Abstract
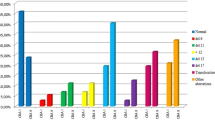
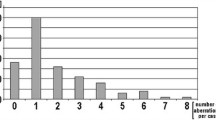
Although interphase fluorescence in situ hybridization (FISH) is routinely used in chronic lymphocytic leukemia (CLL), differences in the chromosomal pattern with respect to morphological subtypes of CLL (typical CLL, CLL/PL, PLL) are still under debate. We studied 153 patients with CLL and correlated cytomorphology on peripheral blood stains with FISH analysis and other prognostic markers. The percentage of prolymphocytes was calculated as a continuous variable and followed published thresholds in parallel while being correlated to FISH analysis. Higher percentages of prolymphocytes were associated significantly with deletion of 17p13. Deletion of 17p13 was most frequently observed in patients with more than 30% prolymphocytes. Trisomy 12 was found mainly in cases with 6–30% prolymphocytes. The percentage of prolymphocytes did not correlate with deletions of 11q23 or with 13q14 abnormalities. In conclusion, we suggest that further research focus on the percentage of prolymphocytes in CLL. Doing so, biologically relevant thresholds for the percentages of prolymphocytes in the peripheral blood and their association to underlying genetic markers could be investigated together with other biologically and especially prognostic markers.
Similar content being viewed by others
References
Bennett JM, Gatovsky D, Daniel MT (1989) Proposals for the classification of chronic (mature) B and T lymphoid leukemias. French-American-British (FAB) Cooperative Group. J Clin Pathol 42:567–584
Bennett JM (2002) The FAB/MIC/WHO proposals for the classification of the chronic lymphoid leukemias. Rev Clin Exp Hematol 6:330–334
Bigoni R, Cuneo A, Roberti MG, Bardi A, Rigolin GM, Piva N, Scapoli G, Spanedda R, Negrini M, Bullrich F, Veronese ML, Croce CM, Castoldi G (1997) Chromosome aberrations in atypical chronic lymphocytic leukemia: a cytogenetic and interphase cytogenetic study. Leukemia 11:1933–1940
Buhmann R, Kurzeder C, Rehklau J, Westhaus D, Bursch S, Hiddemann W, Haferlach T, Hallek M, Schoch C (2002) CD40L stimulation enhances the ability of conventional metaphase cytogenetics to detect chromosome aberrations in B-cell chronic lymphocytic leukemia cells. Br J Haematol 118:968–975
Cano I, Martinez E, Quevedo J, Pinilla J, Martin-Recio A, Rodriguez A, Castaneda A, Lopez R, Perez-Pino T, Hernandez-Navarro F (1996) Trisomy 12 and p53 deletion in chronic lymphocytic leukemia detected by fluorescence in situ hybridization: association with morphology and resistance to conventional chemotherapy. Cancer Genet Cytogenet 90:118–124
Criel A, Michaux L, De Wolf-Peeters C (1999) The concept of typical and atypical chronic lymphocytic leukemia. Leuk Lymphoma 33:33–45
Criel A, Verhoef G, Vlietinck R, Mecucci C, Billiet J, Michaux L, Meeus P, Louwagie A, Van Orshoven A, Van Hoff A, Boogaerts M, Van den Berghe H, De Wolf-Peeters C (1997) Further characterization of morphologically defined typical and atypical CLL: a clinical, immunophenotypic, cytogenetic and prognostic study on 390 cases. Br J Haematol 97:383–391
D’Arena G, Dell’Olio M, Cascavilla N, Dell’Olio M, Di Renzo N, Perla G, Savino L, Carotenuto M (2001) CD38 expression correlates with adverse biological features and predicts poor clinical outcome in B-cell chronic lymphocytic leukemia. Leuk Lymphoma 42:109–114
