Abstract
Purpose
To investigate the association between UDP-glucuronosyltransferase (UGT)1A polymorphisms and irinotecan-treatment efficacy in a Chinese population with metastatic colorectal cancer (mCRC).
Methods
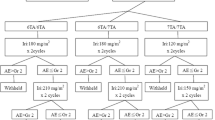
The present study was based on a prospective multicenter trial of Chinese mCRC patients treated with irinotecan-based chemotherapy (NCT01282658, registered at http://www.clinicaltrials.gov). Fifteen single-nucleotide polymorphisms (SNPs) in four UGT1A genes were selected for genotyping in 164 patients. Kaplan–Meier and Cox regression analyses were used to assess the association between potential signatures and survival outcome.
Results
We found that UGT1A1*28 variant genotype was significantly associated with decreased progression-free survival (PFS) [adjusted hazard ratio (HR), 1.803; 95% confidence interval (CI), 1.217–2.671] and overall survival (OS) (adjusted HR 1.979; 95% CI 1.267–3.091) compared with wild-type genotype. Patients carrying (TA)7 allele showed a median PFS of 7.5 (95% CI 5.5–9.6) months compared with 9.8 (95% CI 8.6–10.9) months for patients with wild-type genotype. Median OSs were 13.3 (95% CI 10.3–16.2), and 20.8 (95% CI 18.7–23.0) months for (TA)6/7 or (TA)7/7, and (TA)6/6 patients, respectively. Similarly but more significantly, the copy number of haplotype III (composed by rs3755321-T, rs3821242-C, rs4124874-G and rs3755319-C) constructed among the selected SNPs also correlated with survival outcome.
Conclusions
UGT1A polymorphisms are predictive of survival outcome of irinotecan-treated Chinese mCRC patients. After validation, UGT1A polymorphisms might be helpful in facilitating stratification of mCRC patients for individualized treatment options.



Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65(2):87–108. https://doi.org/10.3322/caac.21262
McPhail S, Johnson S, Greenberg D, Peake M, Rous B (2015) Stage at diagnosis and early mortality from cancer in England. Br J Cancer 112 Suppl 1:S108–S115. https://doi.org/10.1038/bjc.2015.49
Yoo PS, Lopez-Soler RI, Longo WE, Cha CH (2006) Liver resection for metastatic colorectal cancer in the age of neoadjuvant chemotherapy and bevacizumab. Clin Colorectal Cancer 6(3):202–207. https://doi.org/10.3816/CCC.2006.n.036
Liu X, Cheng D, Kuang Q, Liu G, Xu W (2014) Association of UGT1A1*28 polymorphisms with irinotecan-induced toxicities in colorectal cancer: a meta-analysis in Caucasians. Pharmacogenomics J 14(2):120–129. https://doi.org/10.1038/tpj.2013.10
Cai X, Cao W, Ding H, Liu T, Zhou X, Wang M, Zhong M, Zhao Z, Xu Q, Wang L (2013) Analysis of UGT1A1*28 genotype and SN-38 pharmacokinetics for irinotecan-based chemotherapy in patients with advanced colorectal cancer: results from a multicenter, retrospective study in Shanghai. J Cancer Res Clin Oncol 139(9):1579–1589. https://doi.org/10.1007/s00432-013-1480-7
Iyer L, King CD, Whitington PF, Green MD, Roy SK, Tephly TR, Coffman BL, Ratain MJ (1998) Genetic predisposition to the metabolism of irinotecan (CPT-11). Role of uridine diphosphate glucuronosyltransferase isoform 1A1 in the glucuronidation of its active metabolite (SN-38) in human liver microsomes. J Clin Investig 101(4):847–854. https://doi.org/10.1172/JCI915
Hanioka N, Ozawa S, Jinno H, Ando M, Saito Y, Sawada J (2001) Human liver UDP-glucuronosyltransferase isoforms involved in the glucuronidation of 7-ethyl-10-hydroxycamptothecin. Xenobiotica 31(10):687–699. https://doi.org/10.1080/00498250110057341
