Abstract
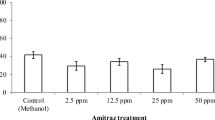
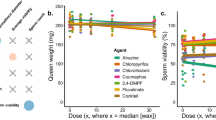
Pollinator diversity and abundance in North America have been at a steep decline over the last two decades due to the combinatorial effects of several environmental and anthropogenic stressors. In particular, managed honey bees (Apis mellifera) face multiple health risks including nutritional stress, exposure to pests and pathogens, poor queen quality, and pesticide contamination, which cause problems at the individual and colony levels. One of the gravest problems faced by honey bees is parasitization by the mite Varroa destructor, which is typically controlled through the application of miticides such as tau-fluvalinate, coumaphos, and amitraz. In addition to miticides, colonies are also exposed to pesticides brought by foragers from agricultural settings, including the fungicide chlorothalonil and the insecticide chlorpyrifos. Here, we explored whether exposure of wax to combinations of these pesticides during development affects honey bee queen physiology and worker behavior. To do this, we reared queens in plastic cups coated with molten beeswax that was either pesticide-free or containing field-relevant concentrations of tau-fluvalinate and coumaphos, amitraz, or chlorothalonil and chlorpyrifos. Once queens mated naturally, we placed them in observation hives to measure egg-laying rate and worker retinue size. We then dissected the queens and used the contents of their mandibular glands to measure worker attractiveness in caged bioassays and to analyze their chemical components using GC-MS. Exposure of wax to field-relevant concentrations of the tested pesticides during queen development significantly lowered the adult queens’ egg-laying rate and worker retinue size. Miticide exposure during development also lowered the attractiveness of queen mandibular gland contents to workers and affected the relative amounts of the glands’ chemical components. Our results support the ideas that mandibular gland pheromones act as honest indicators of queen reproductive fitness and that pesticide exposure of wax during bee development is an important and concerning factor impairing honey bee health.
Significance statement
Pollinator health is currently being threatened by multiple environmental factors, including poor nutrition, habitat fragmentation, and pesticide contamination. In particular, honey bee health is facing challenges caused by combinations of these factors, leading to an annual mortality of over 30% of all managed colonies in the USA. The conditions in which honey bee queens are reared can cause severe physiological and behavioral effects in adults, which in turn can affect the behavior of workers that tend to those queens, thus affecting colony productivity, growth, and survival. In this study, we show that queens reared in beeswax contaminated with miticides or agrochemicals lay fewer eggs over time and exhibit mandibular gland pheromone profiles that differ from those of queens reared in pesticide-free wax. Workers discern these differences, preferentially investigating the gland contents and forming larger retinues (thus performing more tending behaviors) for queens reared in pesticide-free wax. These results indicate that pesticide contamination of the queen-rearing wax directly affects queen physiology and behavior and further affirm that queen mandibular pheromones seem to act as honest indicators to workers of a queen’s reproductive capacity and overall health.