D’Arena G, Dell’Olio M, Pellegrino M, Cascavilla N, Perla G, Savino L, Greco MM (2001) Morphologically typical and atypical B-cell chronic lymphocytic leukemias display a different pattern of surface antigenic density. Leuk Lymphoma 42:649–654
Döhner H, Stilgenbauer S, Benner A, Leupolt E, Krober A, Bullinger L, Döhner K, Bentz M, Lichter P (2000) Genomic aberrations and survival in chronic lymphocytic leukemia. N Engl J Med 343:1910–1916
Frater JL, McCarron K, Hammel J, Shapiro JL, Miller ML, Tubbs RR, Pettay J, Hsi ED (2001) Typical and atypical chronic lymphocytic leukemia differ clinically and immunophenotypically. Am J Clin Pathol 116:655–664
Geisler CH, Philip P, Christensen E, Hou-Jensen K, Tinggaard Pedersen N, Myhre Jensen O, Thorling K, Andersen E, Birgens HS, Drivsholm A, Ellegaard J, Larsen JK, Plesner T, Brown P, Krah Andersen P, Mork Hansen M (1997) In B-cell chronic lymphocytic leukemia chromosome 17 abnormalities and not trisomy 12 are the single most important cytogenetic abnormalities for the prognosis: a cytogenetic and immunophenotypic study of 480 unselected newly diagnosed patients. Leuk Res 21:1011–1023
Hallek M, Langenmayer I, Nerl C, Knauf W, Dietzfelbinger H, Ostwald M, Busch R, Kuhn-Hallek I, Thiel E, Emmerich B (1999) Elevated serum thymidine kinase levels identify a subgroup at high risk of disease progression in early, nonsmoldering chronic lymphocytic leukemia. Blood 93:1732–1737
Hallek M, Wanders L, Ostwald M, Busch R, Senekowitsch RSS, Schick HD, Kuhn-Hallek I, Emmerich B (1996) Serum beta(2)-microglobulin and serum thymidine kinase are independent predictors of progression-free survival in chronic lymphocytic leukemia and immunocytoma. Leuk Lymphoma 22:439–447
Hercher C, Robain M, Davi F, Garand R, Flandrin G, Valensi F, Vandeputte H, Albert A, Maynadie M, Troussard X, Hoerni Simon G, Lespinasse J, Portefaix G, Merle-Beral H (2001) A multicentric study of 41 cases of B-prolymphocytic leukemia: two evolutive forms. Leuk Lymphoma 42:981–987
Hjalmar V, Kimby E, Matutes E, Sundström C, Wallvik J, Hast R (2000) Atypical lymphocytes in B-cell chronic lymphatic leukemia and trisomy 12 studied by conventional staining combined with fluorescence in situ hybridization. Leuk Lymphoma 37:571–576
Hjalmar V, Hast R, Kimby E (2002) Cell surface expression of CD 25, CD 54, and CD 95 on B- and T-cells in chronic lymphocytic leukemia in relation to trisomy 12, atypical morphology and clinical course. Eur J Haematol 68:127–134
Jaffe ES, Harris NL, Stein H, Vardiman JW (2001) Pathology and genetics of tumors and hematopoietic and lymphoid tissues. World Health Organization classification of tumours. IARCPress, Lyon
Juliusson G, Oscier DG, Fitchett M, Ross FM, Stockdill G, Mackie MJ, Parker AC, Castoldi GL, Guneo A, Knuutila S (1990) Prognostic subgroups in B-cell chronic lymphocytic leukemia defined by specific chromosomal abnormalities. N Engl J Med 323:720–724
Lens D, Matutes E, Catovsky D, Coignet LJA (2000) Frequent deletions at 11q23 and 13q14 in B cell prolymphocytic leukemia (B-PLL). Leukemia 14:427–430
Lens D, Dyer M, Garcia-Marco J, De Schouwer PJJ, Hamoudi R, Jones D, Farahat N, Matutes E (1997) p53 abnormalities in CLL are associated with excess of prolymphocytes and poor prognosis. Br J Haematol 99:848–857
