Abstract
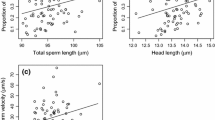
Some theoretical models of sperm competition make the assumption that in fish species with external fertilisation, sperm length relates positively to swimming speed at the expense of sperm longevity. Few studies have tested this assumption. We used the three-spined stickleback, Gasterosteus aculeatus L., to study functional sperm morphology. In this study, the relationship between males’ mean sperm length and fertilisation rate was investigated in vitro in a non-competitive situation. Fertilisation at different time points after sperm release was taken into account, and sperm morphology was quantified from scanning electron microscopy images. The time series of artificial fertilisations demonstrated that males which produced sperm with a longer tail fertilised faster, but their sperm had a shorter lifespan (or activity period). It was further suggested that males that produced sperm with a larger midpiece had greater fertilisation chances later on in the fertilisation process. Thus, in sticklebacks, there exists functional variation in sperm morphology, and sperm tail length is traded off against sperm longevity (or activity).


Similar content being viewed by others
References
Anderson MJ, Dixson AF (2002) Motility and the midpiece in primates. Nature 416:496
Anderson MJ, Dixson AS, Dixson AF (2006) Mammalian sperm and oviducts are sexually selected: evidence for co-evolution. J Zool 270:682–686
Bakker TCM, Zbinden M, Frommen JG, Weiss A, Largiadèr CR (2006) Slow fertilization of stickleback eggs: the result of sexual conflict? BMC Ecol 6:7
Ball MA, Parker GA (1996) Sperm competition games: external fertilization and “adaptive” infertility. J Theor Biol 180:141–150
Ball MA, Parker GA (1997) Sperm competition games: inter- and intraspecies results of a continuous external fertilization model. J Theor Biol 186:459–466
Balshine S, Leach BJ, Neat F, Werner NY, Montgomerie R (2001) Sperm size of African cichlids in relation to sperm competition. Behav Ecol 12:726–731
Barber I, Arnott SA (2000) Split-clutch IVF: a technique to examine indirect fitness consequences of mate preferences in sticklebacks. Behaviour 137:1129–1140
Barber I, Arnott SA, Braithwaite VA, Andrew J, Huntingford FA (2001) Indirect fitness consequences of mate choice in sticklebacks: offspring of brighter males grow slowly but resist parasitic infections. Proc R Soc Lond B 268:71–76
Becker WA (1992) Manual of quantitative genetics, 5th edn. Academic, Pullman
Bell MA, Foster SA (eds) (1994) The evolutionary biology of the threespine stickleback. Oxford University Press, Oxford
Birkhead TR, Hosken DJ, Pitnick S (eds) (2009) Sperm biology: an evolutionary perspective. Academic, Amsterdam
Briskie JV, Montgomerie R (1992) Sperm size and sperm competition in birds. Proc R Soc Lond B 247:89–95
Burness G, Casselman SJ, Schulte-Hostedde AI, Moyes CD, Montgomerie R (2004) Sperm swimming speed and energetics vary with sperm competition risk in bluegill (Lepomis macrochirus). Behav Ecol Sociobiol 56:65–70
Butts IAE, Johnson K, Wilson CC, Pitcher TE (2013) Ovarian fluid enhances sperm velocity based on relatedness in lake trout, Salvelinus namaycush. Theriogenology 78:2105–2109
Candolin U, Vlieger L (2013) Should attractive males sneak: the trade-off between current and future offspring. PLoS ONE 8:e57992
de Fraipont M, FitzGerald GJ, Guderley H (1993) Age-related differences in reproductive tactics in the three-spined stickleback, Gasterosteus aculeatus. Anim Behav 46:961–968
Dean MD, Nachman MW (2009) Faster fertilization rate in conspecific versus heterospecific matings in house mice. Evolution 63:20–28
Dziminski MA, Roberts JD, Beveridge M, Simmons LW (2009) Sperm competitiveness in frogs: slow and steady wins the race. Proc R Soc Lond B 276:3955–3961
