Abstract
Background and Aims
The aim of this single-center, open-label phase II study was to assess the efficacy of image-guided high-dose-rate (HDR) brachytherapy (iBT) compared with conventional transarterial embolization (cTACE) in unresectable hepatocellular carcinoma.
Methods
Seventy-seven patients were treated after randomization to iBT or cTACE, as single or repeated interventions. Crossover was allowed if clinically indicated. The primary endpoint was time to untreatable progression (TTUP). Eligibility criteria included a Child–Pugh score of ≤ 8 points, absence of portal vein thrombosis (PVT) at the affected liver lobe, and ≤ 4 lesions. Survival was analyzed by using the Cox proportional hazard model with stratification for Barcelona Clinic Liver Cancer (BCLC) stages.
Results
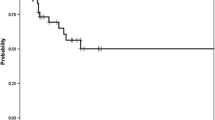
Twenty patients were classified as BCLC-A (iBT/cTACE 8/12), 35 as BCLC-B (16/19), and 22 as BCLC-C (13/9). The 1-, 2-, and 3-year TTUP probabilities for iBT compared with cTACE were 67.5% versus 55.2%, 56.0% versus 27.4%, and 29.5% versus 11.0%, respectively, with an adjusted hazard ratio (HR) of 0.49 (95% confidence interval 0.27–0.89; p = 0.019). The 1-, 2-, and 3-year TTPs for iBT versus cTACE were 56.0% versus 28.2%, 23.9% versus 6.3%, and 15.9% versus 6.3%, respectively, with an adjusted HR of 0.49 (0.29–0.85; p = 0.011). The 1-, 2-, and 3-year OS rates were 78.4% versus 67.7%, 62.0% versus 47.3%, and 36.7% versus 27.0%, respectively, with an adjusted HR of 0.62 (0.33–1.16; p = 0.136).
Conclusions
This explorative phase II trial showed a superior outcome of iBT compared with cTACE in hepatocellular carcinoma and supports proceeding to a phase III trial.




Similar content being viewed by others
Abbreviations
- AASL:
-
American Association for the Study of the Liver
- BCLC:
-
Barcelona Clinic Liver Cancer (staging system)
- CI:
-
Confidence interval
- CLIP:
-
Cancer of the Liver Italian Program
- CT:
-
Computed tomography
- cTACE:
-
Conventional transarterial chemoembolization
- CTCAE:
-
Common Terminology Criteria for Adverse Events
- DEB-TACE:
-
Drug-eluting beads transarterial chemoembolization
- EASL:
-
European Association for the Study of the Liver
- HCC:
-
Hepatocellular carcinoma
- HDR:
-
High dose rate
- HR:
-
Hazard ratio
- iBT:
-
Interstitial brachytherapy
- OS:
-
Overall survival
- PVT:
-
Portal vein thrombosis
- RFA:
-
Radiofrequency ablation
- SBRT:
-
Stereotactic body radiotherapy
- TTP:
-
Time to progression
- TTUP:
-
Time to untreatable progression
References
Jaeck D, Bachellier P, Oussoultzoglou E, Weber JC, Wolf P. Surgical resection of hepatocellular carcinoma. Post-operative outcome and long-term results in Europe: an overview. Liver Transpl. 2004;10:S58–63.
Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet. 2018;391:1301–14.
Cheng BQ, Jia CQ, Liu CT, et al. Chemoembolization combined with radiofrequency ablation for patients with hepatocellular carcinoma larger than 3 cm: a randomized controlled trial. JAMA. 2008;299:1669–77.
Helmberger T, Dogan S, Straub G, et al. Liver resection or combined chemoembolization and radiofrequency ablation improve survival in patients with hepatocellular carcinoma. Digestion. 2007;75:104–12.
Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: chemoembolization improves survival. Hepatology. 2003;37:429–42.
Vogl TJ, Naguib NN, Nour-Eldin NE, et al. Review on transarterial chemoembolization in hepatocellular carcinoma: palliative, combined, neoadjuvant, bridging, and symptomatic indications. Eur J Radiol. 2009;72:505–16.
Cucchetti A, Piscaglia F, Cescon M, Ercolani G, Pinna AD. Systematic review of surgical resection vs radiofrequency ablation for hepatocellular carcinoma. World J Gastroenterol. 2013;19:4106–18.
Cucchetti A, Piscaglia F, Cescon M, et al. An explorative data-analysis to support the choice between hepatic resection and radiofrequency ablation in the treatment of hepatocellular carcinoma. Dig Liver Dis. 2014;46:257–63.
Takayasu K, Arii S, Ikai I, et al. Overall survival after transarterial lipiodol infusion chemotherapy with or without embolization for unresectable hepatocellular carcinoma: propensity score analysis. AJR Am J Roentgenol. 2010;194:830–7.
Collettini F, Schnapauff D, Poellinger A, et al. Hepatocellular carcinoma: computed-tomography-guided high-dose-rate brachytherapy (CT-HDRBT) ablation of large (5–7 cm) and very large (> 7 cm) tumours. Eur Radiol. 2012;22:1101–9.
