Abstract
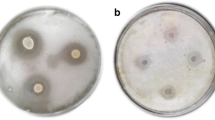
Mycotoxins may be present in nuts, coffee, cereals, and grapes, among other products. Increasing concerns about human health and environmental protection have driven the application of biological control techniques that can inhibit fungal contaminants. In this study, the growth inhibition of the ochratoxigenic fungus Aspergillus carbonarius Ac 162 was evaluated using 5 lactic acid bacteria (LAB). The LAB studied were Lactobacillus plantarum MZ801739 (J), Lactobacillus plantarum MZ809351 (31) and Lactobacillus plantarum MZ809350 (34), isolated in the Ivory Coast, and Lactobacillus plantarum MN982928 (3) and Leuconostoc citreum MZ801735 (23), isolated in Mexico. J, 31, 34, 3 and 23 are the internal strain codes from our laboratory. LAB were cultivated in De Man, Rogosa and Sharpe (MRS) broth, and different polyols (glycerol, mannitol, sorbitol, and xylitol) were added to the culture broth to stimulate the production of antifungal compounds. The fungal inhibition studies were performed using the poisoned food technique. The highest inhibition of A. carbonarius growth was obtained by cultivating L. plantarum MZ809351 in the presence of xylitol and glycerol. Under these conditions, 1 L of the L. plantarum MZ809351 cultures were used to identify antifungal compounds. The compounds were concentrated by solid-phase extraction and then characterized by GC–MS. In addition to 9-octadecenoic acid, 3 diketopiperazines or cyclic dipeptides were identified, including cyclo (Leu-Leu), cyclo (Pro-Gly) and cyclo (Val-Phe), which were compounds related to microbial antifungal activities. Xylitol and glycerol induced the production of these antifungal compounds against A. carbonarius Ac 162. On the other hand, adding xylitol and glycerol to the MRS broth reduced the Ochratoxin A (OTA) content to 56.8 and 54.7%, respectively. This study shows the potential for using L. plantarum MZ809351 as a biocontrol agent to prevent the growth of A. carbonarius and reduce the production of OTA in foods.





Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Leyva Salas M, Mounier J, Valence F, Coton M, Thierry A, Coton E (2017) Antifungal microbial agents for food biopreservation—a review. Microorganisms 5:1–37. https://doi.org/10.3390/microorganisms5030037
Marin S, Ramos AJ, Cano-Sancho G, Sanchis V (2013) Mycotoxins: occurrence, toxicology, and exposure assessment. Food Chem Toxicol 60:218–237. https://doi.org/10.1016/j.fct.2013.07.047
Amézqueta S, Schorr-Galindo S, Murillo-Arbizu M, González-Peñas E, López de Cerain A, Guiraud JP (2012) OTA-producing fungi in foodstuffs: a review. Food Control 6:259–268. https://doi.org/10.1016/j.foodcont.2012.01.042
Ogunade IM, Martinez-Tuppia C, Queiroz OCM, Jiang Y, Drouin P, Wu F, Vyas D, Adesogan AT (2018) Silage review: mycotoxins in silage: occurrence, effects, prevention, and mitigation. J Dairy Sci 101:4034–4059. https://doi.org/10.3168/jds.2017-13788
Djossou O, Perraud-Gaime I, Lakhal Mirleau F, Rodriguez-Serrano G, Karou G, Niamke S, Ouzari M, Boudabous A, Roussos S (2011) Robusta coffee beans post-harvest microflora: Lactobacillus plantarum sp. as potential antagonist of Aspergillus carbonarius. Anaerobe 17:267–272. https://doi.org/10.1016/j.anaerobe.2011.03.006
Lee H, Churey JJ, Worobo RW (2008) Purification and structural characterization of bacillomycin F produced by a bacterial honey isolate active against Byssochlamys fulva H25. J Appl Microbiol 105:663–673. https://doi.org/10.1111/j.1365-2672.2008.03797.x
Wu L, Wu HJ, Qiao J, Gao X, Borriss R (2015) Novel routes for improving biocontrol activity of Bacillus based bioinoculants. Front Microbiol 6:1–13. https://doi.org/10.3389/fmicb.2015.01395
Kumar N, Singh N, Jaryal R, Bhandari C, Singh J, Thakur P et al (2019) Purification, characterization and antibacterial spectrum of a compound produced by Bacillus cereus MTCC 10072. Arch Microbiol 201:1195–1205. https://doi.org/10.1007/s00203-019-01685-1
Gerez CL, Torres MJ, Font de Valdez G, Rollán G (2013) Control of spoilage fungi by lactic acid bacteria. Biol Control 64:231–237. https://doi.org/10.1016/j.biocontrol.2012.10.009
