Abstract
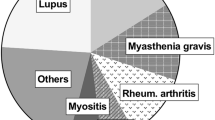
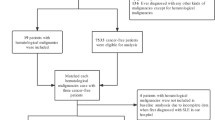
The incidence and clinical implications of autoimmune diseases (ADs) in patients with non-Hodgkin’s lymphoma(NHL) remain unclear. The aim of this study was to examine the prevalence of ADs in NHL and define the clinical characteristics and prognosis of AD-associated NHL patients. Patients diagnosed with NHL in our institute between 1995 and 2017 were retrospectively reviewed to assess the incidence of ADs. Of 4880 patients with NHL, 140 (2.9%) presented with autoimmunity, with a total of 24 ADs. The most common AD was Sjögren syndrome, followed by autoimmune cytopenia, psoriasis, rheumatoid arthritis, etc. Psoriasis and rheumatoid arthritis were significantly associated with pre-existing ADs, whereas autoimmune cytopenia was significantly associated with secondary AD. Sjögren syndrome was significantly associated with B-cell lymphoma, and systemic vasculitis was significantly associated with T-cell lymphoma. Patients with AD-associated NHL had a high frequency of extranodal involvement(87%), with significant associations between specific extranodal sites of lymphoma and subtypes of ADs. Among patients with available data on pre-treatment peripheral blood Epstein-Barr virus (EBV) DNA(n = 68), elevated EBV-DNA load was observed in a variety of NHL subtypes, including 20% of marginal zone lymphoma and 14.3% of follicular lymphoma patients. In a matched-pair analysis, survival did not differ significantly between NHL patients with and without ADs. However, for NHL patients with pre-existing ADs, a prior history of systemic corticosteroids therapy was significantly associated with worse survival (HR = 7.33, P = 0.006). Taken together, our data suggest that a broad spectrum of ADs is associated with NHL, and AD-associated NHL has distinct features with regard to clinical manifestations and prognosis.






Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL et al (2012) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108
Chen W, Zheng R, Baade PD et al (2016) Cancer statistics in China, 2015. CA Cancer J Clin 66:115–132
Smedby KE, Hjalgrim H, Askling J, Chang ET, Gregersen H, Porwit-MacDonald A, Sundström C, Åkerman M, Melbye M, Glimelius B, Adami HO (2006) Autoimmune and chronic inflammatory disorders and risk of non-Hodgkin lymphoma by subtype. J Natl Cancer Inst 98:51–60
Ekstrom Smedby K, Vajdic CM, Falster M, Engels EA, Martinez-Maza O, Turner J, Hjalgrim H, Vineis P, Seniori Costantini A, Bracci PM, Holly EA, Willett E, Spinelli JJ, la Vecchia C, Zheng T, Becker N, de Sanjose S, Chiu BCH, Dal Maso L, Cocco P, Maynadie M, Foretova L, Staines A, Brennan P, Davis S, Severson R, Cerhan JR, Breen EC, Birmann B, Grulich AE, Cozen W (2008) Autoimmune disorders and risk of non-Hodgkin lymphoma subtypes: a pooled analysis within the InterLymph consortium. Blood 111:4029–4038
Fallah M, Liu X, Ji J, Försti A, Sundquist K, Hemminki K (2014) Autoimmune diseases associated with non-Hodgkin lymphoma: a nationwide cohort study. Ann Oncol 25:2025–2030
Hauswirth AW, Skrabs C, Schützinger C, Gaiger A, Lechner K, Jäger U (2007) Autoimmune hemolytic anemias, Evans' syndromes, and pure red cell aplasia in non-Hodgkin lymphomas. Leuk Lymphoma 48:1139–1149
Jardin F (2008) Development of autoimmunity in lymphoma. Expert Rev Clin Immunol 4:247–266
Baecklund E, Smedby KE, Sutton LA, Askling J, Rosenquist R (2014) Lymphoma development in patients with autoimmune and inflammatory disorders—what are the driving forces? Semin Cancer Biol 24:61–70