Elgee KE, Evans JP, Ramnarine IW, Rush SA, Pitcher TE (2010) Geographic variation in sperm traits reflects predation risk and natural rates of multiple paternity in the guppy. J Evol Biol 23:1331–1338
Elofsson H, McAllister BG, Kime DE, Mayer I, Borg B (2003) Long lasting stickleback sperm; is ovarian fluid a key to success in fresh water? J Fish Biol 63:240–253
Evans JP (2011) Patterns of genetic variation and covariation in ejaculate traits reveal potential evolutionary constraints in guppies. Heredity 106:869–875
Falconer DS (1989) Introduction to quantitative genetics, 3rd edn. Longman, Harlow
Fauvel C, Savoye O, Dreanno C, Cosson J, Suquet M (1999) Characteristics of sperm of captive seabass in relation to its fertilization potential. J Fish Biol 54:356–369
Firman RC, Simmons LW (2009) Experimental evolution of sperm quality via postcopulatory sexual selection in house mice. Evolution 64:1245–1256
Firman RC, Simmons LW (2010) Sperm midpiece length predicts sperm swimming velocity in house mice. Biol Lett 6:513–516
Fitzpatrick JL, Desjardins JK, Milligan N, Montgomerie R, Balshine S (2007) Reproductive-tactic-specific variation in sperm swimming speeds in a shell-brooding cichlid. Biol Reprod 77:280–284
Fitzpatrick JL, Montgomerie R, Desjardins JK, Stiver KA, Kolm N, Balshine S (2009) Female promiscuity promotes the evolution of faster sperm in cichlid fishes. Proc Natl Acad Sci U S A 106:1128–1132
Fitzpatrick JL, Garcia-Gonzalez F, Evans JP (2010) Linking sperm length and velocity: the importance of intramale variation. Biol Lett 6:797–799
Fitzpatrick JL, Simmons LW, Evans JP (2012) Complex patterns of multivariate selection on the ejaculate of a broadcast spawning marine invertebrate. Evolution 66:2451–2460
Gage MJG, Macfarlane C, Yeates S, Shackleton R, Parker GA (2002) Relationships between sperm morphometry and sperm motility in the Atlantic salmon. J Fish Biol 61:1528–1539
Gasparini C, Pilastro A (2011) Cryptic female preference for genetically unrelated males is mediated by ovarian fluid in the guppy. Proc R Soc Lond B 278:2495–2501
Gomendio M, Roldan ERS (1991) Sperm competition influences sperm size in mammals. Proc R Soc Lond B 243:181–186
Helfenstein F, Podevin M, Richner H (2010) Sperm morphology, swimming velocity, and longevity in the house sparrow Passer domesticus. Behav Ecol Sociobiol 64:557–565
Higginson DM, Miller KB, Segraves KA, Pitnick S (2012) Female reproductive tract form drives the evolution of complex sperm morphology. Proc Natl Acad Sci U S A 109:4538–4543
Humphries S, Evans JP, Simmons LW (2008) Sperm competition: linking form to function. BMC Evol Biol 8:319
Jamieson BGM (1991) Fish evolution and systematics: evidence from spermatozoa. Cambridge University Press, Cambridge
Jamieson BGM (2009) Ultrastructure of spermatozoa: Acanthopterygii continued: Percomorpha. In: Jamieson BGM (ed) Reproductive biology and phylogeny of fishes (agnathans and bony fishes), part A. Science Publishers, Enfield, pp 503–684
Johnson DW, Monro K, Marshall DJ (2012) The maintenance of sperm variability: context-dependent selection on sperm morphology in a broadcast spawning invertebrate. Evolution 67:1383–1395
Karr TL, Swanson WJ, Snook RR (2009) The evolutionary significance of variation in sperm-egg interactions. In: Birkhead TR, Hosken DJ, Pitnick S (eds) Sperm biology: an evolutionary perspective. Academic, Amsterdam, pp 305–365
Katz DF, Drobins EZ (1990) Analysis and interpretation of the forces generated by spermatozoa. In: Bavister BD, Cummins J, Roldan ERS (eds) Fertilization in mammals. Serono Symposia, Norwell, pp 125–137
Kleven O, Fossøy F, Laskemoen T, Robertson RJ, Rudolfsen G, Lifjeld JT (2009) Comparative evidence for the evolution of sperm swimming speed by sperm competition and female sperm storage duration in passerine birds. Evolution 63:2466–2473