Stoianova LG, Ustiugova EA, Netrusov AI (2012) Antibacterial metabolites of lactic acid bacteria: their diversity and properties. Appl Biochem Microbiol 48:229–243. https://doi.org/10.1134/S0003683812030143
Todorov SD, Dicks LMT (2005) Characterization of bacteriocins produced by lactic acid bacteria isolated from spoiled black olives. J Basic Microbiol 45:312–322. https://doi.org/10.1002/jobm.200410532
Yang EJ, Kim YS, Chang HC (2011) Purification and characterization of antifungal δ-dodecalactone from Lactobacillus plantarum AF1 isolated from kimchi. J Food Prot 74:651–657. https://doi.org/10.4315/0362-028X.JFP-10-512
Kwak MK, Liu R, Kwon JO, Kim MK, Kim AHJ, Kang SO (2013) Cyclic dipeptides from lactic acid bacteria inhibit proliferation of the influenza a virus. J Microbiol 51:836–843. https://doi.org/10.1007/s12275-013-3521-y
Kharazian ZA, Salehi Jouzani G, Aghdasi M, Khorvash M, Zamani M, Mohammadzadeh H (2017) Biocontrol potential of Lactobacillus strains isolated from corn silages against some plant pathogenic fungi. Biol Control 110:33–43. https://doi.org/10.1016/j.biocontrol.2017.04.004
Reis JA, Paula AT, Casarotti SN, Penna ALB (2012) Lactic acid bacteria antimicrobial compounds: characteristics and applications. Food Eng Rev 4:124–140. https://doi.org/10.1007/s12393-012-9051-2
Özogul F, Hamed I (2018) The importance of lactic acid bacteria for the prevention of bacterial growth and their biogenic amines formation: a review. Crit Rev Food Sci Nutr 58:1660–1670. https://doi.org/10.1080/10408398.2016.127797217
Martins MB, Carvalho I (2007) Diketopiperazines: biological activity and synthesis. Tetrahedron 63:9923–9932. https://doi.org/10.1016/j.tet.2007.04.105
Dalié DKD, Deschamps AM, Richard-Forget F (2010) Lactic acid bacteria—potential for control of mould growth and mycotoxins: a review. Food Control 21:370–380. https://doi.org/10.1016/j.foodcont.2009.07.011
Toplaghaltsyan A, Bazukyan I, Trchounian A (2016) The effects of different carbon sources on the antifungal activity by lactic acid bacteria. Curr Microbiol 74:168–174. https://doi.org/10.1007/s00284-016-1168-8
Lipińska L, Klewicki R, Klewicka E, Kołodziejczyk K, Sójka M, Nowak A (2016) Antifungal activity of Lactobacillus sp. Bacteria in the presence of xylitol and galactosyl-xylitol. Biomed Res Int. https://doi.org/10.1155/2016/5897486
Su YK, Willis LB, Jeffries TW (2015) Effects of aeration on growth, ethanol and polyol accumulation by Spathaspora passalidarum NRRL Y-27907 and Scheffersomyces stipitis NRRL Y-7124. Biotechnol Bioeng 112:457–469. https://doi.org/10.1002/bit.25445
Balouiri M, Sadiki M, Ibnsouda SK (2016) Methods for in vitro evaluating antimicrobial activity: a review. J Pharm Anal 6:71–79. https://doi.org/10.1016/j.jpha.2015.11.005
Durand N (2012) Dynamique des populations microbiennes au cours du traitement post récolte du café et relations interspécifiques entre souches ochratoxinogènes. Dissertation, Université Montpellier II. Dynamics of microbial populations during post-harvest coffee processing and interspecific relationships between ochratoxinogenic strains. Dissertation, University of Montpellier II. Retrieved from http://www.theses.fr/2012MON20232. Accessed 10 Feb 2022
Stamatelopoulou E, Agriopoulou S, Dourtoglou E, Chatzilazarou A, Drosou F, Marinea M et al (2018) Diketopiperazines in wines. J Wine Res 29(1):37–48. https://doi.org/10.1080/09571264.2018.1433137
Lipińska L, Klewicki R, Sójka M, Bonikowski R, Żyżelewicz D, Kołodziejczyk K, Klewicka E (2018) Antifungal activity of Lactobacillus pentosus ŁOCK 0979 in the presence of polyols and galactosyl-polyols. Probiotics Antimicrob Proteins 10:186–200. https://doi.org/10.1007/s12602-017-9344-0
Lind H, Sjögren J, Gohil S, Kenne L, Schnürer J, Broberg A (2007) Antifungal compounds from cultures of dairy propionibacteria type strains. FEMS Microbiol Lett 271:310–315. https://doi.org/10.1111/j.1574-6968.2007.00730.x
