Abstract
Purpose
To retrospectively determine whether hepatobiliary phase (HBP) sequence outperforms unenhanced T1-weighted imaging (uT1wI) in distinguishing the ablation margin (AM) from hepatocellular carcinoma (HCC) 24 h after thermoablation.
Material and methods
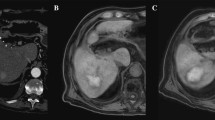
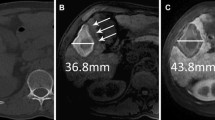
Ninety-one patients [mean age, 65.7 years; 68 M/23F] with 138 HCCs (>6 months follow-up) underwent pre- and postablation gadoxetate disodium-enhanced MRI. AM showed a hyperintense middle zone (MZ) surrounding central hypo- or hyperintense HCCs on uT1wI, and an intermediate-intense MZ encompassing central hypo- or hyperintense HCCs during HBP. The visible AM was defined as persistent MZ around HCCs, which were demarcated from MZ, or peripherally band encompassing MZ, which were not demarcated from HCC. The indefinite AM was defined as no demarcating HCCs from MZ. The ability to distinguish AM from HCC was classified as visible or indefinite on axial (ax)-uT1wI, ax-HBP, coronal (cor)-HBP, and combined all images. To investigate the AM visibility during HBP, significance of differences upon comparison of ax-uT1wI with combined images was analyzed. Preablation liver-tumor contrast ratio (LTCR) on ax-uT1wI and ax-HBP sequence is compared between the visible and indefinite AM.
Results
The McNemar test demonstrated a significant increase (p < 0.05) in visible AM from ax-uT1wI (60), to ax-HBP (70), cor-HBP (79), and combined images (83). TLCR with visible AM was significantly higher than that with indefinite AM on ax-uT1wI (0.4 vs. 0.2, p = 0.001) and ax-HBP sequence (0.9 vs. 0.6, p = 0.004).
Conclusions
HBP sequence might have higher feasibility to distinguish AM from tumor than ax-uT1wI. The TLCR value in visible AM was higher than that in indefinite AM on both ax-uT1wI and ax-HBP sequences.








Similar content being viewed by others
References
Sainani NI, Gervais DA, Mueller PR, Arellano RS (2013) Imaging after percutaneous radiofrequency ablation of hepatic tumors: Part 1, normal findings. AJR Am J Roentgenol 200:184–193
Sainani NI, Gervais DA, Mueller PR, Arellano RS (2013) Imaging after percutaneous radiofrequency ablation of hepatic tumors: Part 2, abnormal findings. AJR Am J Roentgenol 200:194–204
Kierans AS, Elazzazi M, Braga L, et al. (2010) Thermoablative treatments for malignant liver lesions: 10-year experience of MRI appearances of treatment response. AJR Am J Roentgenol 194:523–529
Kei SK, Rhim H, Choi D, et al. (2008) Local tumor progression after radiofrequency ablation of liver tumors: analysis of morphologic pattern and site of recurrence. AJR Am J Roentgenol 190:1544–1551
Mulier S, Ni Y, Jamart J, et al. (2005) Local recurrence after hepatic radiofrequency coagulation: multivariate meta-analysis and review of contributing factors. Ann Surg 242:158–171
Ng KK, Poon RT, Lo CM, et al. (2008) Analysis of recurrence pattern and its influence on survival outcome after radiofrequency ablation of hepatocellular carcinoma. Gastrointest Surg 12:183–191
Goldberg SN, Gazelle GS, Mueller PR (2000) Thermal ablation therapy for focal malignancy: a unified approach to underlying principles, techniques, and diagnostic imaging guidance. AJR Am J Roentgenol 174:323–331
Kim YS, Rhim H, Lim HK, et al. (2001) Coagulation necrosis induced by radiofrequency ablation in the liver: histopathologic and radiologic review of usual to extremely rare changes. Radiographics 31:377–390