Kraak SBM, Bakker TCM, Mundwiler B (1999) Correlates of the duration of the egg collecting phase in the three-spined stickleback. J Fish Biol 54:1038–1049
Largiadèr CR, Fries V, Bakker TCM (2001) Genetic analysis of sneaking and egg-thievery in a natural population of the three-spined stickleback (Gasterosteus aculeatus L.). Heredity 86:459–468
Laskemoen T, Kleven O, Fossøy F, Robertson RJ, Rudolfsen G, Lifjeld JT (2010) Sperm quantity and quality effects on fertilization success in a highly promiscuous passerine, the tree swallow Tachycineta bicolour. Behav Ecol Sociobiol 64:1473–1483
Laskemoen T, Albrecht T, Bonisoli-Alquati A, Cepak J, de Lope F et al (2013) Variation in sperm morphometry and sperm competition among barn swallow (Hirundo rustica) populations. Behav Ecol Sociobiol 67:301–309
Lemke MJ, Chiva M, Coyle B, Kulak D, MacKay M, Kasinsky H (1999) Variability of sperm nuclear basic proteins in the three-spined stickleback and related species of Gasterosteoidei. Comp Biochem Physiol B 122:339–353
Levitan DR (2000) Sperm velocity and longevity trade off each other and influence fertilization in the sea urchin Lytechinus variegates. Proc R Soc Lond B 267:531–534
Lifjeld JT, Laskemoen T, Kleven O, Albrecht T, Robertson RJ (2010) Sperm length variation as a predictor of extrapair paternity in passerine birds. PLoS ONE 5:e13456
Lifjeld JT, Laskemoen T, Kleven O, Pedersen ATM, Lampe HM, Rudolfsen G, Schmoll T, Slagsvold T (2012) No evidence for pre-copulatory sexual selection on sperm length in a passerine bird. PLoS ONE 7:e32611
Lüpold S, Calhim S, Immler S, Birkhead TR (2009) Sperm morphology and sperm velocity in passerine birds. Proc R Soc Lond B 276:1175–1181
Malo AF, Gomendio M, Garde J, Lang-Lenton B, Soler AJ, Roldan ERS (2006) Sperm design and sperm function. Biol Lett 2:246–249
Mattei X (1991) Spermatozoon ultrastructure and its systematic implications in fishes. Can J Zool 69:3038–3055
Mehlis M, Frommen JG, Rahn AK, Bakker TCM (2012) Inbreeding in three-spined sticklebacks (Gasterosteus aculeatus L.): effects on testis and sperm traits. Biol J Linn Soc 107:510–520
Mehlis M, Hilke LK, Bakker TCM (2013) Attractive males have faster sperm in three-spined sticklebacks Gasterosteus aculeatus. Curr Zool 59:761–768
Montgomerie R, Fitzpatrick JL (2009) Testes, sperm, and sperm competition. In: Jamieson BGM (ed) Reproductive biology and phylogeny of fishes (agnathans and bony fishes), part B. Science Publishers, Enfield, pp 1–53
Moore PJ, Harris WE, Montrose VT, Levin D, Moore AJ (2004) Constraints on evolution and postcopulatory sexual selection: trade-offs among ejaculate characteristics. Evolution 58:1773–1780
Mortimer D (1994) Practical laboratory andrology. Oxford University Press, Oxford
Parker GA (1970) Sperm competition and its evolutionary consequences in the insects. Biol Rev Camb Philos Soc 45:525–567
Parker GA (1993) Sperm competition games: sperm size and sperm number under adult control. Proc R Soc Lond B 253:245–254
Parker GA (1998) Sperm competition and the evolution of ejaculates: towards a theory base. In: Birkhead TR, Møller AP (eds) Sperm competition and sexual selection. Academic, San Diego, pp 3–54
Pinheiro JC, Bates D, DebRoy S, Sarkar D (2009) nlme: linear and nonlinear mixed effects models. R Packag 3:1–93
Pitnick S, Hosken DJ, Birkhead TR (2009a) Sperm morphological diversity. In: Birkhead TR, Hosken DJ, Pitnick S (eds) Sperm biology: an evolutionary perspective. Academic, Amsterdam, pp 69–149
Pitnick S, Wolfner MF, Suarez SS (2009b) Ejaculate-female and sperm-female interactions. In: Birkhead TR, Hosken DJ, Pitnick S (eds) Sperm biology: an evolutionary perspective. Academic, Amsterdam, pp 247–304
Pizzari T, Parker GA (2009) Sperm competition and sperm phenotype. In: Birkhead TR, Hosken DJ, Pitnick S (eds) Sperm biology: an evolutionary perspective. Academic, Amsterdam, pp 207–245
