Abstract
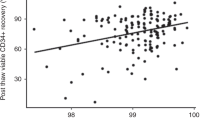
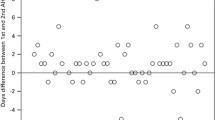
Cryopreservation of peripheral blood stem cells (PBSC) mobilized by chemotherapy combined with or without granulocyte colony-stimulating factor (G-CSF) is an essential part of procedure for anti-cancer strategies. We evaluated whether a higher cell concentration (2×108/ml) without the use of a programmed freezer was acceptable for the storage of mobilized PBSC in an autologous setting. Mobilized PBSC were enriched to mononuclear cells (MNC) by Percoll separation and then frozen at cell concentrations of 2–5×107/ml (group I, n=20) or 2×108/ml (group II, n=44) without the use of a programmed freezer using 5% DMSO, 6% hydroxy ethyl starch, and 4% autologous serum or human albumin. CD34+ cells purified by ISOLEX300 were frozen at 2×107/ml (group III, n=22) using the same method. The median recovery rates of CD34+ cells and CFU-GM were, respectively, n.d. (not determined) and 88% in group I, 103 and 64% in group II, and 98 and 53% in group III. There was a statistical significance between the recovery rate of CFU-GM in group III and that in group I (p=0.02). The median percentage of cell viability after thawing in each group was 89, 87, and 75%, respectively. The median numbers of days after PBSCT to achieve a WBC of >1.0×109/l, an absolute neutrophil count of >0.5×109/l, and a platelet count of >50×109/l were, respectively, 11, 11 and 15 in group I; 12, 12 and 16 in group II; and 12, 12 and 27 in group III. These results suggest that enriched MNC from mobilized PBSC could be frozen at a higher cell concentration (2×108/ml) without the use of a programmed freezer, leading to reduction of the toxicities associated with infusion of thawed cells and of costly space required for cell storage.
Similar content being viewed by others
References
Goldman JM, Schmitz N, Niethammer D, Gratwohl A (1998) Allogeneic and autologous transplantation for hematological disease, solid tumors and immune disorders: current practice in Europe in 1998. Bone Marrow Transplant 21:1–7
Goldman JM, Th’ng KH, Park DS, Spiers AS, Lowenthal RM, Ruutu T (1979) Collection, cryopreservation and subsequent viability of haemopoietic stem cells intended for treatment of chronic granulocytic leukemia in blast-cell transformation. Br J Haematol 40:185–195
Stroncek DF, Fautsh SK, Lasky LC, Hurd DD, Ramsay NK, McCullough J (1991) Adverse reactions in patients transfused with cryopreserved marrow. Transfusion 31:521–526
Okamoto Y, Takaue Y, Saito S, Shimizu T, Suzue T, Abe T, Sato J, Hirao A, Watanabe T, Kawano Y, Kuroda Y (1993) Toxicities associated with cryopreserved and thawed peripheral blood stem cell autografts in children with cancer. Transfusion 33:578–581
Al Alessandrino P, Bernasconi P, Caldera D, Colombo A, Bonfichi M, Malcovati L, Klersy C, Martinelli G, MaiocchiM, Pagnucco G, Varettoni M, Perotti C, Bernasconi C (1999) Adverse events occurring during bone marrow or peripheral blood progenitor cell infusion: analysis of 126 cases. Bone Marrow Transplant 23:533–537
Rowley SD (1992) Hematopoietic stem cell cryopreservation: a review of current techniques. J Hematother 1:233–250
Makino S, Harada M, Akashi K, Taniguchi S, Shibuya T, Inaba S, Niho Y (1991) A simplified method for cryopreservation of peripheral blood stem cells at −80°C without rate-controlled freezing. Bone Marrow Transplant 8:239–244
Takaue Y, Abe T, Kawano Y, Suzue T, Saito S, Hirao A, Sato J, Makimoto A, Kawahito M, Watanabe T, Shimokawa T, Kuroda Y (1994) Comparative analysis of engraftment after peripheral blood stem cell autografts cryopreserved by controlled vs uncontrolled-rate method. Bone Marrow Transplant 13:801–804
Rowley SD, Bensinger WI, Gooley TA, Buckner CD (1994) Effect of cell concentration on bone marrow and peripheral blood stem cell cryopreservation. Blood 83:2731–2736