Lee JD, Lee JM, Kim SW, Kim CS, Mun WS (2001) MR imaging-histopathological correlation of radiofrequency thermal ablation in a rabbit liver model: observation during acute and chronic stages. Korean J Radiol 2:151–158
Onishi H, Matsushita M, Murakami T, et al. (2004) MR appearances of radiofrequency thermal ablation region: histopathologic correlation with dog liver models and an autopsy case. Acad Radiol 11:1180–1189
Hyodoh H, Furuse M, Kawamoto C, et al. (2000) Microwave coagulation therapy: ex vivo comparison of MR imaging and histopathology. J Magn Reson Imaging 11:168–173
Clasen S, Pereira PL (2008) Magnetic resonance guidance for radiofrequency ablation of liver tumors. J Magn Reson Imaging 27:421–433
Rempp H, Clasen S, Pereira PL (2012) Image-based monitoring of magnetic resonance-guided thermoablative therapies for liver tumors. Cardiovasc Interv Radiol 35:1281–1294
Khankan A, Murakami T, Onishi H, et al. (2008) Hepatocellular carcinoma treated with radio frequency ablation: an early evaluation with magnetic resonance imaging. J Magn Reson Imaging 27:546–551
Koda M, Tokunaga S, Miyoshi K, et al. (2012) Assessment of ablative margin by unenhanced magnetic resonance imaging after radiofrequency ablation for hepatocellular carcinoma. Eur J Radiol 81:2730–2736
Yoon JH, Lee EJ, Cha SS, et al. (2010) Comparison of gadoxetic acid-enhanced MR imaging versus four-phase multi-detector row computed tomography in assessing tumor regression after radiofrequency ablation in subjects with hepatocellular carcinomas. J Vasc Interv Radiol 21:348–356
American College of Radiology website. (2014) Liver imaging reporting and data system. www.acr.org/ Quality-Safety/Resources/LIRADS. Accessed March 21.
Mori K, Fukuda K, Asaoka H, et al. (2009) Radiofrequency ablation of the liver: determination of ablative margin at MR imaging with impaired clearance of ferucarbotran-feasibility study. Radiology 251:557–565
Ringe KI, Wacker F, Raatschen HJ (2015) Is there a need for MRI within 24 hours after CT-guided percutaneous thermoablation of the liver? Acta Radiol 56:10–17
Braga L, Semelka RC (2005) Magnetic resonance imaging features of focal liver lesions after intervention. Top Magn Reson Imaging 16:99–106
Ringe KI, Husarik DB, Sirlin CB, Merkle EM (2010) Gadoxetate disodium-enhanced MRI of the liver: Part 1, protocol optimization and lesion appearance in the noncirrhotic liver. AJR Am J Roentgenol 195:13–28
Clasen S, Boss A, Schmidt D, et al. (2006) Magnetic resonance imaging for hepatic radiofrequency ablation. Eur J Radiol 59:140–148
Zech CJ, Bartolozzi C, Bioulac-Sage P, et al. (2013) Consensus report of the fifth international forum for liver MRI. AJR Am J Roentgenol 201:97–107
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Nobuyuki Takeyama declares that he has no conflict of interest. Sirachat Vidhyarkorn declares that she has no conflict of interest. Dong Jin Chung declares that he has no conflict of interest. Surachate Siripongsakun declares that he has no conflict of interest. Author B declares that he has no conflict of interest. Hyun J. Kim declares that she has no conflict of interest. David S.K. Lu, declares that he has no conflict of interest. Steven S. Raman declares that he has no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Takeyama, N., Vidhyarkorn, S., Chung, D.J. et al. Does hepatobiliary phase sequence qualitatively outperform unenhanced T1-weighted imaging in assessment of the ablation margin 24 hours after thermal ablation of hepatocellular carcinomas?. Abdom Radiol 41, 1942–1955 (2016). https://doi.org/10.1007/s00261-016-0796-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-016-0796-6