Similar content being viewed by others
References
Ahn K, Xie X, Riddle J, Pettis J, Huang ZY (2012) Effects of long distance transportation on honey bee physiology. Psyche. https://doi.org/10.1155/2012/193029
Amsalem E, Grozinger CM (2018) The importance of holistically evaluating data: a comment on Holman. Behav Ecol 29(6):1210–1211. https://doi.org/10.1093/beheco/ary082
Arena M, Sgolastra F (2014) A meta-analysis comparing the sensitivity of bees to pesticides. Ecotoxicology 23:324–334. https://doi.org/10.1007/s10646-014-1190-1
Belsky J, Joshi NK (2019) Impact of biotic and abiotic stressors on managed and feral bees. Insects 10:233. https://doi.org/10.3390/insects10080233
Boecking O, Genersch E (2008) Varroosis – the ongoing crisis in beekeeping. J Verbr Lebensm 3:221–228. https://doi.org/10.1007/s00003-008-0331-y
Büchler R (1994) Varroa tolerance in honey bees—occurrence, characters and breeding. Bee World 75:54–70. https://doi.org/10.1080/0005772X.1994.11099201
Burley LM, Fell R, Saacke R (2008) Survival of honey bee (Hymenoptera: Apidae) spermatozoa incubated at room temperature from drones exposed to miticides. J Econ Entomol 101:1081–1087. https://doi.org/10.1603/0022-493(2008)101[1081:SOHBHA]2.0.CO;2
Butler CG (1961) The scent of queen honeybees (Apis mellifera L.) that causes partial inhibition of queen rearing. J Insect Physiol 7:258–264. https://doi.org/10.1016/0022-1910(61)90076-2
Calderone NW (2012) Insect pollinated crops, insect pollinators and US agriculture: trend analysis of aggregate data for the period 1992–2009. PLoS One 7(5):e37235. https://doi.org/10.1371/journal.pone.0037235
Calis JNM, Boot WJ, Beetsma J, van den Eijnde JHPM, de Ruijter A, van der Steen JJM (1999) Effective biotechnical control of varroa: applying knowledge on brood cell invasion to trap honey bee parasites in drone brood. J Apic Res 38:49–61. https://doi.org/10.1080/00218839.1999.11100995
Chaimanee V, Evans JD, Chen Y, Jackson C, Pettis JS (2016) Sperm viability and gene expression in honey bee queens (Apis mellifera) following exposure to the neonicotinoid insecticide imidacloprid and the organophosphate acaricide coumaphos. J Insect Physiol 89. https://doi.org/10.1016/j.jinsphys.2016.03.004
Collins AM, Pettis JS (2013) Correlation of queen size and spermathecal contents and effects of miticides exposure during development. Apidologie 44:351–356. https://doi.org/10.1007/s13592-012-0186-1
Collins AM, Pettis JS, Wilbanks R, Feldlaufer MF (2004) Performance of honey bee (Apis mellifera) queens reared in beeswax cells impregnated with coumaphos. J Apic Res 43:128–134. https://doi.org/10.1371/journal.pone.0076536
Dai P, Jack CJ, Mortensen AN, Ellis JD (2017) Acute toxicity of five pesticides to Apis mellifera larvae reared in vitro. Pest Manag Sci 73:2282–2286. https://doi.org/10.1002/ps.4608
Doublet V, Labarussias M, de Miranda JR, Moritz RFA, Paxton RJ (2014) Bees under stress: sublethal doses of a neonicotinoid pesticide and pathogens interact to elevate honey bee mortality across the life cycle. Environ Microbiol 17:969–983. https://doi.org/10.1111/1462-2920.12426
Dussaubat C, Maisonnasse A, Crauser D, Tchamitchian S, Bonnet M, Cousin M, Kretzschmar A, Brunet J-L, Le Conte Y (2016) Combined neonicotinoid pesticide and parasite stress alter honeybee queens’ physiology and survival. Sci Rep 6:31430. https://doi.org/10.1038/srep31430
Elzen PJ, Westervelt D (2002) Detection of coumaphos resistance in Varroa destructorin Florida. Am Bee J 142:291–292
Elzen PJ, Eischen FA, Baxter JB, Pettis J, Elzen GW, Wilson WT (1998) Tau-fluvalinate resistance in Varroa jacoboni from several geographic locations. Am Bee J 138:674–676