Magnac C, Porcher R, Davi F, Nataf J, Payelle-Brogard B, Tang RP, Oppezzo P, Levy V, Dighiero G (2003) Predictive value of serum thymidine kinase level for Ig-V mutational status in B-CLL leukemia. 17:133–137
Matutes E, Oscier D, Garcia-Marco J, Ellis J, Copplestone A, Gillingham A, Gillingham R, Hamblin T, Lens D, Swansbury J, Catovsky D (1996) Trisomy 12 defines a group of CLL with atypical morphology: correlation between cytogenetic, clinical and laboratory features in 544 patients Br J Haematol 92:382–388
Matutes E, Polliack A (2000) Morphological and immunophenotypic features of chronic lymphocytic leukemia. Rev Clin Exp Hematol 4:22–47
Mauro FR, Gentile M, Mancini F, Giannarelli D, Guarini A, De Propriis MS, Cerretti R, Foa R (2002) Prognostic significance of lymphocyte morphology in patients with advanced chronic lymphocytic leukemia treated with first line therapy of fludarabine and prednisone. Haematologica 87:602–608
Montillo M, Tedeschi A, O’Brien S, Di Raimondo F, Lerner S, Ferrajoli A, Morra E, Keating MJ (2003) Phase II study of cladribine and cyclophosphamide in patients with chronic lymphocytic leukemia and prolymphocytic leukemia. Blood 97:114–120
O’Connor SJM, Su‘ut L, Morgan GJ, Jack AS (2000) The relationship between typical and atypical B-CLL. A comparative genomic hybridization-based study. Hematopathology 114:448–458
Oscier DG, Gardiner A, Mould S, Glide S, Davis ZA, Ibbotson RE, Corcoran MM, Chapman RM, Thomas PW, Copplestone JA, Orchard JA, Hamblin TJ (2002) Multivariate analysis of prognostic factors in CLL: clinical stage, IGVH gene mutational status, and loss or mutation of the p53 gene are independent prognostic factors. Blood 100:1177–1184
Oscier DG, Matutes E, Copplestone A, Pickering RM, Chapman R, Gillingham R, Catovsky D, Hamblin TJ (1997) Atypical lymphocyte morphology: an adverse prognostic factor for disease progression in stage A CLL independent of trisomy 12. Br J Haematol 98:934–939
Que TH, Garcia Marco J, Ellis J, Matutes E, Brito Babapulle VB, Boyle S, Catovsky D (1993) Trisomy 12 in chronic lymphocytic leukemia detected by fluorescence in situ hybridization: analysis by stage, immunophenotype, and morphology. Blood 82:571–575
Shvidel L, Shtalrid M, Bassous L, Bassous L, Klepfish A, Vorst E, Berrebi A (1999) B-cell prolymphocytic leukemia: a survey of 35 patients emphasizing heterogeneity, prognostic factors and evidence for a group with an indolent course. Leuk Lymphoma 33:169–179
Wiestner A, Rosenwald A, Barry TS, Wright G, Davis RE, Henrickson SE, Zhao H, Ibbotson RE, Orchard JA, Davis Z, Stetler-Stevenson M, Raffeld M, Arthur DC, Marti GE, Wilson WH, Hamblin TJ, Oscier DG, Staudt LM (2003) ZAP-70 expression identifies a chronic lymphocytic leukemia subtype with unmutated immunoglobulin genes, inferior clinical outcome, and distinct gene expression profile. Blood 101:4944–4951
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bacher, U., Kern, W., Schoch, C. et al. Discrimination of chronic lymphocytic leukemia (CLL) and CLL/PL by cytomorphology can clearly be correlated to specific genetic markers as investigated by interphase fluorescence in situ hybridization (FISH). Ann Hematol 83, 349–355 (2004). https://doi.org/10.1007/s00277-004-0869-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-004-0869-4