Gagne JF, Montminy V, Belanger P, Journault K, Gaucher G, Guillemette C (2002) Common human UGT1A polymorphisms and the altered metabolism of irinotecan active metabolite 7-ethyl-10-hydroxycamptothecin (SN-38). Mol Pharmacol 62(3):608–617
Kadakol A, Ghosh SS, Sappal BS, Sharma G, Chowdhury JR, Chowdhury NR (2000) Genetic lesions of bilirubin uridine-diphosphoglucuronate glucuronosyltransferase (UGT1A1) causing Crigler–Najjar and Gilbert syndromes: correlation of genotype to phenotype. Hum Mutation 16 (4):297–306. https://doi.org/10.1002/1098-1004(200010)16:4<297::AID-HUMU2>3.0.CO;2-Z
Gupta E, Lestingi TM, Mick R, Ramirez J, Vokes EE, Ratain MJ (1994) Metabolic fate of irinotecan in humans: correlation of glucuronidation with diarrhea. Cancer Res 54(14):3723–3725
Chen X, Liu L, Guo Z, Liang W, He J, Huang L, Deng Q, Tang H, Pan H, Guo M, Liu Y, He Q, He J (2017) UGT1A1 polymorphisms with irinotecan-induced toxicities and treatment outcome in Asians with lung cancer: a meta-analysis. Cancer Chemother Pharmacol 79(6):1109–1117. https://doi.org/10.1007/s00280-017-3306-9
Zhang X, Yin J-F, Zhang J, Kong S-J, Zhang H-Y, Chen X-M (2017) UGT1A1*6 polymorphisms are correlated with irinotecan-induced neutropenia: a systematic review and meta-analysis. Cancer Chemother Pharmacol 80(1):135–149. https://doi.org/10.1007/s00280-017-3344-3
Toffoli G, Cecchin E, Corona G, Russo A, Buonadonna A, D’Andrea M, Pasetto LM, Pessa S, Errante D, De Pangher V, Giusto M, Medici M, Gaion F, Sandri P, Galligioni E, Bonura S, Boccalon M, Biason P, Frustaci S (2006) The role of UGT1A1*28 polymorphism in the pharmacodynamics and pharmacokinetics of irinotecan in patients with metastatic colorectal cancer. J Clin Oncol 24(19):3061–3068. https://doi.org/10.1200/JCO.2005.05.5400
Shulman K, Cohen I, Barnett-Griness O, Kuten A, Gruber SB, Lejbkowicz F, Rennert G (2011) Clinical implications of UGT1A1*28 genotype testing in colorectal cancer patients. Cancer 117(14):3156–3162. https://doi.org/10.1002/cncr.25735
Lara PN Jr, Natale R, Crowley J, Lenz HJ, Redman MW, Carleton JE, Jett J, Langer CJ, Kuebler JP, Dakhil SR, Chansky K, Gandara DR (2009) Phase III trial of irinotecan/cisplatin compared with etoposide/cisplatin in extensive-stage small-cell lung cancer: clinical and pharmacogenomic results from SWOG S0124. J Clin Oncol 27(15):2530–2535. https://doi.org/10.1200/JCO.2008.20.1061
Xu C, Tang X, Qu Y, Keyoumu S, Zhou N, Tang Y (2016) UGT1A1 gene polymorphism is associated with toxicity and clinical efficacy of irinotecan-based chemotherapy in patients with advanced colorectal cancer. Cancer Chemother Pharmacol 78(1):119–130. https://doi.org/10.1007/s00280-016-3057-z
Dias MM, Pignon JP, Karapetis CS, Boige V, Glimelius B, Kweekel DM, Lara PN, Laurent-Puig P, Martinez-Balibrea E, Paez D, Punt CJ, Redman MW, Toffoli G, Wadelius M, McKinnon RA, Sorich MJ (2014) The effect of the UGT1A1*28 allele on survival after irinotecan-based chemotherapy: a collaborative meta-analysis. Pharmacogenomics J 14(5):424–431. https://doi.org/10.1038/tpj.2014.16
Liu X, Cheng D, Kuang Q, Liu G, Xu W (2013) Association between UGT1A1*28 polymorphisms and clinical outcomes of irinotecan-based chemotherapies in colorectal cancer: a meta-analysis in Caucasians. PloS One 8(3):e58489. https://doi.org/10.1371/journal.pone.0058489
Liu XH, Lu J, Duan W, Dai ZM, Wang M, Lin S, Yang PT, Tian T, Liu K, Zhu YY, Zheng Y, Sheng QW, Dai ZJ (2017) Predictive value of UGT1A1*28 polymorphism in irinotecan-based chemotherapy. J Cancer 8(4):691–703. https://doi.org/10.7150/jca.17210
