Abstract
Aim
Cetuximab is an essential drug for the treatment of wild-type K-RAS colorectal cancer (CRC). It improves the overall survival of patients. However, acquired resistance prevents its clinical efficacy. Tumor heterogeneity may be a nonnegligible reason for cetuximab resistance. We attempted to explore the corresponding molecular mechanism.
Methods
Cetuximab-resistant CRC cell RKO and cetuximab-sensitive CRC cell Caco-2 were applied in this study. Cells were centrifuged to determine the concentration in the culture supernatant (CS). MTT, EdU, and colony formation assays were utilized to evaluate cell survival and proliferation. Chromatin immunoprecipitation (ChIP) and promoter-luciferase reporter assays were employed to confirm the direct binding of transcription factors. Western blot and reverse transcription-polymerase chain reaction (RT-PCR) assays were used to detect the expression of molecular markers in the pathway.
Results
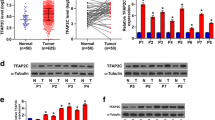
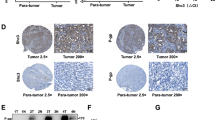
Hepatocyte growth factor (HGF) was up-regulated in RKO cell culture supernatant and induced cetuximab resistance in Caco-2 cells. SRY-Box Transcription Factor 8 (SOX8) bound to the promoter sequence of HGF. HGF activated the HGF/MET bypass pathway and induced cetuximab resistance in Caco-2 cells.
Conclusion
The SOX8/HGF/MET axis played a crucial role in the communication between cetuximab-resistant cells and cetuximab-sensitive cells, inducing treatment resistance.





Similar content being viewed by others
Availability of data and material
The data used to support the findings of this study are available from the corresponding author upon request.
References
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics 2020. CA Cancer J Clin 70(1):7–30. https://doi.org/10.3322/caac.21590
Cohen R, Pudlarz T, Delattre JF, Colle R, André T (2020) Molecular targets for the treatment of metastatic colorectal cancer. Cancers. https://doi.org/10.3390/cancers12092350
Mendelsohn J (2002) Targeting the epidermal growth factor receptor for cancer therapy. J Clin Oncol Off J Am Soc Clin Oncol 20(18 Suppl):1s–13s
Price TJ, Tang M, Gibbs P, Haller DG, Peeters M, Arnold D, Segelov E, Roy A, Tebbutt N, Pavlakis N, Karapetis C, Burge M, Shapiro J (2018) Targeted therapy for metastatic colorectal cancer. Expert Rev Anticancer Ther 18(10):991–1006. https://doi.org/10.1080/14737140.2018.1502664
Berger MD, Ning Y, Stintzing S, Heinemann V, Cao S, Zhang W, Yang D, Miyamoto Y, Suenaga M, Schirripa M, Hanna DL, Soni S, Puccini A, Tokunaga R, Naseem M, Battaglin F, Cremolini C, Falcone A, Loupakis F, Lenz HJ (2020) A polymorphism within the R-spondin 2 gene predicts outcome in metastatic colorectal cancer patients treated with FOLFIRI/bevacizumab: data from FIRE-3 and TRIBE trials. Eur J Cancer 131:89–97. https://doi.org/10.1016/j.ejca.2020.02.048
Yaeger R, Kotani D, Mondaca S, Parikh AR, Bando H, Van Seventer EE, Taniguchi H, Zhao H, Thant CN, de Stanchina E, Rosen N, Corcoran RB, Yoshino T, Yao Z, Ebi H (2019) Response to anti-EGFR Therapy in patients with BRAF non-V600-mutant metastatic colorectal cancer. Clin Cancer Res Off J Am Assoc Cancer Res 25(23):7089–7097. https://doi.org/10.1158/1078-0432.Ccr-19-2004
Gharib E, Salmanipour R, Nazemalhosseini Mojarad E, Yaghoob Taleghani M, Sarlak S, Malekzade-Moghani M, Nasrabadi PN, Meiary MA, Asadzadeh Aghdaei H, Zali MR (2019) HER2(+) mCRC patients with exon 20 R784G substitution mutation do not respond to the cetuximab therapy. J Cell Physiol 234(8):13137–13144. https://doi.org/10.1002/jcp.27984
