Abstract
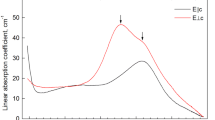
Polarized absorption spectra, σ and π, in the spectral range 30000–400 cm−1 (3.71–0.05 eV) were obtained on crystal slabs // [001] of deep blue rutile at various temperatures from 88 to 773 K. The rutile crystals were grown in Pt-capsules from carefully dried 99.999% TiO2 rutile powder at 50 kbar/1500 °C using graphite heating cells in a belt-type apparatus. Impurities were below the detection limits of the electron microprobe (about 0.005 wt% for elements with Z≥13). The spectra are characterized by an unpolarized absorption edge at 24300 cm−1, two weak and relatively narrow (Δν1/2≈3500–4000 cm−1), slightly σ-polarized bands ν1 at 23500 cm−1 and ν2 at 18500 cm−1, and a complex, strong band system in the NIR (near infra red) with sharp weak peaks in the region of the OH stretching fundamentals superimposed on the NIR system in the σ-spectra. The NIR band system and the UV edge produce an absorption minimum in both spectra, σ and π, at 21000 cm−1, i.e. in the blue, which explains the colour of the crystals. Bands ν1 and ν2 are assigned to dd transitions to the Jahn-Teller split upper Eg state of octahedral Ti3+. The NIR band system can be fitted as a sum of three components. Two of them are partly π-polarized, nearly Gaussian bands, both with large half widths 6000–7000 cm−1, ν3 at 12000 cm–1 and the most intense ν4 at 6500 cm−1. The third NIR band ν5 of a mixed Lorentz-Gaussian shape with a maximum at 3000 cm−1 forms a shoulder on the low-energy wing of ν4. Energy positions, half band widths and temperature behaviour of these bands are consistent with a small polaron type of Ti3+Ti4+ charge transfer (CT). Polarization dependence of CT bands can be explained on the basis of the structural model of defect rutile by Bursill and Blanchin (1983) involving interstitial titanium. Two OH bands at 3322 and 3279 cm−1 in σ-spectra show different stability during annealing, indicating two different positions of proton in the rutile structure, one of them probably connected with Ti3+ impurity. Total water concentration in blue rutile determined by IR spectroscopy is 0.10 wt-% OH. The EPR spectra measured in the temperature interval 20–295 K show the presence of an electron centre at temperatures above 100 K and Ti3+ ions in more than one structural position, but predominantly in compressed interstitial octahedral sites, at lower temperatures. These results are in good agreement with the conclusions based on the electronic absorption data.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 24 March 1997 / Revised, accepted: 14 October 1997
Rights and permissions
About this article
Cite this article
Khomenko, V., Langer, K., Rager, H. et al. Electronic absorption by Ti3+ ions and electron delocalization in synthetic blue rutile. Phys Chem Min 25, 338–346 (1998). https://doi.org/10.1007/s002690050124
Issue Date:
DOI: https://doi.org/10.1007/s002690050124