Abstract
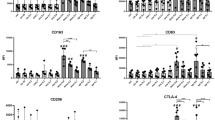
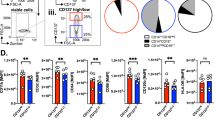
Macrophages have been shown to infiltrate a wide range of malignancies and are often considered to promote tumour survival, growth and spread. However, the source and behaviour of discrete tumour-associated macrophage populations are still poorly understood. Here we show a novel method for the rational development of bone marrow-derived monocytes appropriate for the study of processes which involve the contribution of circulating inflammatory monocytes. We have shown that in response to tumour-conditioned medium, these cells upregulate CD206 and CD115, markers traditionally associated with M2-type macrophages. Treated cells show reduced capacity for cytokine secretion but significantly impact CD4+ and CD8+ T-cell proliferation and polarization. Coculture with conditioned bone marrow-derived monocytes significantly reduced CD4+ T-cell proliferation but increased CD8+ T-cell proliferation and granzyme B expression with significant induction of IFNγ secretion by both CD4+ and CD8+ T cells, indicating that these cells may have a role in promoting anti-cancer immunity.






Similar content being viewed by others
Abbreviations
- B16CM:
-
B16F10-conditioned medium
- BMDM:
-
Bone marrow-derived monocytes
- iNOS:
-
Inducible nitric oxide synthase
- mRPMI:
-
Macrophage RPMI-1640
- NTCM:
-
No treatment concentrated medium
- RANTES:
-
Regulated on activation, normal T cell expressed and secreted
- RBC:
-
Red blood cell
- TAM:
-
Tumour-associated macrophage
References
Murray PJ, Allen JE, Biswas SK, Fisher EA, Gilroy DW, Goerdt S, Gordon S, Hamilton JA, Ivashkiv LB, Lawrence T, Locati M, Mantovani A, Martinez FO, Mege JL, Mosser DM, Natoli G, Saeij JP, Schultze JL, Shirey KA, Sica A, Suttles J, Udalova I, van Ginderachter JA, Vogel SN, Wynn TA (2014) Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41(1):14–20. https://doi.org/10.1016/j.immuni.2014.06.008
Mills CD (2012) M1 and M2 macrophages: oracles of health and disease. Crit Rev Immunol 32(6):463–488
Tremble LF, Forde PF (2017) Clinical evaluation of macrophages in cancer: role in treatment, modulation and challenges. Cancer Immunol Immunother 66(12):1509–1527. https://doi.org/10.1007/s00262-017-2065-0
Lee CM, Hu J (2013) Cell density during differentiation can alter the phenotype of bone marrow-derived macrophages. Cell Biosci 3:30. https://doi.org/10.1186/2045-3701-3-30
Rostam HM, Singh S, Salazar F, Magennis P, Hook A, Singh T, Vrana NE, Alexander MR, Ghaemmaghami AM (2016) The impact of surface chemistry modification on macrophage polarisation. Immunobiology 221(11):1237–1246. https://doi.org/10.1016/j.imbio.2016.06.010
Shima M, Teitelbaum SL, Holers VM, Ruzicka C, Osmack P, Ross FP (1995) Macrophage-colony-stimulating factor regulates expression of the integrins alpha 4 beta 1 and alpha 5 beta 1 by murine bone marrow macrophages. Proc Natl Acad Sci USA 92(11):5179–5183
Geissmann F, Jung S, Littman DR (2003) Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 19(1):71–82
Yang J, Zhang L, Yu C, Yang X-F, Wang H (2014) Monocyte and macrophage differentiation: circulation inflammatory monocyte as biomarker for inflammatory diseases. Biomark Res 2(1):1. https://doi.org/10.1186/2050-7771-2-1
Zheng J, Yang M, Shao J, Miao Y, Han J, Du J (2013) Chemokine receptor CX3CR9 contributes to macrophage survival in tumor metastasis. Mol Cancer 12(1):141. https://doi.org/10.1186/1476-4598-12-141