Weickhardt AJ, Tebbutt NC, Mariadason JM (2010) Strategies for overcoming inherent and acquired resistance to EGFR inhibitors by targeting downstream effectors in the RAS/PI3K pathway. Curr Cancer Drug Targets 10(8):824–833. https://doi.org/10.2174/156800910793357961
Qu L, Ding J, Chen C, Wu ZJ, Liu B, Gao Y, Chen W, Liu F, Sun W, Li XF, Wang X, Wang Y, Xu ZY, Gao L, Yang Q, Xu B, Li YM, Fang ZY, Xu ZP, Bao Y, Wu DS, Miao X, Sun HY, Sun YH, Wang HY, Wang LH (2016) Exosome-transmitted lncARSR promotes sunitinib resistance in renal cancer by acting as a competing endogenous RNA. Cancer Cell 29(5):653–668. https://doi.org/10.1016/j.ccell.2016.03.004
Vitale I, Shema E, Loi S, Galluzzi L (2021) Intratumoral heterogeneity in cancer progression and response to immunotherapy. Nat Med 27(2):212–224. https://doi.org/10.1038/s41591-021-01233-9
Piao HY, Guo S, Wang Y, Zhang J (2020) Exosome-transmitted lncRNA PCGEM1 promotes invasive and metastasis in gastric cancer by maintaining the stability of SNAI1. Clin trans oncol off publ Fed Span Oncol Soc Natl Cancer Inst Mexico. https://doi.org/10.1007/s12094-020-02412-9
Guan X (2015) Cancer metastases: challenges and opportunities. Acta Pharm Sin B 5(5):402–418. https://doi.org/10.1016/j.apsb.2015.07.005
Czarnecka AM, Bartnik E, Fiedorowicz M, Rutkowski P (2020) Targeted therapy in melanoma and mechanisms of resistance. Int J Mol Sci. https://doi.org/10.3390/ijms21134576
Piao H-y, Guo S, Jin H, Wang Y, Zhang J (2021) LINC00184 involved in the regulatory network of ANGPT2 via ceRNA mediated miR-145 inhibition in gastric cancer. J Cancer 12(8):2336–2350
Lu H, Zhang H, Weng ML, Zhang J, Jiang N, Cata JP, Ma D, Chen WK, Miao CH (2020) Morphine promotes tumorigenesis and cetuximab resistance via EGFR signaling activation in human colorectal cancer. J Cell Physiol. https://doi.org/10.1002/jcp.30161
Wu T, Dai Y (2017) Tumor microenvironment and therapeutic response. Cancer Lett 387:61–68. https://doi.org/10.1016/j.canlet.2016.01.043
Shi Y, Riese DJ 2nd, Shen J (2020) The role of the CXCL12/CXCR4/CXCR7 chemokine axis in cancer. Front Pharmacol 11:574667. https://doi.org/10.3389/fphar.2020.574667
Mhaidly R, Mechta-Grigoriou F (2020) Fibroblast heterogeneity in tumor micro-environment: role in immunosuppression and new therapies. Semin Immunol 48:101417. https://doi.org/10.1016/j.smim.2020.101417
Comoglio PM, Trusolino L, Boccaccio C (2018) Known and novel roles of the MET oncogene in cancer: a coherent approach to targeted therapy. Nat Rev Cancer 18(6):341–358. https://doi.org/10.1038/s41568-018-0002-y
Qin H, Yang Y, Jiang B, Pan C, Chen W, Diao W, Ding M, Cao W, Zhang Z, Chen M, Gao J, Zhao X, Qiu X, Guo H (2021) SOX9 in prostate cancer is upregulated by cancer-associated fibroblasts to promote tumor progression through HGF/c-Met-FRA1 signaling. Febs J. https://doi.org/10.1111/febs.15816
Xing F, Zhao D, Wu SY, Tyagi A, Wu K, Sharma S, Liu Y, Deshpande R, Wang Y, Cleary J, Miller LD, Chittiboyina AG, Yalamanchili C, Mo YY, Watabe K (2021) Epigenetic and post-transcriptional modulation of SOS1 can promote breast cancer metastasis through obesity-activated c-Met signaling in African American women. Cancer Res. https://doi.org/10.1158/0008-5472.Can-19-4031
