Abstract
Purpose
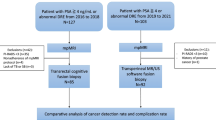
The aim of this study was to compare the complication rates between transrectal ultrasound (TRUS) systematic prostate biopsy and multiparametric magnetic resonance imaging (MRI)-TRUS fusion prostate biopsy techniques.
Materials and methods
This is a single-center retrospective study, institutional review board approved. Systematic TRUS and MRI-TRUS fusion prostate biopsy complication rates were compared in 967 men. A total of 319 patients were received systematic TRUS prostate biopsy and 648 patients underwent systematic TRUS + MRI-TRUS fusion prostate biopsy. Complications were divided into immediate (those that occurred during the hospital observation period) and late (those that occurred within 5 days after biopsy).
Results
Seventeen complications were observed in patients who received either a systematic prostate biopsy or MRI-TRUS fusion prostate biopsy. Severe complications were not observed in both groups. Among patients who underwent systematic prostate biopsy, 6 (1.9%) cases of complications were observed and between those who received MRI-TRUS fusion prostate biopsy 11 (1.7%) cases of complications after the procedure (p = 0.873) were observed, with no statistical difference between groups. Also, no statistical differences between early and late complication groups (p > 0.999) were observed.
Conclusions
The complication rates were low in both groups, with no critical clinical outcomes and no significant difference of complication rates between systematic TRUS prostate biopsy and MRI-TRUS fusion prostate biopsy techniques.





Similar content being viewed by others
References
Huang H, Wang W, Lin T, et al. (2016) Comparison of the complications of traditional 12 cores transrectal prostate biopsy with image fusion guided transperineal prostate biopsy. BMC Urol. 16(1):68
Loeb S, Vellekoop A, Ahmed HU, et al. (2013) Systematic review of complications of prostate biopsy. Eur Urol. 64(6):876–892
Baco E, Rud E, Eri LM, et al. (2016) A randomized controlled trial to assess and compare the outcomes of two-core prostate biopsy guided by fused magnetic resonance and transrectal ultrasound images and traditional 12-core systematic biopsy. Eur Urol. 69(1):149–156
Bjurlin MA, Carter HB, Schellhammer P, et al. (2013) Optimization of initical prostate biopsy in clinical practice: sampling, labeling and specimen processing. J Urol. 189(6):2039–2046
Beyersdorff D, Winkel A, Hamm B, et al. (2005) MR imaging—guided prostate biopsy with a closed MR Unit at 1.5 T: initial results. Radiology 234(2):576–581
Puech P, Rouviere O, Renard-Penna R, et al. (2013) Prostate cancer diagnosis: multiparametric MR-targeted biopsy with cognitive and transrectal US-MR fusion guidance versus systematic biopsy—prospective multicenter study. Radiology 268:461–469
Fiard G, Hohn N, Descotes JL, et al. (2013) Targeted MRI-guided prostate biopsies for the detection of prostate cancer: initial clinical experience with real-time 3-dimensional transrectal ultrasound guidance and magnetic resonance/transrectal ultrasound image fusion. Urology 81:1372–1378
Hoeks CM, Schoten MG, Bomers JG, et al. (2012) Three-Tesla magnetic resonance-guided prostate biopsy in men with increases prostete-specific antigen and repeated, negative, random, systematic, transrectal ultrasound biopsies: detection of clinically significant prostate cancers. Eur Urol. 62(5):902–909
Siddiqui MM, Rais-Bahrami S, Turkbey B, et al. (2015) Comparison of MR/ultrasound fusion-guided biopsy with ultrasound-guided biopsy for the diagnosis of prostate cancer. JAMA 313:390–397
Moore CM, Robertson NL, Arsanious N, et al. (2013) Image-guided pratate biopsy using magnetic resonance imaging-derived targets: a systematic review. Eur Urol. 63(1):125–140
Rosario DJ, Lane JA, Metcalfe C, et al. (2012) Short term outcomes of prostate biopsy in men tested for cancer by prostate specific antigen: prospective evaluation within ProtecT study. BMJ 344:d7894
Gayet M, Van Der Aa A, Beerlage HP, Schrier BP, Mulders PF (2016) Wijkstra H The value of magnetic resonance imaging and ultrasonography (MRI/US)-fusion biopsy platforms in prostate cancer detection: a systematic review. BJU Int. 117(3):392–400
Common Terminology Criteria for Adverse Events (CTCAE) version 4.0; edition 28.05.2009
Sonn GA, Natarajan S, Margolis DJ, et al. (2013) Targeted biopsy in the detection of prostate cancer using an office based magnetic resonance ultrasound fusion device. J Urol. 189:86–91
Rosenkratz AB, Verma S, Choyke P, et al. (2016) Prostate magnetic resonance imaging and magnetic resonance imaging targeted biopsy in patients with a prior negative biopsy: a consensus statement by AUA and SAR. J Urol 196(6):1613–1618
Ahmed HU, Bosaily AES, Brown LC, et al. (2017) Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet 389(10071):815–822
Borghesi M, Ahmed H, Nam R, et al. (2017) Complications after systematic random and image-guided prostate biopsy. Eur Urol 71(3):353–365
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Queiroz, M.R.G., Falsarella, P.M., Mariotti, G.C. et al. Comparison of complications rates between multiparametric magnetic resonance imaging-transrectal ultrasound (TRUS) fusion and systematic TRUS prostatic biopsies. Abdom Radiol 44, 732–738 (2019). https://doi.org/10.1007/s00261-018-1782-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-018-1782-y