Abstract
The effect of the Escherichia coli (E. coli) Rosetta (DE3) system on the expression of recombinant papain-like cysteine protease inhibitors (SnuCalCpIs) was evaluated, and the inhibition mode of the expressed inhibitor was determined. SnuCalCpI08 and SnuCalCpI17, which previously had not been expressed in the E. coli BL21 (DE3) system due to rare codons of more than 10%, were successfully expressed in E. coli Rosetta (DE3) since the strain provides tRNAs for six rare codons. Initially, both inhibitors were expressed as inclusion bodies; however, the water solubility of SnuCalCpI17 could be improved by lowering the incubation temperature, reducing the IPTG concentration, and increasing the induction time. In contrast, the other inhibitor could not be solubilized in water. To validate whether the inhibitor was expressed with correct protein folding, a papain inhibition assay was performed with SnuCalCpI17. SnuCalCpI17 showed a half-maximal inhibitory concentration (IC50) of 105.671 ± 9.857 µg/mL and a slow-binding inhibition mode against papain at pH 7.0 with a Kiapp of 75.80 μg/mL. The slow-binding inhibitor has a slow dissociation from the inhibitor-target complex, resulting in a long residence time in vivo, and thus can effectively inhibit the target at doses far below the IC50 of the inhibitor.
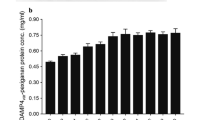
Graphical abstract

Key points
• Propeptide inhibitor (SnuCalCpI17) containing rare codons was expressed in E. coli Rosetta (DE3).
• The slow-binding inhibition was shown by plotting the apparent first-order rate constant (k obs ).
• Protein–protein interaction between SnuCalCpIs and papain was verified by docking simulation.







Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abe M, Abe K, Kuroda M, Arai S (1992) Corn kernel cysteine proteinase inhibitor as a novel cystatin superfamily member of plant origin. Eur J Biochem 209:933–937. https://doi.org/10.1111/j.1432-1033.1992.tb17365.x
Baici A (2015) Slow-onset enzyme inhibition. In: Baici A (ed) Kinetics of enzyme-modifier interactions, 1st edn. Springer, Vienna, pp 367–444. https://doi.org/10.1007/978-3-7091-1402-5_8
Billington CJ, Mason P, Magny M-C, Mort JS (2000) The slow-binding inhibition of cathepsin K by its propeptide. Biochem Biophys Res Commun 276:924–929. https://doi.org/10.1006/bbrc.2000.3553
Brégeon D, Colot V, Radman M, Taddei F (2001) Translational misreading: a tRNA modification counteracts a +2 ribosomal frameshift. Genes Dev 15:2295–2306. https://doi.org/10.1101/gad.207701
Cha S (1975) Tight-binding inhibitors—I: kinetic behavior. Biochem Pharmacol 24:2177–2185. https://doi.org/10.1016/0006-2952(75)90050-7
Chen R (2012) Bacterial expression systems for recombinant protein production: E. coli and beyond. Biotechnol Adv 30:1102–1107. https://doi.org/10.1016/j.biotechadv.2011.09.013
Cohen SN, Chang AC, Boyer HW, Helling RB (1973) Construction of biologically functional bacterial plasmids in vitro. Proc Natl Acad Sci USA 70:3240–3244. https://doi.org/10.1073/pnas.70.11.3240
Copeland RA (2000) Time-dependent inhibition. In: Copeland RA (ed) Enzymes: a practical introduction to structure, mechanism, and data analysis, 2nd edn. Wiley-VCH, New York (NY), pp 318–349. https://doi.org/10.1002/0471220639.ch10
Copeland RA, Pompliano DL, Meek TD (2006) Drug–target residence time and its implications for lead optimization. Nat Rev Drug Discov 5:730–739. https://doi.org/10.1038/nrd2082
Eggen M, Schindler J (2018) Impact and evolution of biophysics in medicinal chemistry. In: Canales A (ed) Biophysical techniques in drug discovery, 1st edn. Royal Society of Chemistry, London (UK), pp 1–22
Fox T, De Miguel E, Mort JS, Storer AC (1992) Potent slow-binding inhibition of cathepsin B by its propeptide. Biochemistry 31:12571–12576. https://doi.org/10.1021/bi00165a005
Francis DM, Page R (2010) Strategies to optimize protein expression in E. coli. Curr Protoc Protein Sci 61:5.24.21-25.24.29. https://doi.org/10.1002/0471140864.ps0524s61
Fu W, Lin J, Cen P (2007) 5-Aminolevulinate production with recombinant Escherichia coli using a rare codon optimizer host strain. Appl Microbiol Biotechnol 75:777–782. https://doi.org/10.1007/s00253-007-0887-y
Ghavim M, Abnous K, Arasteh F, Taghavi S, Nabavinia MS, Alibolandi M, Ramezani M (2017) High level expression of recombinant human growth hormone in Escherichia coli: crucial role of translation initiation region. Res Pharm Sci 12:168–175. https://doi.org/10.4103/1735-5362.202462
Gustafsson C, Govindarajan S, Minshull J (2004) Codon bias and heterologous protein expression. Trends Biotechnol 22:346–353. https://doi.org/10.1016/j.tibtech.2004.04.006
