Abstract
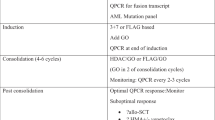
Acute myeloid leukaemia (AML) with t (8;21) or inv (16), called core binding factor (CBF) AML, has a favourable prognosis. However, some CBF-AML patients have persistent measurable residual disease (MRD) and are more likely to relapse after standard chemotherapy treatment. The CAG regimen, composed of cytarabine, aclarubicin and granulocyte colony-stimulating factor, has been proven to be effective and safe in treating refractory AML patients. We performed a retrospective study to evaluate the efficacy of the CAG regimen to eliminate MRD detected by RUNX1::RUNX1T1 and CBFβ::MYH11 transcript levels by quantitative polymerase chain reaction (Q-PCR) among 23 patients. Molecular response was defined as the ratio of fusion transcript after treatment to that before treatment less than or equal to 0.5. The molecular response rate and median decrease ratio of fusion transcripts at the molecular level of the CAG regimen were 52% and 0.53, respectively. The median fusion transcripts before CAG treatment was 0.25% whereas after CAG was 0.11%. Among the 15 patients who had a poor molecular response to the high/intermediate-dose cytarabine regimen, the median decrease ratios of transcripts at the molecular level of high/intermediate-dose cytarabine and CAG were 1.55 and 0.53 (P = 0.028), respectively, and 6 of 15 patients achieved a molecular response to CAG (40%). The median disease-free survival was 18 months, and the overall survival rate at 3 years among all patients was 72.7% ± 10.7%. The common grades 3–4 adverse events were nausea (100%), thrombocytopenia (39%) and neutropenia (37.5%). The CAG regimen may have activity in CBF-AML patients and could provide a new option for patients who have a poor molecular response to high/intermediate-dose cytarabine.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Opatz S, Bamopoulos SA, Metzeler KH et al (2020) The clinical mutatome of core binding factor leukemia. Leukemia 34:1553–1562
Speck NA, Gilliland DG (2002) Core-binding factors in haematopoiesis and leukaemia. Nat Rev Cancer 2:502–513
Döhner H, Estey E, Grimwade D et al (2017) Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 129:424–447
Borthakur G, Kantarjian H (2021) Core binding factor acute myelogenous leukemia-2021 treatment algorithm. Blood Cancer J 11:114
Löwenberg B, Downing JR, Burnett A (1999) Acute myeloid leukemia. N Engl J Med 341:1051–1062
Phillips GL, Reece DE, Shepherd JD et al (1991) High dose cytarabine and daunorubicin induction and postremission chemotherapy for the treatment of acute myelogenous leukemia in adults. Blood 77:1429–1435
Appelbaum FR, Kopecky KJ, Tallman MS et al (2006) The clinical spectrum of adult acute myeloid leukaemia associated with core binding factor translocations. Br J Haematol 135:165–173
Schlenk RF, Benner A, Krauter J et al (2004) Individual patient data-based meta-analysis of patients aged 16 to 60 years with core binding factor acute myeloid leukemia: a survey of the German Acute Myeloid Leukemia Intergroup. J Clin Oncol 22:3741–3750
Marcucci G, Mrózek K, Ruppert AS et al (2005) Prognostic factors and outcome of core binding factor acute myeloid leukemia patients with t(8;21) differ from those of patients with inv(16): a cancer and leukemia group B study. J Clin Oncol 23:5705–5717
Loken MR, Alonzo TA, Pardo L et al (2012) Residual disease detected by multidimensional flow cytometry signifies high relapse risk in patients with de novo acute myeloid leukemia: a report from Children’s Oncology Group. Blood 120:1581–1588
Freeman SD, Virgo P, Couzens S et al (2013) Prognostic relevance of treatment response measured by flow cytometric residual disease detection in older patients with acute myeloid leukemia. J Clin Oncol: Off J Ame Soc Clin Oncol 31:4123–4131
Grimwade D, Freeman SD (2014) Defining minimal residual disease in acute myeloid leukemia: which platforms are ready for “prime time”? Blood 124:3345–3355
Boddu P, Gurguis C, Sanford D et al (2018) Response kinetics and factors predicting survival in core-binding factor leukemia. Leukemia 32:2698–2701
Ouyang J, Goswami M, Peng J et al (2016) Comparison of multiparameter flow cytometry immunophenotypic analysis and quantitative RT-PCR for the detection of minimal residual disease of core binding factor acute myeloid leukemia. Am J Clin Pathol 145:769–777