Hoshida Y, Xu JX, Fujita S et al (2007) Lymphoproliferative disorders in rheumatoid arthritis: clinicopathological analysis of 76 cases in relation to methotrexate medication. J Rheumatol 34:322–331
Kotlyar DS, Lewis JD, Beaugerie L, Tierney A, Brensinger CM, Gisbert JP, Loftus EV Jr, Peyrin-Biroulet L, Blonski WC, van Domselaar M, Chaparro M, Sandilya S, Bewtra M, Beigel F, Biancone L, Lichtenstein GR (2015) Risk of lymphoma in patients with inflammatory bowel disease treated with azathioprine and 6-mercaptopurine: a meta-analysis. Clin Gastroenterol Hepatol 13:847–858
Ichikawa A, Arakawa F, Kiyasu J, Sato K, Miyoshi H, Niino D, Kimura Y, Takeuchi M, Yoshida M, Ishibashi Y, Nakashima S, Sugita Y, Miura O, Ohshima K (2013) Methotrexate iatrogenic lymphoproliferative disorders in rheumatoid arthritis: histology, Epstein–Barr virus, and clonality are important predictors of disease progression and regression. Eur J Haematol 91:20–28
Vos AC, Bakkal N, Minnee RC et al (2011) Risk of malignant lymphoma in patients with inflammatory bowel diseases: a Dutch nationwide study. Inflamm Bowel Dis 17:1837–1845
Martin DN, Mikhail IS, Landgren O (2009) Autoimmunity and hematologic malignancies: associations and mechanisms. Leuk Lymphoma 50:541–550
Duhrsen U, Augener W, Zwingers T, Brittinger G (1987) Spectrum and frequency of autoimmune derangements in lymphoproliferative disorders: analysis of 637 cases and comparison with myeloproliferative diseases. Br J Haematol 67:235–239
Váróczy L, Gergely L, Zeher M et al (2002) Malignant lymphoma-associated autoimmune diseases—a descriptive epidemiological study. Rheumatol Int 22:233–237
Váróczy L, Páyer E, Kádár Z, Gergely L, Miltényi Z, Magyari F, Szodoray P, Illés Á (2012) Malignant lymphomas and autoimmunity—a single center experience from Hungary. Clin Rheumatol 31:219–224
Jachiet V, Mekinian A, Carrat F, Grignano E, Retbi A, Boffa JJ, Ronco P, Rondeau E, Sellam J, Berenbaum F, Chazouillères O, Capron J, Alamowitch S, Chasset F, Frances C, Coppo P, Fain O, on behalf of French Network of systemic and immune disorders associated with hemopathies and cancer (MINHEMON) (2017) Autoimmune manifestations associated with lymphoma: characteristics and outcome in a multicenter retrospective cohort study. Leuk Lymphoma 59:1399–1405. https://doi.org/10.1080/10428194.2017.1379075
Swerdlow SH, Campo E, Harris NL et al (2008) WHO classification of tumours of haematopoietic and lymphoid tissues. In: Bosman FT, Jaffe ES, Lakhani SR, Ohgaki H (eds) World Health Organization classification of Tumours. IARC, Lyon, p 439
Hochberg MC (1997) Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40:1725
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO, Birnbaum NS, Burmester GR, Bykerk VP, Cohen MD, Combe B, Costenbader KH, Dougados M, Emery P, Ferraccioli G, Hazes JM, Hobbs K, Huizinga TW, Kavanaugh A, Kay J, Kvien TK, Laing T, Mease P, Menard HA, Moreland LW, Naden RL, Pincus T, Smolen JS, Stanislawska-Biernat E, Symmons D, Tak PP, Upchurch KS, Vencovsky J, Wolfe F, Hawker G (2010) 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis 69:1580–1588
Koff JL, Rai A, Flowers CR (2018) Characterizing autoimmune disease-associated diffuse large B-cell lymphoma in a SEER-Medicare cohort. Clin Lymphoma Myeloma Leuk 18:e115–e121
Johnsen SJ, Brun JG, Gøransson LG, Småstuen MC, Johannesen TB, Haldorsen K, Harboe E, Jonsson R, Meyer PA, Omdal R (2013) Risk of non-Hodgkin’s lymphoma in primary Sjögren’s syndrome: a population-based study. Arthritis Care Res (Hoboken) 65:816–821