Elzen PJ, Baxter JB, Spivak M, Wilson WT (2000) Control of Varroa jacobsoni Oud. Resistant to fluvalinate and amitraz using coumaphos. Apidologi 31:437–441. https://doi.org/10.1051/apido:2000134
Fine JD, Shpigler HY, Ray AM, Beach NJ, Sankey AL, Cash-Ahmed A, Huang ZY, Astrauskaite I, Chao R, Zhao H, Robinson GE (2018) Quantifying the effects of pollen nutrition on honey bee queen egg laying with a new laboratory system. PLoS One 13(9):e0203444. https://doi.org/10.1371/journal.pone.0203444
Fisher A II, Rangel J (2018) Exposure to pesticides during development negatively affects honey bee (Apis mellifera) drone sperm viability. PLoS One 13(12):e0208630. https://doi.org/10.1371/journal.pone.0208630
Gallai N, Salles J-M, Settele J, Vaissière BE (2009) Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol Econ 68(3):810–821. https://doi.org/10.1016/j.ecolecon.2008.06.014
Gradish AE, van der Steen J, Scott-Dupree CD, Cabrera AR, Cutler GC, Goulson D, Klein O, Lehmann DM, Lückmann J, O'Neill B, Raine NE, Sharma B, Thompson H (2019) Comparison of pesticide exposure in honey bees (Hymenoptera: Apidae) and bumble bees (Hymenoptera: Apidae): implications for risk assessments. Environ Entomol 48(1):12–21. https://doi.org/10.1093/ee/nvy168
Gregorc A, Alburaki M, Werle C, Knight PR, Adamczyk J (2017) Brood removal or queen caging combined ith oxalic acid treatment to control varroa mites (Varroa destructor) in honey bee colonies (Apis mellifera). Apidologie 48:821–832. https://doi.org/10.1007/s13592-017-0526-2
Gregorc A, Alburaki M, Rinderer N, Sampson B, Knight PR, Karim S, Adamczyk J (2018) Effects of coumaphos and imidacloprid on honey bee (Hymenoptera: Apidae) lifespan and antioxidant gene regulations in laboratory experiments. Sci Rep 8:1–13. https://doi.org/10.1038/s41598-018-33348-4
Grozinger CM, Sharabash NM, Whitfield CW, Robinson GE (2003) Pheromone-mediated gene expression in the honey bee brain. Proc Natl Acad Sci U S A 100:14519–14525. https://doi.org/10.1073/pnas.2335884100
Guzmán-Novoa E, Eccles L, Calvete Y, Mcgowan J, Kelly PG, Correa-Benitez A (2010) Varroa destructor is the main culprit for the death and reduced populations of overwintered honey bee (Apis mellifera) colonies in Ontario, Canada. Apidologie 41(4). https://doi.org/10.1051/apido/2009076
Haarmann T, Spivak M, Weaver D, Weaver B, Glenn T (2002) Effects of fluvalinate and coumaphos on queen honey bees (Hymenoptera: Apidae) in two commercial queen rearing operations. J Econ Entomol 95:28–35. https://doi.org/10.1371/journal.pone.0132014
Haber AI, Steinhauer NA, van Engelsdorp D (2019) Use of chemical and nonchemical methods for the control of Varroa destructor (Acari: Varroidae) and associated winter colony losses in U.S. beekeeping operations. J Econ Entomol:1–17. https://doi.org/10.1093/jee/toz088
Holman L (2018) Queen pheromones and reproductive division of labor: a meta-analysis. Behav Ecol 29(6):1199–1209. https://doi.org/10.1093/beheco/ary023
Hoover SER, Keeling CI, Winston ML, Slessor KN (2003) The effect of queen pheromones on worker honey bee ovary development. Naturwissenschaften 90:477–480. https://doi.org/10.1007/s00114-003-0462-z
Hunt G, Given JK, Tsuruda JM, Andino GK (2016) Breeding mite-biting bees to control Varroa. Bee Cult 8:41–47
Johnson RM, Ellis MD, Mullin CA, Frazier M (2010) Pesticides and honey bee toxicity-USA. Apidologie 41(3):312–331. https://doi.org/10.1051/apido/2010018
Johnson RM, Dahlgren L, Siegfried BD, Ellis MD (2013) Effect of in-hive miticides on drone honey bee survival and sperm viability. J Apic Res 52:88–95. https://doi.org/10.3896/IBRA.1.52.2.18
Keeling CI, Slessor KN, Higo HA, Winston ML (2003) New components of the honey bee (Apis mellifera L.) queen retinue pheromone. Proc Natl Acad Sci U S A 100:4486–4491. https://doi.org/10.1186/1742-9994-7-18
