isolates. Mycol Res 105:769–772
Lorito M (1998) Chitinolytic enzymes ands their genes. In: Harman GE, Kubicek CP (eds) Enzymes, biological control and commercial application. (Trichoderma and Gliocladium, vol 2) Taylor and Francis, London, pp 73–99
Lorito M, Harman GE, Hayes CK, Broadway RM, Tronsmo A, Woo SL, Di Pietro A (1993) Chitinolytic enzymes produced by Trichoderma harzianum: antifungal activity of purified endochitinase and chitobiosidase. Phytopathology 83:302–307
Lorito M, Hayes CK, Di Pietro A, Woo SL, Harman GE (1994) Purification, characterization and synergistic activity of a glucan 1,3-β-glucosidase and N-acetyl-β-glucosaminidase from Trichoderma harzianum. Phytopathology 84:398–405
Lorito M, Woo SL, D′Ambrosio M, Harman GE, Hayes CK, Kubicek CP, Scala F (1996) Synergistic interaction between cell wall degrading enzymes and membrane affecting compounds. Mol Plant Microbe Interact 9:206–213
Mach RL, Peterbauer CK, Payer K, Jaksits S, Woo SL, Zeilinger S, Kullnig CM, Lorito M, Kubicek CP (1999) Expression of two major chitinase genes of Trichoderma atroviride (T. harzianum P1) is triggered by different regulatory signals. Appl Environ Microbiol 65:1858–1863
Margolles-Clark E, Harman GE, Penttilä M (1996) Enhanced expression of endochitinase in Trichoderma harzianum with the cbh1 promoter of Trichoderma reesei. Appl Environ Microbiol 62:2152–2155
Mercedes Dana M de la, Limon CM, Meijas R, Mach RL, Pintor-Toro JA, Kubicek CP (2001) Regulation of chitinase 33 (chit33) gene expression in Trichoderma harzianum. Curr Genet 38:335–342
Peterbauer C, Lorito M, Hayes CK, Harman GE, Kubicek CP (1996) Molecular cloning and expression of nag1 (N-acetyl-β-d-glucosaminidase-encoding) gene from Trichoderma harzianum P1. Curr Genet 29:812–820
Peterbauer CK, Brunner K, Mach RL, Kubicek CP (2002a) Identification of the N-acetyl-d-glucosamine-inducible element in the promoter of the Trichoderma atroviride nag1 gene encoding N-acetyl-glucosaminidase. Mol Genet Genomics 267:162–170
Peterbauer CK, Litscher D, Kubicek CP (2002b) The Trichoderma atroviride seb1-(stress response element binding) gene encodes an AGGGG-binding protein which is involved in osmotic stress response. Mol Genet Genomics 268:223–231
Reyes F, Calatayud J, Vazquez C, Martinez MJ (1989a) Beta-N-acetylglucosaminidase from Aspergillus nidulans which degrades chitin oligomers during autolysis. FEMS Microbiol Lett 53:83–87
Reyes F, Calatayud J, Martinez MJ (1989b) Endochitinases from Aspergillus nidulans Beta-N-acetylglucosaminidase from Aspergillus nidulans implicated in the autolysis of the cell-wall. FEMS Microbiol Lett 51:119–124
Sahai AS, Manocha MS (1993) Chitinases of fungi and plants: their involvement in morphogenesis and host-parasite interaction. FEMS Microbiol Rev 11:317–338
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Plainview, N.Y.
Seiboth B, Hakola S, Mach RL, Suominen PL, Kubicek CP (1997) Role of four major cellulases in triggering of cellulase gene expression by cellulose in Trichoderma reesei. J Bacteriol 179:5318–5320
Suominen PL, Mantylä AL, Karhunen T, Hakola S, Nevalainen H (1993) High frequency one-step gene replacement in Trichoderma reesei. II. Effects of deletions of individual cellulase genes. Mol Gen Genet 241:523–530
Viterbo A, Haran S, Friesem D, Ramot O, Chet I (2001) Antifungal activity of a novel endochitinase gene (chit36) from Trichoderma harzianum Rifai TM. FEMS Microbiol Lett 200:169–174
White S, McIntyre M, Berry DR, McNeil B (2002) The autolysis of industrial filamentous fungi. Crit Rev Biotechnol 22:1–14
Woo SL, Donzelli B, Scala F, Mach RL, Harman GE, Kubicek CP, Del Sorbo G, Lorito M (1998) Disruption of ech42 (endochitinase-encoding) gene affects biocontrol activity in Trichoderma harzianum strain P1. Mol Plant Microbe Interact 12:419–429
Wrathall CR, Tatum EL (1973) The peptides of the hyphal wall of Neurospora crassa. J Gen Microbiol 78:139–153
Yanisch-Perron C, Vieira J, Messing J (1985) Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene 33:103–119
Zeilinger S, Galhaup C, Payer K, Woo SL, Mach RL, Fekete C, Lorito M, Kubicek CP (1999) Chitinase gene expression during mycoparasitic interaction of Trichoderma harzianum with its host. Fungal Genet Biol 26:131–140
Acknowledgements
This work was supported by grants from the Austrian Science Foundation to C.K.P. (P-13170 MOB) and S.Z. (P-15483). M.L. and C.P.K. also recognize the support of the EU-funded TRICHOEST project (QLRT-2001-02032). M.L. recognizes the grants FIRB 2002 [Genomica funzionale dell′interazione trapiante e micro-organismi (patogeni, antagonisti e simbiotici): determinanti coinvolti nella produzione agricola e protezione dell′ambiente] and MIPAF (Risorse genetiche di organismi utili per il miglioramento di specie di interesse agrario e per un′agricoltura sostenibile). K.B.′s stay in Naples was supported by a grant from the ÖAD (Scientific-technological collaboration between Austria and Italy) and by the Ausseninstitut of Technische Universität Wien. S.Z. is the recipient of an APART fellowship (number 10764)
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by U. Kück
K. Brunner and C.K. Peterbauer contributed equally to the present work and are listed in alphabetical order
Rights and permissions
About this article
Cite this article
Brunner, K., Peterbauer, C.K., Mach, R.L. et al. The Nag1 N-acetylglucosaminidase of Trichoderma atroviride is essential for chitinase induction by chitin and of major relevance to biocontrol. Curr Genet 43, 289–295 (2003). https://doi.org/10.1007/s00294-003-0399-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-003-0399-y