Mohnike K, Wieners G, Schwartz F, et al. Computed tomography-guided high-dose-rate brachytherapy in hepatocellular carcinoma: safety, efficacy, and effect on survival. Int J Radiat Oncol Biol Phys. 2010;78:172–9.
Ricke J, Mohnike K, Pech M, et al. Local response and impact on survival after local ablation of liver metastases from colorectal carcinoma by computed tomography-guided high-dose-rate brachytherapy. Int J Radiat Oncol Biol Phys. 2010;78:479–85.
Ricke J, Thormann M, Ludewig M, et al. MR-guided liver tumor ablation employing open high-field 1.0T MRI for image-guided brachytherapy. Eur Radiol. 2010;20:1985–93.
Ricke J, Wust P, Stohlmann A, et al. CT-guided interstitial brachytherapy of liver malignancies alone or in combination with thermal ablation: phase I–II results of a novel technique. Int J Radiat Oncol Biol Phys. 2004;58:1496–505.
Ricke J, Wust P, Wieners G, et al. Liver malignancies: CT-guided interstitial brachytherapy in patients with unfavorable lesions for thermal ablation. J Vasc Interv Radiol. 2004;15:1279–86.
Tselis N, Chatzikonstantinou G, Kolotas C, Milickovic N, Baltas D, Zamboglou N. Computed tomography-guided interstitial high dose rate brachytherapy for centrally located liver tumours: a single institution study. Eur Radiol. 2013;23:2264–70.
Mohnike K, Neumann K, Hass P, et al. Radioablation of adrenal gland malignomas with interstitial high-dose-rate brachytherapy: efficacy and outcome. Strahlenther Onkol. 2017;193:612–9.
Mohnike K, Wolf S, Damm R, et al. Radioablation of liver malignancies with interstitial high-dose-rate brachytherapy: complications and risk factors. Strahlenther Onkol. 2016;192:288–96.
Hata M, Tokuuye K, Sugahara S, et al. Proton beam therapy for hepatocellular carcinoma with portal vein tumor thrombus. Cancer. 2005;104:794–801.
Lee SU, Park JW, Kim TH, et al. Effectiveness and safety of proton beam therapy for advanced hepatocellular carcinoma with portal vein tumor thrombosis. Strahlenther Onkol. 2014;190:806–14.
Sugahara S, Nakayama H, Fukuda K, et al. Proton-beam therapy for hepatocellular carcinoma associated with portal vein tumor thrombosis. Strahlenther Onkol. 2009;185:782–8.
Mohnike K, Sauerland H, Seidensticker M, et al. Haemorrhagic complications and symptomatic venous thromboembolism in interventional tumour ablations: the impact of peri-interventional thrombosis prophylaxis. Cardiovasc Intervent Radiol. 2016;39:1716–21.
Cannistra SA. Phase II trials in journal of clinical oncology. J Clin Oncol. 2009;27:3073–6.
Bruix J, Sherman M, Llovet JM, et al. Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the study of the liver. J Hepatol. 2001;35:421–30.
Bruix J, Sherman M. Practice Guidelines Committee AAftSoLD. Management of hepatocellular carcinoma. Hepatology. 2005;42:1208–36.
Llovet JM, Real MI, Montana X, et al. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet. 2002;359:1734–9.
Lo CM, Ngan H, Tso WK, et al. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology. 2002;35:1164–71.
Lencioni R. Chemoembolization in patients with hepatocellular carcinoma. Liver Cancer. 2012;1:41–50.
Lammer J, Malagari K, Vogl T, et al. Prospective randomized study of doxorubicin-eluting-bead embolization in the treatment of hepatocellular carcinoma: results of the PRECISION V study. Cardiovasc Intervent Radiol. 2010;33:41–52.
Forner A, Gilabert M, Bruix J, Raoul JL. Treatment of intermediate-stage hepatocellular carcinoma. Nat Rev Clin Oncol. 2014;11:525–35.
Funding
This study was funded exclusively by the University of Magdeburg.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
Consent for publication was obtained for every individual person’s data included in the study.
Financial Support
This work was funded exclusively by the University of Magdeburg.
Ethical Considerations
The study was conducted in accordance with the protocol, the ethical principles that have their origin in the Declaration of Helsinki, and ICH-GCP. The study protocol and all study-related documentation were approved by all relevant authorities (Ethics Committee of the Medical Faculty, University of Magdeburg, 44/06).
Rights and permissions
About this article
Cite this article
Mohnike, K., Steffen, I.G., Seidensticker, M. et al. Radioablation by Image-Guided (HDR) Brachytherapy and Transarterial Chemoembolization in Hepatocellular Carcinoma: A Randomized Phase II Trial. Cardiovasc Intervent Radiol 42, 239–249 (2019). https://doi.org/10.1007/s00270-018-2127-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-018-2127-5