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45(2):228–247. https://doi.org/10.1016/j.ejca.2008.10.026
Tournigand C, Andre T, Achille E, Lledo G, Flesh M, Mery-Mignard D, Quinaux E, Couteau C, Buyse M, Ganem G, Landi B, Colin P, Louvet C, de Gramont A (2004) FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol 22(2):229–237. https://doi.org/10.1200/JCO.2004.05.113
Meropol NJ, Gold PJ, Diasio RB, Andria M, Dhami M, Godfrey T, Kovatich AJ, Lund KA, Mitchell E, Schwarting R (2006) Thymidine phosphorylase expression is associated with response to capecitabine plus irinotecan in patients with metastatic colorectal cancer. J Clin Oncol 24(25):4069–4077. https://doi.org/10.1200/JCO.2005.05.2084
Fuchs CS, Moore MR, Harker G, Villa L, Rinaldi D, Hecht JR (2003) Phase III comparison of two irinotecan dosing regimens in second-line therapy of metastatic colorectal cancer. J Clin Oncol 21(5):807–814
National Cancer Institute (U.S.) (2009) Common terminology criteria for adverse events (CTCAE). NIH publication, vol no 10-5410, Rev. edn. U.S. Dept. of Health and Human Services. National Institutes of Health, National Cancer Institute, Bethesda
Yu QQ, Qiu H, Zhang MS, Hu GY, Liu B, Huang L, Liao X, Li QX, Li ZH, Yuan XL (2016) Predictive effects of bilirubin on response of colorectal cancer to irinotecan-based chemotherapy. World J Gastroenterol WJG 22(16):4250–4258. https://doi.org/10.3748/wjg.v22.i16.4250
Jurinke C, Oeth P, van den Boom D (2004) MALDI-TOF mass spectrometry: a versatile tool for high-performance DNA analysis. Mol Biotechnol 26(2):147–164. https://doi.org/10.1385/MB:26:2:147
Loupakis F, Yang D, Yau L, Feng S, Cremolini C, Zhang W, Maus MK, Antoniotti C, Langer C, Scherer SJ, Muller T, Hurwitz HI, Saltz L, Falcone A, Lenz HJ (2015) Primary tumor location as a prognostic factor in metastatic colorectal cancer. J Natl Cancer Inst 107(3):dju427. https://doi.org/10.1093/jnci/dju427
Hsieh TY, Shiu TY, Huang SM, Lin HH, Lee TC, Chen PJ, Chu HC, Chang WK, Jeng KS, Lai MM, Chao YC (2007) Molecular pathogenesis of Gilbert’s syndrome: decreased TATA-binding protein binding affinity of UGT1A1 gene promoter. Pharmacogenet Genom 17(4):229–236. https://doi.org/10.1097/FPC.0b013e328012d0da
Stewart CF, Panetta JC, O’Shaughnessy MA, Throm SL, Fraga CH, Owens T, Liu T, Billups C, Rodriguez-Galindo C, Gajjar A, Furman WL, McGregor LM (2007) UGT1A1 promoter genotype correlates with SN-38 pharmacokinetics, but not severe toxicity in patients receiving low-dose irinotecan. J Clin Oncol 25(18):2594–2600. https://doi.org/10.1200/JCO.2006.10.2301
Strassburg CP (2010) Gilbert–Meulengracht’s syndrome and pharmacogenetics: is jaundice just the tip of the iceberg? Drug Metab Rev 42(1):168–181. https://doi.org/10.3109/03602530903209429
Okuyama Y, Hazama S, Nozawa H, Kobayashi M, Takahashi K, Fujikawa K, Kato T, Nagata N, Kimura H, Oba K, Sakamoto J, Mishima H (2011) Prospective phase II study of FOLFIRI for mCRC in Japan, including the analysis of UGT1A1 28/6 polymorphisms. Jpn J Clin Oncol 41(4):477–482. https://doi.org/10.1093/jjco/hyr001
Sunakawa Y, Ichikawa W, Fujita K, Nagashima F, Ishida H, Yamashita K, Mizuno K, Miwa K, Kawara K, Akiyama Y, Araki K, Yamamoto W, Miya T, Narabayashi M, Ando Y, Hirose T, Saji S, Sasaki Y (2011) UGT1A1*1/*28 and *1/*6 genotypes have no effects on the efficacy and toxicity of FOLFIRI in Japanese patients with advanced colorectal cancer. Cancer Chemother Pharmacol 68(2):279–284. https://doi.org/10.1007/s00280-010-1485-8