Pizzari T, Worley K, Burke T, Froman DP (2008) Sperm competition dynamics: ejaculate fertilising efficiency changes differentially with time. BMC Evol Biol 8:332
Rice WD, Gaines SD (1994) Extending nondirectional heterogeneity tests to evaluate simply ordered alternative hypotheses. Proc Natl Acad Sci U S A 91:225–226
Rick IP, Mehlis M, Eßer E, Bakker TCM (2013) The influence of ambient UV light on sperm quality and sexual ornamentation in three-spined sticklebacks (Gasterosteus aculeatus). Oecologia (published online, doi:10.1007/s00442-013-2773-x)
Simpson JL, Humphries S, Evans JP, Simmons LW, Fitzpatrick JL (2013) Relationship between sperm length and speed differ among three internally and three externally fertilizing species. Evolution. doi:10.1111/evo.12199
Skinner AMJ, Watt PJ (2007) Phenotypic correlates of spermatozoon quality in the guppy, Poecilia reticulata. Behav Ecol 18:47–52
Smith CC, Ryan MJ (2010) Evolution of sperm quality but not quantity in the internally fertilized fish Xiphophorus nigrensis. J Evol Biol 23:1759–1771
Snook RR (2005) Sperm in competition: not playing by the numbers. Trends Ecol Evol 20:46–53
Stockley P, Gage MJG, Parker GA, Møller AP (1996) Female reproductive biology and the coevolution of ejaculate characteristics in fish. Proc R Soc Lond B 263:451–458
Stockley P, Gage MJG, Parker GA, Møller AP (1997) Sperm competition in fishes: the evolution of testis size and ejaculate characteristics. Am Nat 149:933–954
Swarup H (1958) Stages in the development of the stickleback Gasterosteus aculeatus (L.). J Embryol Exp Morphol 6:373–383
Tourmente M, Gomendio M, Roldan ERS (2011) Sperm competition and the evolution of sperm design in mammals. BMC Evol Biol 11:12
Tuset VM, Dietrich GJ, Wojtczak M, Słowinska M, de Monserrat J, Ciereszko A (2008) Relationships between morphology, motility and fertilization capacity in rainbow trout (Oncorhynchus mykiss) spermatozoa. J Appl Ichthyol 24:393–397
Urbach D, Folstad I, Rudolfsen G (2005) Effects of ovarian fluid on sperm velocity in Arctic charr (Salvelinus alpinus). Behav Ecol Sociobiol 57:438–444
Wootton RJ (1976) The biology of the sticklebacks. Academic, London
Wootton RJ (1984) A functional biology of sticklebacks. Croom Helm, London
Zbinden M, Largiadèr CR, Bakker TCM (2001) Sperm allocation in the three-spined stickleback. J Fish Biol 59:1287–1297
Acknowledgments
We thank the “Bakker Research Group” for discussions and two anonymous reviewers for constructive comments on the manuscript. Timo Thünken is acknowledged for pointing out a mistake in the calculation of sperm numbers in previous estimations. We are very grateful to the late Erika Müller-Schulte for competence both in practical assistance, making scanning electron microscope images and measurements of part of sperm morphology. We thank George Wintermans, Texel, the Netherlands and Peter Snelderwaart, University of Leiden, the Netherlands for catching and caring for the 2001 Texel sticklebacks, respectively. We thank Joachim G. Frommen for the 2005 Texel fish samples and the Euskirchen 2005 samples. We thank Jan Hottentot for catching the sticklebacks, which were used in the control experiment. We acknowledge the permission of Jürgen Wittler to catch sticklebacks at the Euskirchen field site and the Swiss National Science Foundation and the Deutsche Forschungsgemeinschaft (DFG) (BA 2885/4-1) for financial support.
Ethical standards
The experiments comply with the current laws of Germany in which they were performed.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. Lindström
Rights and permissions
About this article
Cite this article
Bakker, T.C.M., Hollmann, M., Mehlis, M. et al. Functional variation of sperm morphology in sticklebacks. Behav Ecol Sociobiol 68, 617–627 (2014). https://doi.org/10.1007/s00265-013-1676-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-013-1676-5