Luo H, Hong H, Slater MR, Graves SA, Shi S, Yang Y, Nickles RJ, Fan F, Cai W (2015) PET of c-Met in cancer with 64Cu-labeled hepatocyte growth factor. J Nucl Med 56(5):758–763. https://doi.org/10.2967/jnumed.115.154690
Miura D, Miura Y, Yagasaki K (2004) Resveratrol inhibits hepatoma cell invasion by suppressing gene expression of hepatocyte growth factor via its reactive oxygen species-scavenging property. Clin Exp Metastasis 21(5):445–451. https://doi.org/10.1007/s10585-004-2698-1
Miyata Y, Asai A, Mitsunari K, Matsuo T, Ohba K, Mochizuki Y, Sakai H (2014) Met Urol Cancers Cancers 6(4):2387–2403. https://doi.org/10.3390/cancers6042387
Song S, Bi M (2014) Research progress of HGF/MET signaling pathway in EGFR-TKI resistance in non-small cell lung cancer. Zhongguo Fei Ai Za Zhi 17(10):755–759. https://doi.org/10.3779/j.issn.1009-3419.2014.10.08
Pfeifer D, Poulat F, Holinski-Feder E, Kooy F, Scherer G (2000) The SOX8 gene is located within 700 kb of the tip of chromosome 16p and is deleted in a patient with ATR-16 syndrome. Genomics 63(1):108–116. https://doi.org/10.1006/geno.1999.6060
Wang Y, Yang W, Liu T, Bai G, Liu M, Wang W (2019) Over-expression of SOX8 predicts poor prognosis in colorectal cancer: a retrospective study. Medicine (Baltimore) 98(27):e16237. https://doi.org/10.1097/md.0000000000016237
Dong L, Zhou D, Xin C, Liu B, Sun P (2020) MicroRNA-139 suppresses the tumorigenicity of triple negative breast cancer cells by targeting SOX8. Cancer Manag Res 12:9417–9428. https://doi.org/10.2147/cmar.S268378
Tang H, Chen B, Liu P, Xie X, He R, Zhang L, Huang X, Xiao X, Xie X (2019) SOX8 acts as a prognostic factor and mediator to regulate the progression of triple-negative breast cancer. Carcinogenesis 40(10):1278–1287. https://doi.org/10.1093/carcin/bgz034
Sun H, Wang H, Wang X, Aoki Y, Wang X, Yang Y, Cheng X, Wang Z, Wang X (2020) Aurora-A/SOX8/FOXK1 signaling axis promotes chemoresistance via suppression of cell senescence and induction of glucose metabolism in ovarian cancer organoids and cells. Theranostics 10(15):6928–6945. https://doi.org/10.7150/thno.43811
Liska D, Chen CT, Bachleitner-Hofmann T, Christensen JG, Weiser MR (2011) HGF rescues colorectal cancer cells from EGFR inhibition via MET activation. Clin Cancer Res Off J Am Assoc Cancer Res 17(3):472–482. https://doi.org/10.1158/1078-0432.Ccr-10-0568
Peng S, Wang R, Zhang X, Ma Y, Zhong L, Li K, Nishiyama A, Arai S, Yano S, Wang W (2019) EGFR-TKI resistance promotes immune escape in lung cancer via increased PD-L1 expression. Mol Cancer 18(1):165. https://doi.org/10.1186/s12943-019-1073-4
Acknowledgements
Liaoning S&T Project (2020-ZLLH-46, 2021-MS-068).
Author information
Authors and Affiliations
Contributions
PHY performed the majority of experiments and analyzed the data, and drafted the manuscript; LYP designed the research; QJL collected and analyzed the data.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethics approval and consent to participate
The study was reviewed and approved by the Faculty of Science Ethics Committee at Liaoning Cancer Hospital & Institute (Cancer Hospital of China Medical University, 20201144K).
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Piao, Hy., Qu, JL. & Liu, YP. SOX8 promotes cetuximab resistance via HGF/MET bypass pathway activation in colorectal cancer. Cancer Chemother Pharmacol 89, 441–449 (2022). https://doi.org/10.1007/s00280-021-04378-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-021-04378-z