Díaz-Cárdenas C, Cantillo A, Rojas LY, Sandoval T, Fiorentino S, Robles J, Ramos FA, Zambrano MM, Baena S (2017) Microbial diversity of saline environments: searching for cytotoxic activities. AMB Express 7:1–16. https://doi.org/10.1186/s13568-017-0527-6
Kumar SN, Sreekala SR, Chandrasekaran D, Nambisan B, Anto RJ (2014) Biocontrol of Aspergillus species on peanut kernels by antifungal diketopiperazine producing Bacillus cereus associated with entomopathogenic nematode. PLoS ONE 9:1–14. https://doi.org/10.1371/journal.pone.0106041
Dong QF, Wang JL, Zhang SF, Wang Z, Zhang CX, Gao H et al (2008) Antifungal activity of crude extracts and fat-soluble constituents of Holotrichia diomphalia larvae. Bioresour Technol 99:8521–8523. https://doi.org/10.1016/j.biortech.2008.03.010
Mishra AK, Choi J, Choi SJ, Baek KH (2017) Cyclodipeptides: an overview of their biosynthesis and biological activity. Molecules 22:1–13. https://doi.org/10.3390/molecules22101796
Nissen L, Pérez-Martínez G, Yebra MJ (2005) Sorbitol synthesis by an engineered Lactobacillus casei strain expressing a sorbitol-6-phosphate dehydrogenase gene within the lactose operon. FEMS Microbiol Lett 249:177–183. https://doi.org/10.1016/j.femsle.2005.06.010
Wisselink HW, Weusthuis RA, Eggink G, Hugenholtz J, Grobben GJ (2002) Mannitol production by lactic acid bacteria: a review. Int Dairy J 12:151–161. https://doi.org/10.1016/S0958-6946(01)00153-4
Adeyeye SAO (2016) Fungal mycotoxins in foods: a review. Cogent Food Agric 2:1–11. https://doi.org/10.1080/23311932.2016.1213127
Bhatt SM, Mohan A, Srivastava SK (2013) Challenges in enzymatic route of mannitol production. ISRN Biotechnol 2013:1–13. https://doi.org/10.5402/2013/914187
Li H, Liu L, Zhang S, Cui W, Lv J (2012) Identification of antifungal compounds produced by Lactobacillus casei AST18. Curr Microbiol 65:156–161. https://doi.org/10.1007/s00284-012-0135-2
Acknowledgements
RES acknowledges a PhD fellowship from the Consejo Nacional de Ciencia y Tecnología, CONACYT (National Council for Science and Technology), Mexico 613157. The authors acknowledge project support (ANGELICA 273656) from CONACYT Mexico and Agence Nationale de la Recherche ANR (National Agence for Research), France. This work is dedicated to the memory of Prof. Rosa O. González-Robles.
Funding
This study was supported by a scientific collaboration between Mexico and France. The details are provided in the acknowledgements.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conceptualisation and experimental design of the study. The materials preparation, data collection and analysis of data were performed by RE-S, VT-G, IP-G, GMR-S, ROG-R, EIC-M, and GS-C. The first draft of the manuscript was written by RE-S, and all the authors revised it critically. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical Approval
Not applicable.
Consent to Participate
All the authors gave their consent to participate in this manuscript.
Consent for Publication
All the authors gave their consent to the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
284_2022_2761_MOESM1_ESM.tif
Supplementary file1 Consumption of polyols (%) in Man, Rogosa and Sharpe (MRS) broths by five lactic acid bacteria. Lactobacillus plantarum MZ801739 (J), Lactobacillus plantarum MZ809351 (31) and Lactobacillus plantarum MZ809350 (34) were isolated in the Ivory Coast and Lactobacillus plantarum MN982928 (3) and Leuconostoc citreum MZ801735 (23) were isolated in Mexico. J, 31, 34, 3 and 23 are the internal strain codes from our laboratory (TIF 3217 kb)
284_2022_2761_MOESM2_ESM.tif
Supplementary file2 Growth inhibition of A. carbonarius using the poisoned food technique of eluate 1 (negative control) of treatments with Lactobacillus plantarum MZ809351 (31) cultured in the presence of xylitol, X; and glycerol, G (TIF 8084 kb)
Rights and permissions
About this article
Cite this article
Espinosa-Salgado, R., Tamayo-Galván, V., Perraud-Gaime, I. et al. Polyols Induce the Production of Antifungal Compounds by Lactobacillus plantarum. Curr Microbiol 79, 99 (2022). https://doi.org/10.1007/s00284-022-02761-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-022-02761-4