Liang Y, Yang Z, Qin B, Zhong R (2014) Primary Sjogren’s syndrome and malignancy risk: a systematic review and meta-analysis. Ann Rheum Dis 73:1151–1156
Willemze R, Jansen PM, Cerroni L, Berti E, Santucci M, Assaf C, Canninga-van Dijk MR, Carlotti A, Geerts ML, Hahtola S, Hummel M, Jeskanen L, Kempf W, Massone C, Ortiz-Romero PL, Paulli M, Petrella T, Ranki A, Peralto JLR, Robson A, Senff NJ, Vermeer MH, Wechsler J, Whittaker S, Meijer CJLM (2008) Subcutaneous panniculitis-like T-cell lymphoma: definition, classification and prognostic factors. An EORTC cutaneous lymphoma group study of 83 cases. Blood 111:838–845
Tisi MC, Cupelli E, Santangelo R, Maiolo E, Alma E, Giachelia M, Martini M, Bellesi S, D’Alò F, Voso MT, Pompili M, Leone G, Larocca LM, Hohaus S (2016) Whole blood EBV-DNA predicts outcome in diffuse large B-cell lymphoma. Leuk Lymphoma 57:628–634
Ito Y, Kimura H, Maeda Y, Hashimoto C, Ishida F, Izutsu K, Fukushima N, Isobe Y, Takizawa J, Hasegawa Y, Kobayashi H, Okamura S, Kobayashi H, Yamaguchi M, Suzumiya J, Hyo R, Nakamura S, Kawa K, Oshimi K, Suzuki R (2012) Pretreatment EBV-DNA copy number is predictive of response and toxicities to SMILE chemotherapy for extranodal NK/T-cell lymphoma, nasal type. Clin Cancer Res 18:4183–4190
Kim YR, Kim SJ, Cheong JW et al (2017) Pretreatment Epstein-Barr virus DNA in whole blood is a prognostic marker in peripheral T-cell lymphoma. Oncotarget 8:92312–92323
Grywalska E, Rolinski J (2015) Epstein-Barr virus-associated lymphomas. Semin Oncol 42:291–303
Shih YH, Yang Y, Chang KH, Chen YH, Teng CLJ (2018) Clinical features and outcome of lymphoma patients with pre-existing autoimmune diseases. Int J Rheum Dis 21:93–101
Xing K, Gu B, Zhang P, Wu X (2015) Dexamethasone enhances programmed cell death 1 (PD-1) expression during T cell activation: an insight into the optimum application of glucocorticoids in anti-cancer therapy. BMC Immunol 16:39
Schmidt S, Rainer J, Ploner C, Presul E, Riml S, Kofler R (2004) Glucocorticoid-induced apoptosis and glucocorticoid resistance: molecular mechanisms and clinical relevance. Cell Death Differ 11(Suppl 1):S45–S55
Acknowledgments
The authors thank the patients, medical staff, and physicians who participated in this study. The authors thank the assistance of department of hematology and department of rheumatology for study collaboration. The authors also acknowledge the department of medical record library for medical record provision.
Author information
Authors and Affiliations
Contributions
S.H., D.Z., and W.Z. designed the research study; all authors contributed to the acquisition of data and the statistical analysis and interpretation of data; all authors contributed in drafting the article or revising it critically for important intellectual content; all authors gave the final approval of the version published.
Corresponding author
Ethics declarations
Conflict of interest disclosures
The authors declare no competing financial interests for this study.
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. Informed consent was obtained from all patients for being included in the study.
Electronic supplementary material
Supplementary Table 1
(DOCX 12 kb)
Supplementary Table 2
(DOCX 12 kb)
Rights and permissions
About this article
Cite this article
Hu, S., Zhou, D., Wu, Y. et al. Autoimmune disease-associated non-Hodgkin’s lymphoma—a large retrospective study from China. Ann Hematol 98, 445–455 (2019). https://doi.org/10.1007/s00277-018-3515-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-018-3515-2