Wei G, Ni W, Chiao J-w et al (2011) A meta-analysis of CAG (cytarabine, aclarubicin, G-CSF) regimen for the treatment of 1029 patients with acute myeloid leukemia and myelodysplastic syndrome. J Hematol Oncol 4:46
Zhu HH, Jiang H, Jiang B et al (2013) Cytarabine, aclarubicin and granulocyte colony-stimulating factor regimen represents an effective and safe salvage regimen for patients with acute myeloid leukemia refractory to first course of induction chemotherapy. Leuk Lymphoma 54:2452–2457
Pollyea DA, Bixby D, Perl A et al (2021) NCCN guidelines insights: acute myeloid leukemia, Version 2.2021. J Natl Compr Canc Netw 19:16–27
Dillon R, Hills R, Freeman S et al (2020) Molecular MRD status and outcome after transplantation in NPM1-mutated AML. Blood 135:680–688
DiNardo CD, Jonas BA, Pullarkat V et al (2020) Azacitidine and venetoclax in previously untreated acute myeloid leukemia. N Engl J Med 383:617–629
Short NJ, Zhou S, Fu C et al (2020) Association of measurable residual disease with survival outcomes in patients with acute myeloid leukemia: a systematic review and meta-analysis. JAMA Oncol 6:1890–1899
Döhner H, Wei AH, Appelbaum FR et al (2022) Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood 140:1345–1377
Cazzaniga ME, Dionisio MR, Riva F (2017) Metronomic chemotherapy for advanced breast cancer patients. Cancer Lett 400:252–258
Shaked Y, Emmenegger U, Man S et al (2005) Optimal biologic dose of metronomic chemotherapy regimens is associated with maximum antiangiogenic activity. Blood 106:3058–3061
Abu Lila AS, Ishida T (2017) Metronomic chemotherapy and nanocarrier platforms. Cancer Lett 400:232–242
Löwenberg B, van Putten W, Theobald M et al (2003) Effect of priming with granulocyte colony-stimulating factor on the outcome of chemotherapy for acute Myeloid Leukemia. N Engl J Med 349:743–752
Tanaka M (1993) Recombinant GM-CSF modulates the metabolism of cytosine arabinoside in leukemic cells in bone marrow. Leuk Res 17:585–592
Miyauchi JKC, Wang C et al (1989) Growth factors influence the sensitivity of leukemic stem cells to cytosine arabinoside in culture. Blood 73:1272–1278
Te Boekhorst PA, L B, Sonneveld P (1994) Hematopoietic growth factor stimulation and cytarabine cytotoxicity in vitro: effects in untreated and relapsed or primary refractory acute myeloid leukemia cells. Leukemia 8:1480–1486
Te Boekhorst PA, L B, Vlastuin M, Sonneveld P (1993) Enhanced chemosensitivity of clonogenic blasts from patients with acute myeloid leukemia by G-CSF, IL-3 or GM-CSF stimulation. Leukemia 7:1191–1198
Löwenberg B, Pabst T, Vellenga E et al (2011) Cytarabine dose for acute myeloid leukemia. N Engl J Med 364:1027–1036
Miyawaki S, Ohtake S, Fujisawa S et al (2011) A randomized comparison of 4 courses of standard-dose multiagent chemotherapy versus 3 courses of high-dose cytarabine alone in postremission therapy for acute myeloid leukemia in adults: the JALSG AML201 study. Blood 117:2366–2372
Acknowledgements
We thank the faculty and staff of the Clinical Unit of the Department of Haematology, the First Affiliated Hospital, Zhejiang University. We also appreciate Zhejiang Province Key Laboratory of Haematology Oncology Diagnosis and Treatment for their services in Q-PCR and quality control.
Author information
Authors and Affiliations
Contributions
ZH designed and supervised the clinical study. WH, ZL, YX and JJ enrolled patients and took care of the patients; SY and CJ collected and analysed the clinical data; SY performed statistical analysis. ZH, SY and ZY drafted and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Institute Research Ethics Committee of the first Affiliated Hospital, College of Medicine, Zhejiang University.
Consent for publication
Written informed consent for publication was obtained.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shen, Yj., Zhang, Y., Chang, J. et al. CAG (cytarabine, aclarubicin and granulocyte colony-stimulating factor) regimen for core binding factor acute myeloid leukaemia with measurable residual disease. Ann Hematol 102, 1731–1738 (2023). https://doi.org/10.1007/s00277-023-05213-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-023-05213-6