Han H-L, Kwon CW, Choi Y, Chang P-S (2022) Antifungal activity of α-helical propeptide SnuCalCpI15 derived from Calotropis procera R. Br. against food spoilage yeasts. Food Control 133:108628. https://doi.org/10.1016/j.foodcont.2021.108628
Holdgate GA, Meek TD, Grimley RL (2017) Mechanistic enzymology in drug discovery: a fresh perspective. Nat Rev Drug Discov 17:115–132. https://doi.org/10.1038/nrd.2017.219
Johnson AR (2013) In vitro and in vivo assays. In: Li JJ, Corey EJ (eds) Drug discovery: practices, processes, and perspectives, 1st edn. John Wiley & Sons, Hoboken (NJ), pp 67–98
Kane JF (1995) Effects of rare codon clusters on high-level expression of heterologous proteins in Escherichia coli. Curr Opin Biotechnol 6:494–500. https://doi.org/10.1016/0958-1669(95)80082-4
Kruger NJ (1994) The bradford method for protein quantitation. In: Walker JM (ed) Basic protein and peptide protocols, 1st edn. Humana Press, Totowa (NJ), pp 9–15
Kwon CW, Yang H, Yeo S, Park KM, Jeong AJ, Lee KW, Ye SK, Chang PS (2018) Molecular cloning and anti-invasive activity of cathepsin L propeptide-like protein from Calotropis procera R. Br. against cancer cells. J Enzyme Inhib Med Chem 33:657–664. https://doi.org/10.1080/14756366.2018.1444609
Ladisch MR, Kohlmann KL (1992) Recombinant human insulin. Biotechnol Prog 8:469–478. https://doi.org/10.1021/bp00018a001
Lu H, Tonge PJ (2010) Drug-target residence time: critical information for lead optimization. Curr Opin Chem Biol 14:467–474. https://doi.org/10.1016/j.cbpa.2010.06.176
Marko G, Jure S (2004) Slow-binding inhibition: a theoretical and practical course for students. Biochem Mol Biol Educ 32:228–235. https://doi.org/10.1002/bmb.2004.494032040358
Pandey KC, Barkan DT, Sali A, Rosenthal PJ (2009) Regulatory elements within the prodomain of falcipain-2, a cysteine protease of the malaria parasite Plasmodium falciparum. PLoS One 4:e5694. https://doi.org/10.1371/journal.pone.0005694
Rosano GL, Ceccarelli EA (2009) Rare codon content affects the solubility of recombinant proteins in a codon bias-adjusted Escherichia coli strain. Microb Cell Fact 8:1–9. https://doi.org/10.1186/1475-2859-8-41
Swinney DC (2004) Biochemical mechanisms of drug action: what does it take for success? Nat Rev Drug Discov 3:801–808. https://doi.org/10.1038/nrd1500
Tegel H, Tourle S, Ottosson J, Persson A (2010) Increased levels of recombinant human proteins with the Escherichia coli strain Rosetta(DE3). Protein Expr Purif 69:159–167. https://doi.org/10.1016/j.pep.2009.08.017
Terpe K (2006) Overview of bacterial expression systems for heterologous protein production: from molecular and biochemical fundamentals to commercial systems. Appl Microbiol Biotechnol 72:211–222. https://doi.org/10.1007/s00253-006-0465-8
Tripathi NK (2016) Production and purification of recombinant proteins from Escherichia coli. Chem Bio Eng Rev 3:116–133. https://doi.org/10.1002/cben.201600002
Ulusu Y, Şentürk SB, Kuduğ H, Gökçe İ (2016) Expression, purification, and characterization of bovine chymosin enzyme using an inducible pTOLT system. Prep Biochem Biotechnol 46:596–601. https://doi.org/10.1080/10826068.2015.1085399
Wu R, Cao J, Liu F, Yang M, Su E (2021) High-level soluble expression of phospholipase D from Streptomyces chromofuscus in Escherichia coli by combinatorial optimization. Electron J Biotechnol 50:1–9. https://doi.org/10.1016/j.ejbt.2020.12.002
Yang Q, Yu C-H, Zhao F, Dang Y, Wu C, Xie P, Sachs MS, Liu Y (2019) eRF1 mediates codon usage effects on mRNA translation efficiency through premature termination at rare codons. Nucleic Acids Res 47:9243–9258. https://doi.org/10.1093/nar/gkz710
Zheng L, Guo Z, Cao S, Zhu B (2021) Elucidating the degradation pattern of a new cold-tolerant pectate lyase used for efficient preparation of pectin oligosaccharides. Bioresour Bioprocess 8:1–11. https://doi.org/10.1186/s40643-021-00475-2
Funding
This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (NRF-2020R1A2B5B01002596) and “Cooperative Research Program for Agriculture Science & Technology Development (Project No. PJ01589402)” provided by Rural Development Administration, Republic of Korea.
Author information
Authors and Affiliations
Contributions
CWK and BC conceived and designed research. BC conducted experiments. CWK, BC, and SHY wrote the manuscript draft. PSC edited the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kwon, C.W., Chung, B., Yoo, SH. et al. Heterologous expression of a papain-like protease inhibitor (SnuCalCpI17) in the E. coli and its mode of inhibition. Appl Microbiol Biotechnol 106, 4563–4574 (2022). https://doi.org/10.1007/s00253-022-12032-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-022-12032-8