Keller L, Nonacs P (1993) The role of queen pheromones in social insects: queen control or queen signal? Anim Behav 45:787–794. https://doi.org/10.1006/anbe.1993.1092
Kocher S, Grozinger C (2011) Cooperation, conflict, and the evolution of queen pheromones. J Chem Ecol 37:1263–1275. https://doi.org/10.1007/s10886-011-0036-z
Kocher SD, Richard F-J, Tarpy DR, Grozinger CM (2009) Queen reproductive state modulates pheromone production and queen-worker interactions in honeybees. Behav Ecol 20(5):1007–1014. https://doi.org/10.1093/beheco/arp090
Kopit AM, Pitts-Singer TL (2018) Routes of pesticide exposure in solitary cavity-nesting bees. Environ Entomol 47:499–510. https://doi.org/10.1093/ee/nvy034
Kralj J, Fuchs S (2006) Parasitic Varroa destructor mites influence flight duration and homing ability of infested Apis mellifera foragers. Apidologie 37:577–587. https://doi.org/10.1051/apido:2006040
Kulhanek K, Steinhauer N, Rennich K, Caron DM, Sagili RR, Pettis JS, Ellis J, Wilson ME, Wilkes JT, Tarpy DR, Rose R, Lee K, Rangel J, vanEngelsdorp D (2017) A national survey of managed honey bee 2015-2016 annual colony losses in the USA. J Apic Res 56:328–340. https://doi.org/10.1080/00218839.2017.1344496
Laidlaw HH, Eckert JE (1964) Queen rearing. University of California Press, CA
Le Conte Y, Hefetz A (2008) Primer pheromones in social hymenoptera. Annu Rev Entomol 53:523–542. https://doi.org/10.1146/annurev.ento.52.110405.091434
Leach ME, Drummond F (2018) A review of native wild bee nutritional health. Int J Ecol. https://doi.org/10.1155/2018/9607246
Lee KV, Steinhauer N, Rennich K, Wilson ME, Tarpy DR, Caron DM, Rose R, Delaplane KS, Baylis K, Lengerich EJ, Pettis J, Skinner JA, Wilkes JT, Sagili R, vanEnglesdorp D (2015) A national survey of managed honey bee 2013-2014 annual colony losses in the USA. Apidologie 46:292–305. https://doi.org/10.1007/s13592-015-0356-z
Lodesani M, Colombo M, Spreafico M (1995) Ineffectiveness of Apistan treatment against the mite Varroa jacobsoni Oud in several districts of Lombardy (Italy). Apidologie 26:67–72. https://doi.org/10.1051/apido:19950109
Malka O, Niño EL, Grozinger CM, Hefetz A (2014) Genomic analysis of the interactions between social environment and social communication systems in honey bees (Apis mellifera). Insect Biochem Mol 47:36–45. https://doi.org/10.1016/j.ibmb.2014.01.001
Matsuura K, Himuro C, Yokoi T, Yamamoto Y, Vargo EL, Keller L (2010) Identification of a pheromone regulating caste differentiation in termites. Proc Natl Acad Sci 107(29):12963–12968. https://doi.org/10.1073/pnas.1004675107
Melathopoulos A, Winston M, Pettis J, Pankiw T (1996) Effect of queen mandibular pheromone on initiation and maintenance of queen cells in the honey bee (Apis mellifera L.). Can Entomol 128:263–272. https://doi.org/10.4039/Ent128263-2
Michener CD (1974) The social behavior of bees: a comparative study. Harvard University Press, MA
Monnin T, Ratnieks FLW, Jones GR, Beard R (2002) Pretender punishment induced by chemical signaling in a queenless ant. Nature 419:61–65. https://doi.org/10.1038/nature00932
Morales MM, Ramos MJG, Vázquez PP, Galiano FJD, Valverde MG, Lopez VG, Flores JM, Fernández-Alba AR (2019) Distribution of chemical residues in the beehive compartments and their transfer to the honeybee brood. Sci Total Environ 710(25):136288. https://doi.org/10.1016/j.scitotenv.2019.136288
Moritz RFA, Simon UE, Crewe RM (2000) Pheromonal contest between honeybee workers. Naturwissenschaften 87:395–397. https://doi.org/10.1007/s001140050748
Moritz RFA, Lattorff HMG, Crewe RM (2004) Honeybee workers (Apis mellifera capensis) compete for producing queen-like pheromone signals. Biol Lett Proc R Soc Lond (Suppl) 271:98–100. https://doi.org/10.1098/rsbl.2003.0113