Hirata K, Nagata N, Kato T, Okuyama Y, Andoh H, Takahashi K, Oba K, Sakamoto J, Hazama S, Mishima H (2014) Prospective phase II trial of second-line FOLFIRI in patients with advanced colorectal cancer including analysis of UGT1A1 polymorphisms: FLIGHT 2 study. Anticancer Res 34(1):195–201
Liu CY, Chen PM, Chiou TJ, Liu JH, Lin JK, Lin TC, Chen WS, Jiang JK, Wang HS, Wang WS (2008) UGT1A1*28 polymorphism predicts irinotecan-induced severe toxicities without affecting treatment outcome and survival in patients with metastatic colorectal carcinoma. Cancer 112(9):1932–1940. https://doi.org/10.1002/cncr.23370
Li M, Wang Z, Guo J, Liu J, Li C, Liu L, Shi H, Liu L, Li H, Xie C, Zhang X, Sun W, Fang S, Bi X (2014) Clinical significance of UGT1A1 gene polymorphisms on irinotecan-based regimens as the treatment in metastatic colorectal cancer. OncoTargets Ther 7:1653–1661. https://doi.org/10.2147/OTT.S67867
Cecchin E, Innocenti F, D’Andrea M, Corona G, De Mattia E, Biason P, Buonadonna A, Toffoli G (2009) Predictive role of the UGT1A1, UGT1A7, and UGT1A9 genetic variants and their haplotypes on the outcome of metastatic colorectal cancer patients treated with fluorouracil, leucovorin, and irinotecan. J Clin Oncol 27(15):2457–2465. https://doi.org/10.1200/JCO.2008.19.0314
Levesque E, Belanger AS, Harvey M, Couture F, Jonker D, Innocenti F, Cecchin E, Toffoli G, Guillemette C (2013) Refining the UGT1A haplotype associated with irinotecan-induced hematological toxicity in metastatic colorectal cancer patients treated with 5-fluorouracil/irinotecan-based regimens. J Pharmacol Exp Therap 345(1):95–101. https://doi.org/10.1124/jpet.112.202242
Hazama S, Mishima H, Tsunedomi R, Okuyama Y, Kato T, Takahashi K, Nozawa H, Ando H, Kobayashi M, Takemoto H, Nagata N, Kanekiyo S, Inoue Y, Hamamoto Y, Fujita Y, Hinoda Y, Okayama N, Oba K, Sakamoto J, Oka M (2013) UGT1A1*6, 1A7*3, and 1A9*22 genotypes predict severe neutropenia in FOLFIRI-treated metastatic colorectal cancer in two prospective studies in Japan. Cancer Sci 104(12):1662–1669. https://doi.org/10.1111/cas.12283
McLeod HL, Sargent DJ, Marsh S, Green EM, King CR, Fuchs CS, Ramanathan RK, Williamson SK, Findlay BP, Thibodeau SN, Grothey A, Morton RF, Goldberg RM (2010) Pharmacogenetic predictors of adverse events and response to chemotherapy in metastatic colorectal cancer: results from North American Gastrointestinal Intergroup Trial N9741. J Clin Oncol 28(20):3227–3233. https://doi.org/10.1200/JCO.2009.21.7943
Williamson SC, Mitter R, Hepburn AC, Wilson L, Mantilla A, Leung HY, Robson CN, Heer R (2013) Characterisations of human prostate stem cells reveal deficiency in class I UGT enzymes as a novel mechanism for castration-resistant prostate cancer. Br J Cancer 109(4):950–956. https://doi.org/10.1038/bjc.2013.399
Funding
This study was funded by Grant no. 81372664 from the National Natural Science Foundation of China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The research was prospectively reviewed and approved by the Ethical Committee of Huazhong University of Science and Technology under reference number NCT01282658 (registered at http://www.clinicaltrials.gov).
Human/animal rights
This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yu, Q., Zhang, T., Xie, C. et al. UGT1A polymorphisms associated with worse outcome in colorectal cancer patients treated with irinotecan-based chemotherapy. Cancer Chemother Pharmacol 82, 87–98 (2018). https://doi.org/10.1007/s00280-018-3595-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-018-3595-7