Abstract.
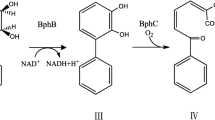
p-Cymene monooxygenase (CMO) from Pseudomonas putida F1 consists of a hydroxylase (CymA1) and a reductase component (CymA2) which initiate p-cymene (p-isopropyltoluene) catabolism by oxidation of the methyl group to p-isopropylbenzyl alcohol (p-cumic alcohol). To study the possible diverse range of substrates catalyzed by CMO, the cymA1A2 genes were cloned in an Escherichia coli pT7-5 expression system and the cells were used in transformation experiments. The tested substrates include different substituents on the aromatic ring at the 2 (ortho), 3 (meta) or 4 (para) position relative to the methyl moiety. As a result, a distinct preference was observed for substrates containing at least an alkyl or heteroatom substituent at the para-position of toluene. The conversion rate of 4-chlorotoluene or 4-methylthiotoluene to the corresponding benzyl alcohol was found to be as good as the canonical substrate, p-cymene. But 3-chlorotoluene, 4-fluorotoluene and 4-nitrotoluene were relatively poor substrates. CMO is also capable of producing styrene oxide from styrene. However, the oxidation of 4-chlorostyrene to 4-chlorostyrene oxide was by far the fastest among the substrates used in this study. The various biotransformation products were identified by a combined solid phase microextraction/gas chromatographic-mass spectrometric analytical technique.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received revision: 7 November 2000
Electronic Publication
Rights and permissions
About this article
Cite this article
Nishio, T., Patel, A., Wang, Y. et al. Biotransformations catalyzed by cloned p-cymene monooxygenase from Pseudomonas putida F1. Appl Microbiol Biotechnol 55, 321–325 (2001). https://doi.org/10.1007/s002530000584
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002530000584