Mullin CA, Frazier M, Frazier JL, Ashcraft S, Simonds R, vanEngelsdorp D, Pettis JS (2010) High levels of miticides and agrochemicals in north American apiaries: implications for honey bee health. PLoS One 5:e9754. https://doi.org/10.1371/journal.pone.0009754
Mumoki FN, Pirk CWW, Yusuf AA, Crewe RM (2018) Reproductive parasitism by worker honey bees suppressed by queens through regulation of worker mandibular secretions. Sci Rep 8:7701. https://doi.org/10.1038/s41598-018-26060-w
Niño EL, Malka O, Hefetz A, Teal P, Hayes J, Grozinger CM (2012) Effects of honey bee (Apis mellifera L.) queen insemination volume on worker behavior and physiology. J Insect Physiol 58(8):1082–1089. https://doi.org/10.1016/j.jinsphys.2012.04.015
Niño EL, Malka O, Hefetz A, Tarpy DR, Grozinger CM (2013) Chemical profiles of two pheromone glands are differentially regulated by distinct mating factors in honey bee queens (Apis mellifera L.). PLoS One 8(11):e78637. https://doi.org/10.1371/journal.pone.0078637
O'Neal ST, Brewster CC, Bloomquist JR, Anderson TD (2017) Amitraz and its metabolite modulate honey bee cardiac function and tolerance to viral infection. J Invertebr Pathol 149:119–126. https://doi.org/10.1016/j.jip.2017.08.005
Ostiguy N, Drummond FA, Aronstein K, Eitzer B, Ellis JD, Spivak M, Sheppard WS (2019) Honey bee exposure to pesticides: a four-year nationwide study. Insects. https://doi.org/10.3390/insects10010013
Pankiw T, Winston ML, Slessor KN (1995) Queen attendance behavior of worker honey bees (Apis mellifera L) that are high and low responding to queen mandibular pheromone. Ins Soc 42:371–378. https://doi.org/10.1007/BF01242165
Peso M, Elgar MA, Barron AB (2014) Pheromonal control: reconciling physiological mechanism with signaling theory. Biol Rev 90:542–559. https://doi.org/10.1111/brv.12123
Pettis JS, Wilson WT, Shimanuki H, Teel PD (1991) Fluvalinate treatment of queen and worker honey bees (Apis mellifera L) and effects on subsequent mortality, queen acceptance and supersedure. Apidologie 22:1–7. https://doi.org/10.1051/apido:19910101
Pettis JS, Collins AM, Wilbanks R, Feldaufer MF (2004) Effects of coumaphos on queen rearing in the honey bee, Apis mellifera. Apidologie 35:605–610. https://doi.org/10.1051/apido:2004056
Plettner E, Slessor KN, Winston ML, Oliver JE (1996) Caste-selective pheromone biosynthesis in honey bees. Science 271:1851–1853. https://doi.org/10.1126/science.271.5257.1851
Potts SG, Biesmeijer JC, Kremen C, Neumann P, Schweiger O, Kunin WE (2010) Global pollinator declines: trends, impacts and drivers. Trends Ecol Evol 25(6):345–353. https://doi.org/10.1016/j.tree.2010.01.007
Princen SA, Oliveira RC, Ernst UR, Millar JG, van Zweden JS, Wenseleers T (2019) Honeybees possess a structurally diverse and functionally redundant set of queen pheromones. Proc R Soc B 286. https://doi.org/10.1098/rspb.2019.0517
Rangel J, Tarpy DR (2015) The combined effects of miticides on the mating health of honey bee (Apis mellifera L.) queens. J Apic Res 54(3):275–283. https://doi.org/10.1080/00218839.2016.1147218
Rangel J, Keller JJ, Tarpy DR (2013) The effects of honey bee (Apis mellifera L.) queen reproductive potential on colony growth. Insect Soc 60:65–73. https://doi.org/10.1007/s00040-012-0267-1
Rangel J, Böröczky K, Schal C, Tarpy DR (2016) Honey bee (Apis mellifera) queen reproductive potential affects queen mandibular gland pheromone composition and worker retinue response. PLoS One 11(6):e0156027. https://doi.org/10.1371/journal.pone.0156027
Ratnieks FL (1993) Egg-laying, egg-removal, and ovary development by workers in queenright honey bee colonies. Behav Ecol Sociobiol 32:191. https://doi.org/10.1007/BF00173777