Richards DM, Hettinger J, Feuerer M (2012) Monocytes and macrophages in cancer: development and functions. Cancer Microenviron 6(2):179–191. https://doi.org/10.1007/s12307-012-0123-x
Haanen JBAG (2013) Immunotherapy of melanoma. EJC Suppl 11(2):97–105. https://doi.org/10.1016/j.ejcsup.2013.07.013
Flecknell P (2002) Replacement, reduction and refinement. Altex 19(2):73–78
Flesch IE, Kaufmann SH (1999) Effect of fetal calf serum on cytokine release by bone marrow-derived macrophages during infection with intracellular bacteria. Immunobiology 200(1):120–127. https://doi.org/10.1016/s0171-2985(99)80037-0
Bertani FR, Mozetic P, Fioramonti M, Iuliani M, Ribelli G, Pantano F, Santini D, Tonini G, Trombetta M, Businaro L, Selci S, Rainer A (2017) Classification of M1/M2-polarized human macrophages by label-free hyperspectral reflectance confocal microscopy and multivariate analysis. Sci Rep 7(1):8965. https://doi.org/10.1038/s41598-017-08121-8
Saha B, Bruneau JC, Kodys K, Szabo G (2015) Alcohol-induced miR-27a regulates differentiation and M2 macrophage polarization of normal human monocytes. J Immunol 194(7):3079–3087. https://doi.org/10.4049/jimmunol.1402190
Soncin I, Sheng J, Chen Q, Foo S, Duan K, Lum J, Poidinger M (2018) The tumour microenvironment creates a niche for the self-renewal of tumour-promoting macrophages in colon adenoma. Nat Commun 9(1):582. https://doi.org/10.1038/s41467-018-02834-8
Schlecker E, Stojanovic A, Eisen C, Quack C, Falk CS, Umansky V, Cerwenka A (2012) Tumor-infiltrating monocytic myeloid-derived suppressor cells mediate CCR17-dependent recruitment of regulatory T cells favoring tumor growth. J Immunol 189(12):5602–5611. https://doi.org/10.4049/jimmunol.1201018
Grossman JG, Nywening TM, Belt BA, Panni RZ, Krasnick BA, DeNardo DG, Hawkins WG, Goedegebuure SP, Linehan DC, Fields RC (2018) Recruitment of CCR18(+) tumor associated macrophage to sites of liver metastasis confers a poor prognosis in human colorectal cancer. Oncoimmunology 7(9):e1470729. https://doi.org/10.1080/2162402x.2018.1470729
Sun LX, Li WD, Lin ZB, Duan XS, Xing EH, Jiang MM, Yang N, Qi HH, Sun Y, Li M, Niu YD, Lu J (2015) Cytokine production suppression by culture supernatant of B16F10 cells and amelioration by Ganoderma lucidum polysaccharides in activated lymphocytes. Cell Tissue Res 360(2):379–389. https://doi.org/10.1007/s00441-014-2083-6
Bardi GT, Smith MA, Hood JL (2018) Melanoma exosomes promote mixed M1 and M2 macrophage polarization. Cytokine 105:63–72. https://doi.org/10.1016/j.cyto.2018.02.002
Zhang F, Wang H, Wang X, Jiang G, Liu H, Zhang G, Wang H, Fang R, Bu X, Cai S, Du J (2016) TGF-β induces M2-like macrophage polarization via SNAIL-mediated suppression of a pro-inflammatory phenotype. Oncotarget 7(32):52294–52306. https://doi.org/10.18632/oncotarget.10561
Wan YY, Flavell RA (2008) TGF-beta and regulatory T cell in immunity and autoimmunity. J Clin Immunol 28(6):647–659. https://doi.org/10.1007/s10875-008-9251-y
Couper KN, Blount DG, Riley EM (2008) IL-10: the master regulator of immunity to infection. J Immunol 180(9):5771–5777. https://doi.org/10.4049/jimmunol.180.9.5771
Maybruck BT, Pfannenstiel LW, Diaz-Montero M, Gastman BR (2017) Tumor-derived exosomes induce CD8(+) T cell suppressors. J Immunother Cancer 5(1):65. https://doi.org/10.1186/s40425-017-0269-7