Cabezudo E, Dalmases C, Ruz M, Sanchez JA, Torrico C, Sola C, Querol S, Garcia J (2000) Leukapheresis components may be cryopreserved at high cell concentrations without additional loss of HPC function. Transfusion 40:1223–1227
Takaue Y, Kawano Y, Kuroda Y (1995) Application of recombinant granulocyte colony-stimulating factor in peripheral blood stem-cell transplantation: a pediatric experience. In: Levitt D, Mertelsmann R (eds) Hematopoietic stem cells. Marcel Dekker, New York, pp 611–630
Makimoto A, Kawano Y, Abe T, Okamoto Y, Sato J, Nakagawa R, Watanabe H, Watanabe T, Kuroda Y, Sweet L, Takaue Y (1999) Comparative evaluation of procedures with a Baxter CS-3000 cell separator for collecting peripheral blood cells from children. J Hematother 8:305–310
Kawano Y, Takaue Y, Watanabe T, Abe T, Okamoto Y, Iwai A, Watanabe A, Ito E, Makimoto A, Nakagawa R, Watanabe H, Sato J, Suenaga K, Suzuya H, Ohnishi T, Kanamaru S, Kaneko S, Kuroda Y (1999) Efficacy of the mobilization of peripheral blood stem cells by granulocyte colony-stimulating factor in pediatric donors. Cancer Res 59:3321–3324
Takaue Y, Kawano Y, Watanabe A, Eguchi H, Abe T, Makimoto A, Okamoto Y, Kuroda Y (1996) Transplantation with purified or unmanipulated mobilized blood stem cells in children. In: Ikehara S, Takaku F, Good RA (eds) Bone marrow transplantation: basic and clinical studies. Springer, Berlin, Heidelberg, New York, pp 246–249
Kawano Y, Takaue Y, Watanabe T, Saito S, Abe T, Hirao A, Sato J, Ninomiya T, Suzue T, Koyama T, Shimokawa T, Yokobayashi A, Asano S, Masaoka T, Takaku F, Kuroda Y (1993) Effects of progenitor cell dose and preleukapheresis use of human recombinant granulocyte-macrophage colony-stimulating factor on the recovery of hematopoiesis after blood stem cell autografting in children. Exp Hematol 21:103–108
Takaue Y, Kawano Y, Abe T, Okamoto Y, Suzue T, Shimizu T, Saito S, Sato J, Makimoto A, Nakagawa R, Watanabe T, Ito M, Kuroda Y (1995) Collection and transplantation of peripheral blood stem cells in very small children weighing 20 kg or less. Blood 86:372–380
To LB, Roberts MM, Haylock DN, Dyson PG, Branford AL, Thorp D, Ho JQ, Dart GW, Horvath N, Davy ML, Olweny LM, Juttner CA (1992) Comparison of haematological recovery times and supportive care requirements of autologous recovery phase peripheral blood stem cell transplants, autologous bone marrow transplants and allogeneic bone marrow transplants. Bone Marrow Transplant 9:277–284
Takaue Y, Watanabe T, Kawano Y, Koyama T, Huq M, Suzue T, Abe T, Sato J, Shimokawa T, Kosaka M, Shimizu M, Ogura T, Ninomiya T, Kuroda Y (1989) Isolation and storage of peripheral blood hematopoietic stem cells for autotransplantation into children with cancer. Blood 74:1245–1251
Keung YK, Cobos E, Morgan D, Park M, Dixon S, Wu K, Park CH (1996) High cellular concentration of peripheral blood progenitor cells during cryopreservation adversely affects CFU-GM but not hematopoietic recovery. J Hematother 5:73–77
Balint B, Ivanovic Z, Petakov M, Taseski J, Jovcic G, Stojanovic N, Milenkovic P (1999) The cryopreservation protocol optimal for progenitor recovery is not optimal for preservation of marrow repopulating ability. Bone Marrow Transplant 23:613–619
Acknowledgements
The authors are grateful to Ms. Yasuda for her excellent technical assistance and to the nursing staff of the pediatric ward at the University Hospital of Tokushima.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kawano, Y., Lee, C.L., Watanabe, T. et al. Cryopreservation of mobilized blood stem cells at a higher cell concentration without the use of a programmed freezer. Ann Hematol 83, 50–54 (2004). https://doi.org/10.1007/s00277-003-0817-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-003-0817-8