Reeves AM, O’Neal ST, Fell RD, Brewster CC, Anderson TD (2018) In-hive acaricides alter biochemical and morphological indicators of honey bee nutrition, immunity, and development. J Insect Sci 18(5). https://doi.org/10.1093/jisesa/iey086
Richard F-J, Tarpy DR, Grozinger CM (2007) Effects of insemination quantity on honey bee queen physiology. PLoS One 2(10):e980. https://doi.org/10.1371/journal.pone.0000980
Rinderer TE, De Guzman LI, Lancaster VA, Delatte GT, Stelzer JA (1999) Varroa in the mating yard: I. the effects of Varroa jacobsoni and Apistan on drone honey bees. Am Bee J 139:134–139
Rinderer TE, Harris JW, Hunt GJ, de Guzman LI (2010) Breeding for resistance to Varroa destructor in North America. Apidologie 41:409–424. https://doi.org/10.1051/apido/2010015
Rosenkranz P, Aumeier P, Ziegelmann B (2010) Biology and control of Varroa destructor. J Invertebr Pathol 103(Suppl 1):S96–S119. https://doi.org/10.1016/j.jip.2009.07.016
Schäfer MO, Dietemann V, Pirk CWW, Neumann P, Crewe RM, Hepburn HR et al (2006) Individual versus social pathway to honeybee worker reproduction (Apis mellifera): pollen or jelly as protein source for oogenesis? J Comp Physiol A 192:761–768. https://doi.org/10.1007/s00359-006-0112-y
Seeley T (1996) The wisdom of the hive. Harvard University Press, Cambridge, MA
Slessor KN, Kaminski L-A, King GGS, Winston ML (1990) Semiochemicals of the honeybee queen mandibular glands. J Chem Ecol 16:851–860. https://doi.org/10.1007/BF01016495
Slessor KN, Winston ML, Le Conte Y (2005) Pheromone communication in the honeybee (Apis mellifera L.). J Chem Ecol 31:2731–2745. https://doi.org/10.1007/s10886-005-7623-9
Smith AA, Hölldobler B, Liebig J (2012) Queen-specific signals and worker punishment in the ant Aphaenogaster cockerelli: the role of the Dufour’s gland. Anim Behav 83(3):587–593. https://doi.org/10.1016/j.anbehav.2011.12.024
Spivak M (1996) Honey bee hygienic behavior and defense against Varroa jacobsoni. Apidologie 27(4):245–260. https://doi.org/10.1051/apido:19960407
Spivak M, Mader E, Vaughan M, Euliss NH (2010) The plight of the bees. Eviron Sci Technol 45:34–38. https://doi.org/10.1021/es101468w
Steinhauer N, Kulhanek K, Antúnez K, Human H, Chantawannakul P, Chauzat M-P, vanEngelsdorp D (2018) Drivers of colony losses. Curr Opin Insect Sci 26:142–148. https://doi.org/10.1016/j.cois.2018.02.004
Sylvester HA, Watts RP, De Guzman LI, Stelzer JA, Rinderer TE (1999) Varroa in the mating yard: II. The effects of Varroa and fluvalinate on drone mating competitiveness. Am Bee J 139:225–227
Tomé HVV, Schmehl DR, Wedde AE, Godoy RSM, Ravaiano SV, Guedes RNC, Martins GF, Ellis JD (2020) Frequently encountered pesticides can cause multiple disorders in developing worker honey bees. Environ Pollut 256:113420. https://doi.org/10.1016/j.envpol.2019.113420
Traynor KS, Pettis JS, Tarpy DR, Mullin CA, Frazier JL, Frazier M, vanEngelsdorp D (2016) In-hive pesticide exposome: assessing risks to migratory honey bees from in-hive pesticide contamination in the eastern United States. Sci Rep 6:33207. https://doi.org/10.1038/srep33207
United States Geological Survey (2017) Pesticide national synthesis project. U.S. Department of the Interior https://www.usgs.gov/mission-areas/water-resources/science/pesticides?qt-science_center_objects=0#qt-science_center_objects. Accessed 10 January 2020
Urlacher E, Tarr IS, Mercer AR (2014) Social modulation of stress reactivity and learning in young worker honey bees. PLoS One 9(12):e113630. https://doi.org/10.1371/journal.pone.0113630