Klarquist J, Tobin K, Farhangi Oskuei P, Henning SW, Fernandez MF, Dellacecca ER, Navarro FC, Eby JM, Chatterjee S, Mehrotra S, Clark JI, Le Poole IC (2016) Ccl22 diverts T regulatory cells and controls the growth of melanoma. Cancer Res 76(21):6230–6240. https://doi.org/10.1158/0008-5472.CAN-16-0618
Chen M-L, Pittet MJ, Gorelik L, Flavell RA, Weissleder R, von Boehmer H, Khazaie K (2005) Regulatory T cells suppress tumor-specific CD8 T cell cytotoxicity through TGF-beta signals in vivo. Proc Natl Acad Sci U S A 102(2):419–424. https://doi.org/10.1073/pnas.0408197102
Jiang H, Gebhardt C, Umansky L, Beckhove P, Schulze TJ, Utikal J, Umansky V (2015) Elevated chronic inflammatory factors and myeloid-derived suppressor cells indicate poor prognosis in advanced melanoma patients. Int J Cancer 136(10):2352–2360. https://doi.org/10.1002/ijc.29297
Gerber AL, Munst A, Schlapbach C, Shafighi M, Kiermeir D, Husler R, Hunger RE (2014) High expression of FOXP3 in primary melanoma is associated with tumour progression. Br J Dermatol 170(1):103–109. https://doi.org/10.1111/bjd.12641
Baumgartner JM, Gonzalez R, Lewis KD, Robinson WA, Richter DA, Palmer BE, Wilson CC, McCarter MD (2009) Increased survival from stage IV melanoma associated with fewer regulatory T Cells. J Surg Res 154(1):13–20. https://doi.org/10.1016/j.jss.2008.04.043
Tucci M, Mannavola F, Passarelli A, Stucci LS, Cives M, Silvestris F (2018) Exosomes in melanoma: a role in tumor progression, metastasis and impaired immune system activity. Oncotarget 9(29):20826–20837. https://doi.org/10.18632/oncotarget.24846
Bland CL, Byrne-Hoffman CN, Fernandez A, Rellick SL, Deng W, Klinke DJ 2nd (2018) Exosomes derived from B16F0 melanoma cells alter the transcriptome of cytotoxic T cells that impacts mitochondrial respiration. FEBS J 285(6):1033–1050. https://doi.org/10.1111/febs.14396
Zhang L, Tao L, Shi T, Zhang F, Sheng X, Cao Y, Zheng S, Wang A, Qian W, Jiang L, Lu Y (2015) Paeonol inhibits B16F10 melanoma metastasis in vitro and in vivo via disrupting proinflammatory cytokines-mediated NF-κB and STAT3 pathways. IUBMB Life 67(10):778–788. https://doi.org/10.1002/iub.1435
Kruger-Krasagakes S, Krasagakis K, Garbe C, Diamantstein T (1995) Production of cytokines by human melanoma cells and melanocytes. Recent Results Cancer Res 139:155–168
Funding
We would like to thank Breakthrough Cancer Research who funded this research.
Author information
Authors and Affiliations
Contributions
LFT: study design, wet lab work and paper writing. ACM and PFF: study design, evaluation of results and paper writing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval and ethical standards
All animal husbandry and experimental procedures were approved and licensed by the Animal Experimentation Ethics Committee (AEEC) in University College Cork under licence 2012-047 and performed according to the Irish Cruelty to Animals Act, 1876.
Animal source
All animals were purchased from Envigo in the UK.
Cell line authentication
The B16F10 cell line was purchased from and authenticated by the Developmental Therapeutics Program, Division of Cancer Treatment and Diagnosis Tumor Repository.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tremble, L.F., Moore, A.C. & Forde, P.F. Melanoma-conditioned medium promotes cytotoxic immune responses by murine bone marrow-derived monocytes despite their expression of ‘M2’ markers. Cancer Immunol Immunother 68, 1455–1465 (2019). https://doi.org/10.1007/s00262-019-02381-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-019-02381-1