Urlacher E, Monchanin C, Rivière C, Richard FJ, Lombardi C, Michelsen-Heath S, Hageman KJ, Mercer AR (2016) Measurements of chlorpyrifos levels in forager bees and comparison with levels that disrupt honey bee odor-mediated learning under laboratory conditions. J Chem Ecol 42(2):127–138. https://doi.org/10.1007/s10886-016-0672-4
Van Oystaeyen A, Oliveira RC, Holman L, van Zweden JS (2014) Conserved class of queen pheromones stops social insect workers from reproducing. Science 343:287–290. https://doi.org/10.1126/science.1244899
Vilcinskas A (2019) Pathogens associated with invasive or introduced insects threaten the health and diversity of native species. Curr Opin Insect Sci 33:43–48. https://doi.org/10.1016/j.cois.2019.03.004
Wallner K (1999) Varroacides and their residues in bee products. Apidologie 30:235–248
Walsh EM, Rangel J (2018) Queen pheromones and mandibular gland dissection. Bee World 95(1):3–5. https://doi.org/10.1080/0005772X.2017.1373511
Williams G, Troxler A, Retschnig G, Roth K, Yanez O, Shutler D, Neumann P, Gauthier L (2015) Neonicotinoid pesticides severely affect honey bee queens. Sci Rep 5:14621. https://doi.org/10.1038/srep14621
Wilson EO (1971) The insect societies. Harvard University Press, Cambridge, MA
Winston ML (1987) The biology of the honey bee. Harvard University Press, Cambridge, Mass
Winston ML, Slessor KN (1998) Honey bee primer pheromones and colony organization: gaps in our knowledge. Apidologie 29:81–95. https://doi.org/10.1051/apido:19980105
Wood SC, Kozii IV, Medici de Mattos I, de Carvalho Macedo Silva R, Klein CD, Dvylyuk I, Moshynskyy I, Epp T, Simko E (2020) Chronic high-dose neonicotinoid exposure decreases overwinter survival of Apis mellifera L. Insects 11:30. https://doi.org/10.3390/insects11010030
Wu JY, Anelli CM, Sheppard WS (2011) Sub-lethal effects of pesticide residues in brood comb on worker honey bee (Apis mellifera) development and longevity. PLoS One 6(2):e14720. https://doi.org/10.1371/journal.pone.0014720
Yao J, Zhu YC, Adamczyk J (2018) Responses of honey bees to lethal and sublethal doses of formulated clothianidin alone and mixtures. J Econ Entomol 111(4):1517–1525. https://doi.org/10.1093/jee/toy140
Zhu W, Schmehl DR, Mullin CA, Frazier JL (2014) Four common pesticides, their mixtures, and a formulation solvent in the hive environment have high oral toxicity to honey bee larvae. PLoS One 9(1):e0077547. https://doi.org/10.1371/journal.pone.0077547
Acknowledgments
The authors thank the anonymous reviewers for their feedback and suggestions on this manuscript. This work was assisted by E.T. Ash, as well as both Joseph and Anne Walsh, for generously sharing their beekeeping assistance and resources throughout the project. Members of the Rangel Honey Bee Laboratory, both past and present, provided insight into this work during all stages of planning, implementation, and completion. The authors also thank the Agriculture Women Excited to Share Opinions, Mentoring and Experiences (AWESOME) faculty group of the College of Agriculture and Life Sciences at Texas A&M University for assistance with editing the manuscript.
Funding
This work was supported in part by the Herb Dean Scholarship to cover EMW’s tuition, funding to JR and NI by a USDA-NIFA award (2015- 67013-23170), a grant from the California State Beekeepers Association, and Texas A&M University’s Hatch Project (TEX09557).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by A. Toth
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Walsh, E.M., Sweet, S., Knap, A. et al. Queen honey bee (Apis mellifera) pheromone and reproductive behavior are affected by pesticide exposure during development. Behav Ecol Sociobiol 74, 33 (2020). https://doi.org/10.1007/s00265-020-2810-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-020-2810-9